Abstracts
The epipelagic flyingfish, Hirundichthys affinis is a major artisanal fishery resource from the Northeastern coastal waters of Brazil. However, biological information about this species has been poorly documented. This paper presents data on the length-weight relationship, sex ratio, length at first sexual maturity, gonadal development and fecundity of H. affinis sampled from the coastal waters of Rio Grande do Norte, Brazil. The total body length and weight for both sexes ranged from 23.4 to 29.4 cm and from 89 to 188g, respectively. The allometric coefficient of males was 2.208 and that of females was 2.985, indicating negatively allometric growth. The sex ratio was 1M:1.6F thus differing from the expected ratio of 1:1 (χ2 = 18.63). The total length at first sexual maturity was estimated at 27.3 cm for males and 27.1 cm for females. The macroscopic characteristics of the gonads indicated four maturation stages. Histological studies of gonads of H. affinis showed seven phases of oocyte development and four phases of spermatocyte development. The mean absolute fecundity was 9092 vitelogenic oocytes. Spawning occurred during the months of March to July. The microscopic descriptions of the stages of gonad maturation indicate that the study area is an important spawning ground of H. affinis.
sex ratio; histology of gonads; fecundity; reproductive period
O peixe epipelágico voador, Hirundichthys affinis é um importante recurso de pesca artesanal das águas costeiras do Nordeste do Brasil. No entanto, as informações biológicas sobre esta espécie tem sido pouco documentada. Este trabalho apresenta dados sobre a relação peso-comprimento, proporção sexual, comprimento de primeira maturação sexual, desenvolvimento gonadal e fecundidade de H. affinis amostrados das águas costeiras do Rio Grande do Norte, Brasil. O comprimento total e o peso total de ambos os sexos variaram 23,4-29,4 cm, e de 89 a 188g, respectivamente. O coeficiente alométrico dos machos foi de 2,208 e o de fêmeas foi 2,985, indicando um crescimento alométrico negativo. A proporção entre os sexos foi 1M:1.6 F, diferindo da proporção esperada de 1:1 (χ2 = 18.63). O comprimento total da primeira maturação sexual foi estimado em 27,3 centímetros para os machos e 27,1 cm para as fêmeas. As características macroscópicas das gônadas indicaram quatro estádios de maturação. Estudos histológicos das gônadas de H. affinis mostram sete fases de desenvolvimento do ovócito e quatro fases de desenvolvimento do espermatócito. A fecundidade absoluta média foi de 9.092 ovócitos vitelogênicos. A desova ocorreu durante os meses de março a julho. As descrições microscópicas dos estágios de maturação gonadal indicam que a área de estudo é uma importante área de desova de H. affinis.
proporção sexual; histologia das gônadas; fecundidade; período reprodutivo
1 Introduction
The flying fish belongs to the family Exocoetidae and are distributed in tropical and
subtropical waters around the world (Oxenford et
al., 1995Oxenford, HA., Mahon, R. and Hunte, W., 1995. Distribution and
relative abundance of flyingfish (Exocoetidae) in the eastern Caribbean. I.
Adults. Marine Ecology Progress Series, vol. 117, p. 11-23.
http://dx.doi.org/10.3354/meps117011.
http://dx.doi.org/10.3354/meps117011...
; Monteiro et al.,
1998Monteiro, A., Vaske JUNIOR, T., LESSA, RP. and El-DEYR, ACA., 1998.
Exocoetidae (Beloniformes) off north-eastern Brazil. Cybium, vol. 22, no. 4, p.
395-403.). They are considered as commercially and ecologically important species
irrespective of their geographical distribution (Gomes et al., 1998Gomes, C., DALES, RBG. and OXENFORD, HA., 1998. The application of
RAPD markers in stock discrimination of the four-wing flyingfish, in the central
western Atlantic. Hirundichthys affinisMolecular Ecology, vol.
7, no. 8, p. 1029-1039.
http://dx.doi.org/10.1046/j.1365-294x.1998.00427.x.
http://dx.doi.org/10.1046/j.1365-294x.19...
; Parin,
2002Parin, NV., 2002. Exocoetidae: flyingfishes. In CARPENTER, KE.
(Ed.). The living marine resources of the western Central Atlantic. Rome: FAO
Species Identification Guide for Fishery Purposes and American Society of
Ichthyologists and Herepetologists Special Publication no. 5. p. 1116-1134. Bony
fishes part 1 (), vol. 2.). The flying fish, Hirundichthys affinis (Günther,
1866) is a commercially important marine fish in the western Atlantic oceanic and
coastal waters. Although the flesh of H. affinis is of a good
quality, it is frequently used as bait to capture larger predatory fish species, but
their eggs are marketed for the production of local caviar. Besides their commercial
importance, the flying fishes are crucial components in the epipelagic food chain,
where they are preferred prey of predators, such as, Coryphaena hippurus,
Thunnus albacares, Tetrapturus albidus, Makaira nigricans and
Prionace glauca which are of high commercial value (Araújo and Chellappa, 2002Araújo, AS. and Chellappa, S., 2002. Estratégia reprodutiva do peixe
voador, . Hirundichthys affinis Günther (Osteichthyes,
Exocoetidae)Revista Brasileira de Zoologia, vol. 19, no. 3, p. 691-702.
http://dx.doi.org/10.1590/S0101-81752002000300006.
http://dx.doi.org/10.1590/S0101-81752002...
; Parin, 1960Parin, NV., 1960. Flying fish (Exocoetidae) of the northwestern part
of the Pacific Ocean. Trudy Instituta Okeanologie Akademii Nauk SSS, vol. 31, p.
205-285.).
It is of fundamental importance to know the average length at which the individuals
reach sexual maturity, in order to manage an effectively exploitable population,
since they can be used to determine a maximum sustainable yield (Chen and Paloheimo, 1994Chen, Y. and Paloheimo, JE., 1994. Estimating fish length and age at
50% maturity using a logistic type model. Aquatic Sciences, vol. 56, no. 3, p.
206-219. http://dx.doi.org/10.1007/BF00879965.
http://dx.doi.org/10.1007/BF00879965...
; King, 1997King, MG., 1997. Fisheries biology, assesment and management.
Oxford: Osney Mead. 341 p. Fishing news books.). This work reports on the
length-weight relationship, sex ratio, length at first sexual maturity, gonadal
development and fecundity of H. affinis sampled from the coastal
waters of Rio Grande do Norte, Northeastern Brazil. The results of the present study
could provide data for the conservation of natural stocks of this important
epipelagic fishery resource.
2 Material and Methods
2.1 Study site and fish capture
Fish were captured monthly from the coastal waters of Caiçara do Norte, Rio Grande do Norte, Brazil, during the period of May, 2011 to April, 2012. The fish samples were captured with the help of artisanal fisherman of the region, who used motor boats and a local fishing gear popularly known as “jereré”. This simple traditional fishing gear has a triangular form which is made of wood, and serves as a support for the suspended nylon material. Fishing operations were carried out on the continental shelf at depths more than 600m, approximately 14 nautical miles from the coast of Caiçara do Norte. Fish samples were numbered, the total body length of each fish was measured to the nearest centimeter (cm) and the body weight was measured to the nearest gram (g). The samples were then dissected to identify the sex. T test was used verify the significant difference between the total length and total weight of males and females.
2.2 Length-weight relationship
The length-weight relationship was determined by the equation, W =
a Lb, where W is the total
weight (g), L is the total length (cm), a is the intercept
(initial rate of growth or condition factor) and b is
allometric coefficient (coefficient of growth or relative growth rate of fish)
(Le Cren, 1951Le Cren, ED., 1951. The length-weight relationship and seasonal
cycle in gonadal weight and condition in the perch (). Perca
fluviatilisJournal of Animal Ecology, vol. 20, no. 2, p. 201-219.
http://dx.doi.org/10.2307/1540.
http://dx.doi.org/10.2307/1540...
; Hayes et al., 1995Hayes, DB., BRODZIAK, JKT. and O’GORMAN, JB., 1995. Efficiency and
bias of estimators and sampling designs for determining length-weight
relationships of fish. Canadian Journal of Animal Science, vol. 52, p.
84-92.; Jobling, 2002Jobling, M., 2002. Environmental factors and rates of development
and growth. In HART, PJ. and REYNOLDS, JD. (Eds.). Handbook of fish biology and
fisheries. Oxford: Blackwell Publishing Ltd. p. 97-122. Fish Biology, vol. 1.
http://dx.doi.org/10.1002/9780470693803.ch5.
http://dx.doi.org/10.1002/9780470693803....
). The t test was performed
to confirm whether the value of b departed significantly from
the isometric value of 3 (Sokal and Rohlf,
1987Sokal, RR. and Rohlf, FJ., 1987. Introduction to biostatistics. 2nd
ed. New York: Freeman. 363 p.).
2.3 Sex ratio
The gonads were removed for sex identification based on the macroscopic observations of gonad characteristics (Mackie and Lewis, 2001Mackie, MC. and Lewis, PD., 2001. Assessment of gonad staging systems and other methods used in the study of the reproductive biology of the narrow-barred Spanish mackerel, Scomberomorus commerson, in Western Australia. Western Australia: Fisheries Department. p. 1-32. Fisheries Research Report, vol. 136.). Sex ratio was given as males: females (M: F), calculated using the equation: total number of males / total number of females. The chi-square (χ²) was used to verify the existence of significant differences between the sex ratio of the study species and commonly expected 1:1 sex ratio (Sokal and Rohlf, 1987Sokal, RR. and Rohlf, FJ., 1987. Introduction to biostatistics. 2nd ed. New York: Freeman. 363 p.).
2.4 Length at first sexual maturity
The first sexual maturation of the species was determined by the relative
frequency distribution of adult males and females in total length classes
registered during this study (Moreno et al.,
2005Moreno, T., Castro, JJ. and Socorro, J., 2005. Reproductive biology
of the sand smelt Cuvier, 1829 (Pisces:Atherinidae) in the central-east
Atlantic. Atherina presbyterFisheries Research, vol. 72, no. 1,
p. 121-131. http://dx.doi.org/10.1016/j.fishres.2004.06.016.
http://dx.doi.org/10.1016/j.fishres.2004...
).
2.5 Macroscopic description and histology of gonads
The characteristics used for macroscopic classification of the gonads were based
on the following external aspects: size, shape, color, presence of blood
vessels, turgidity and the space occupied in the body cavity (Murua et al., 2003Murua, H., Kraus, G., Saborido-Rey, F., Witthames, PR., Thorsen, A.
and Junquera, S., 2003. Procedures to Estimate Fecundity of Marine Fish Species
in Relation to their Reproductive Strategy. Journal of Northwest Atlantic
Fishery Science, vol. 33, p. 33-54.
http://dx.doi.org/10.2960/J.v33.a3.
http://dx.doi.org/10.2960/J.v33.a3...
). In order to avoid
possible variation in the developmental stage of oocytes due to their position
in the ovaries, histological examinations were carried out on sections from the
anterior (cephalic), middle (central), and posterior (caudal) regions of 20
ovaries in different developmental stages. These data were later compared in
order to determine whether samples taken from mid-section of the ovary were
representative of oocyte development (Yoshida,
1964Yoshida, HO., 1964. Skipjack tuna spawning in the Marquesas Islands
and Tuamotu Archipelago. Fish Bulletin, vol. 65, p. 479-488.). Histological verification indicated that gonads were at the
same stage of maturation throughout their length. Subsequently, only a central
transverse section was prepared for each gonad (n= 20).
Fragments of ovaries and testicles selected for histological analysis were fixed
in Bouin solution for 12-24 h (depending on size), washed for 24 hours in
running water to remove excess fixative and were later preserved in 70% Ethyl
alcohol. Fragments of ovaries selected for histological study were embedded in
paraffin, sectioned at 3-5 μm thickness, and stained with Hematoxylin-Eosin (HE)
and periodic acid Schiff (PAS). The histological description of the
developmental stages and classification were performed using the existing
terminology (Wallace and Selman, 1981Wallace, RA. and Selman, K., 1981. Cellular and dynamic aspects of
oocyte growth in teleosts.American Zoologist, vol. 21, p.
325-343.;
West, 1990West, G., 1990. Methods of assessing ovarian development in fishes:
a R eview.Australian Journal of Marine and Freshwater Research,
vol. 41, no. 2, p. 199-222.
http://dx.doi.org/10.1071/MF9900199.
http://dx.doi.org/10.1071/MF9900199...
; Grier and Taylor, 1998Grier, HJ. and Taylor, RG., 1998. Testicular maturation and
regression in the common snook.Journal of Fish Biology, vol.
53, no. 3, p. 521-542.
http://dx.doi.org/10.1111/j.1095-8649.1998.tb00999.x.
http://dx.doi.org/10.1111/j.1095-8649.19...
; Schulz et al., 2010Schulz, RW., de França, LR., Lareyre, JJ., Le Gac, F.,
Chiarini-Garcia, H., Nobrega, RH. and Miura, T., 2010. Spermatogenesis in fish.
General and Comparative Endocrinology, vol. 165, no. 3, p. 390-411.
http://dx.doi.org/10.1016/j.ygcen.2009.02.013. PMid:19348807
http://dx.doi.org/10.1016/j.ygcen.2009.0...
).
2.6 Fecundity
The oocytes from mature ovaries were dissociated using Gilson solution and three
sub-samples of 0.1 g were extracted and the oocytes were counted using Bogorov
counting chamber, a stereo-microscope and an ocular micrometer. Total fecundity
was estimated for the total weight of the ovaries. Fecundity = [(number of
mature oocytes in the fragments of ovary) x (total weight of ovary)] / (weight
of the fragment of the ovary) (Hunter and
Goldberg, 1980Hunter, JR. and Goldberg, SR., 1980. Spawning incidence and batch
fecundity in northern anchovy, Engraulis mordax.Fish Bulletin,
vol. 77, p. 641-652.; Murua et al.,
2003Murua, H., Kraus, G., Saborido-Rey, F., Witthames, PR., Thorsen, A.
and Junquera, S., 2003. Procedures to Estimate Fecundity of Marine Fish Species
in Relation to their Reproductive Strategy. Journal of Northwest Atlantic
Fishery Science, vol. 33, p. 33-54.
http://dx.doi.org/10.2960/J.v33.a3.
http://dx.doi.org/10.2960/J.v33.a3...
).
2.7 Gonadosomatic index (GSI) and reproductive period
The gonadosomatic index (GSI) was calculated according to Wootton et al. (1978)WOOTTON, RJ., EVANS, GW. and MILLS, LA., 1978. Annual cycle in
female three-spined sticklebacks ( L.) from an upland and lowland population.
Gasterosteus aculeatusJournal of Fish Biology, vol. 12, no.
4, p. 331-343.
http://dx.doi.org/10.1111/j.1095-8649.1978.tb04178.x.
http://dx.doi.org/10.1111/j.1095-8649.19...
. Period of breeding was determined
by the temporal relative frequency distribution of the different stages of
ovarian maturation (Martini and Fountain,
1981Martini, EE. and Fountain, R., 1981. Ovarian cycling frequency and
batch fecundity in the queenfish, : attributes representative of serial spawning
fishes. Seriphus politusFish Bulletin, vol. 79, no. 3, p.
547-559.).
3 Results
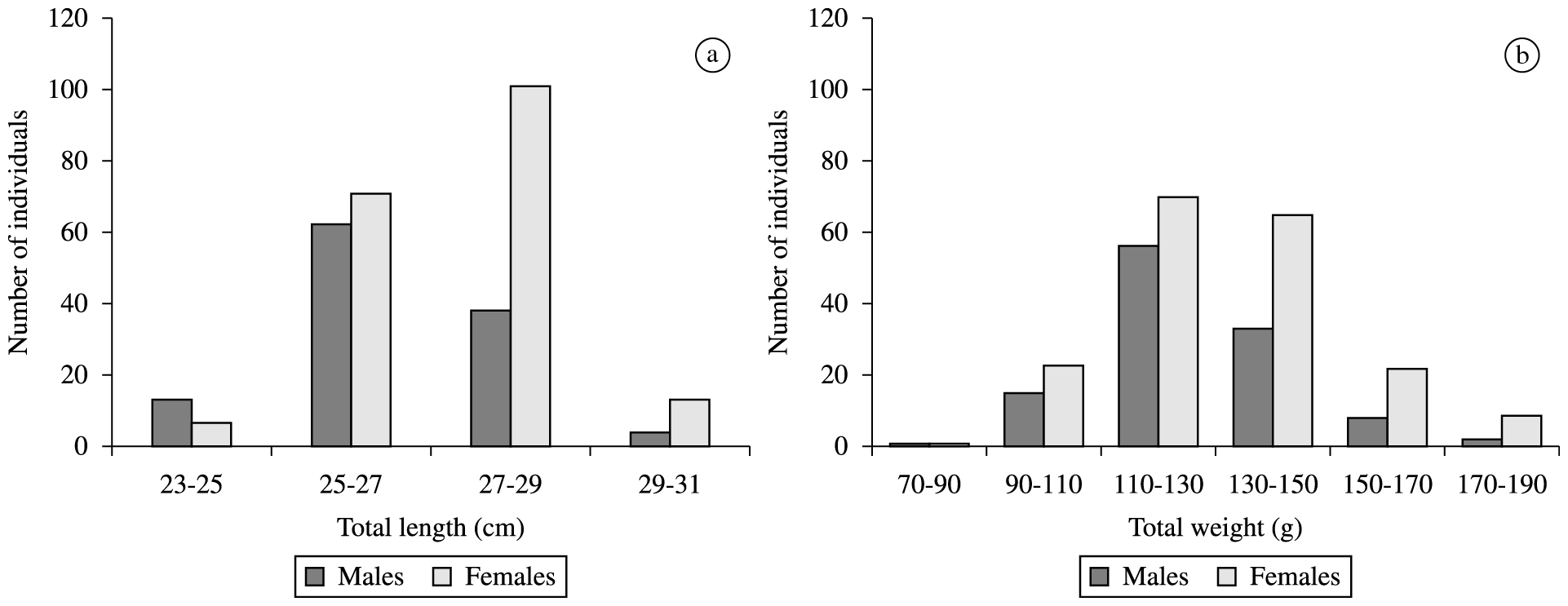
A total of 310 individuals of H. affinis were captured (117 males and 193 females). Total body lengths of all individuals of grouped sex varied from 23.4 to 29.5 cm (mean 26.7 ± 1.2), and the body weights from 70 to 189g (mean 129.2 ± 19.5). Male body lengths varied from 23.4 to 29.1 cm (mean 26.3 ± 1.2) and body weights from 89 to 184.6 g (mean 126.6 ± 17.7). Female body lengths ranged from 23.8 to 29.5 cm (mean 27.05 ± 1.1) and body weights from 70 to 189g (mean 130.8 ± 20.4). The females were larger than males with significant difference in total lengths (t = –5.093; df = 308; p < 0.05). The class distribution of total lengths and weights are shown in Figure 1.
Distribution by total body length classes (a) and (b) total weight classes of Hirundichthys affinis.
3.1 Length-weight relationship
The length-weight relationship function determined for males was Wt(g)=0,0916 Lt(cm)2.208 and for females it was Wt(g)= 0,0068 Lt(cm)2.985. The allometric coefficient of male H. affinis was 2.208 and of female was 2.985, indicating negatively allometric growth. The t-test indicated that the b-values found for both sexes departed significantly from the isometric value (3). The parameters of the equation of the length-weight relationship are presented in Table 1.
Parameter estimates of the length-weight relationship of Hirundichthys affinis (Günther, 1866) (Beloniformes/Exocoetidae).
3.2 Sex ratio
The sex ratio was 1M:1.6F thus differing from the expected ratio of 1:1. This difference was statistically significant (χ2 = 18.63). The females of H. affinis had slight predominance in the sampled population. The females predominated during June to October and the males in the month of May, with significant differences (Figure 2).
Monthly sex ratio of Hirundichthys affinis. * Significant difference of the observed sex ratio from the expected 1:1.
3.3 Length at first maturity
The total length at first sexual maturity was estimated at 27.3 cm for males and 27.1 cm for females. The males of H. affinis mature before the females, shown by the significant difference in size of gonadal maturation of both sexes (t = –5.081; df = 210; p < 0.05).
3.4 Gonad development
H. affinis had paired elongated gonads, the females with lobed ovaries and the males with flattened testes. The testicular walls were fragile when compared to the ovarian walls, and did not show much modification between the different stages of development, unlike the ovaries. The volume, coloration, thickness and blood vessels of ovaries varied according to the stage of maturation, presenting shades of light pink to dark yellow, due to the color of the mature oocytes full of yolk granules (Figures 3a, b, c, d). The testes were whitish from the beginning of maturation to the mature stage (Figures 3e, f, g, h). Table 2 presents the macroscopic characteristics during the gonadal development of H. affinis. During the study period immature fish were not captured.
Macroscopic stages of gonad development in Hirundichthys affinis: (a) ovary in the maturation process, (b) mature ovary, (c) spent ovary, (d) resting ovary, (e) testis in the early maturation process, (f) testis in the late maturation process, (g) mature testis, (h) spent. (Immature individuals were not captured during this study).
3.5 Histology and development of germ cells
3.5.1 The microscopic observations of the different phases of maturation of oocytes
The microscopic stages of ovaries showed seven phases of oocyte development constituting two stages. The first or the pre vitellogenic stage included the young germinative cell phase and the reserve stock phase. The second or vitellogenic stage included five phases: the early yolk vesicle phase, lipid vitellogenic phase, lipid and protein vitellogenic phase, complete vitellogenesis phase and hydrated oocyte phase. Oocytes in atresia and post-ovulatory follicles were also observed.
Young germ cell (Phase I): The smallest cells were found, which appeared grouped in nests placed in the ovigerous lamellae, with little cytoplasm, usually with a single central rounded nucleolus, intensely basophilic (Figure 4a).
Histological aspects of oocyte developmental stages of Hirundichthys affinis: (a) Young germ cell (ygc), (b) Perinucleolus stage or reserve stock (rs); (c) Early yolk vesicle (eyv); (d) Lipid vitellogenesis (lv); (e) Lipid and protein vitellogenesis (lpv) with yolk granules (yg) and lipid droplets (ld); (f) Oocytes with complete vitologenesis; (g) Oocytes in hydration (ho); (h) Oocytes in atresia (a). n,nucleus; nc, nucleolus; pof, post-ovulatory follicle. (a-d: scale bar = 200µm; e-h: scale bar = 50µm).
Perinucleolus stage or reserve stock (Phase II): The nucleus was in the centre, initially with one or two spherical nucleoli (intensely basophilic) and the cytoplasm was well defined and more basophilic than in the previous phase. The nucleoli become more numerous and were present in the periphery of the nucleus (Figure 4b).
Early yolk vesicle (Phase III): The basophilic nucleoli maintain their peripheral position and the cytoplasm became less basophilic. This stage was characterized by small yolk vesicles in the cytoplasm, which appeared as empty unstained vacuoles. The yolk vesicles first appeared at the periphery of the oocyte and gradually spread towards the central nucleus (Figure 4c).
Lipid vitellogenesis (Phase IV): The oocytes showed the central nucleus, cytoplasm was less basophilic than in the previous phase, and with vacuoles representing the lipid deposition (Figure 4d).
Lipid and protein vitellogenesis (Phase V): The oocytes showed the deposition of protein in the form of platelets from peripheral acidophilic cytoplasm (Figure 4e).
Oocytes with complete vitellogenesis (Phase VI): In this stage the lipidic vesicles were not observed and the protein granules were bigger in size. The basophile of the cytoplasm disappeared totally and the nucleus contracted (Figure 4f).
Oocytes in hydration (Phase VII): During this phase the oocytes were was highly modified, the cytoplasm was hydrated, which resulted in the adhesion of the yolk granules (Figure 4g).
Oocytes in atresia: the cellular turgidity was lost, with the rupture of the vitelline membrane. The vitelline granules lost their individuality and constituted an amorphous mass of acidophilic substance (Figure 4h).
Post-ovulatory follicles: the follicular cells were condensed, entering the space which was occupied by the oocytes earlier, thus constituting, a body formed by cellular chords, folded in all directions (Figure 4h).
3.5.2 The microscopic observations of the different phases of maturation of testicles
Histological analyses indicated four developmental phases of the spermatogonia of H. affinis (Figure 5). The spermatogonia were distributed near the tunica albuginea showing restricted distribution.
Histological aspects of spermatocyte developmental stages of Hirundichthys affinis: a) cross section of the testis; b) testis showing stages of spermatogenesis; c) spermatogonia (spg), spermatocyte (spc), spermatid (spt); d) spermatozoid (spz). (a-b: scale bar = 50µm; c-d: scale bar = 200µm).
Spermatogonia: These were the largest spermatogenic cells. They were rounded with large rounded vesicular nuclei. Their cytoplasm was faintly stained. Spermatocytes: These were with clear cytoplasm and relatively large nucleus and chromatin condensed in the poles. Spermatids: They were smaller than the previous germ cells with sparse cytoplasm, spherical and dense nucleus. These cells undergo modifications until they become spermatozoa. Spermatozoa: They are the smallest cells of the spermatogenic lineage, with spherical and very dense nuclei. They concentrate in the seminal lobules after breaking through the cyst wall.
3.6 Fecundity
The absolute fecundity of H. affinis varied from 7398 to 10021 with an average of 9092 (SD ±1153.2) vitellogenic oocytes.
3.7 GSI and Reproductive period
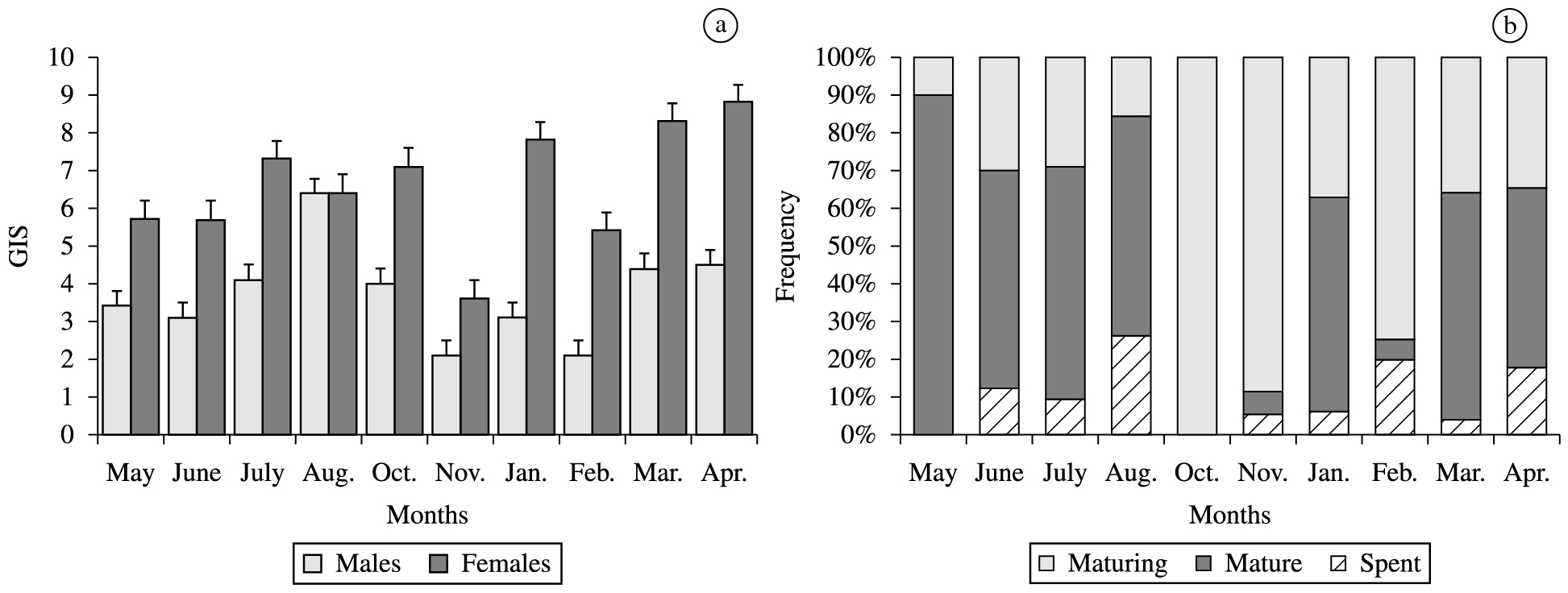
The mean monthly values of GSI for the males and females of H. affinis and the frequency of maturation stages are shown in Figure 6. The mean values of GSI varied from 1.25 to17.1 for the females and from 0.1 to 8.01 for the males. During the entire study period the presence of immature individuals were not registered. There was a predominance of maturing individuals during the period of October to February. Mature fish occurred during March to July. Variations in IGS and the monthly frequency of maturation stages demonstrate that H. affinis reproduces during the period of March to July.
a) Temporal distribution of GSI (± SE) of females and males Hirundichthys affinis; b) monthly frequency of maturity stages for pooled sex.
4 Discussion
It is of fundamental importance to have information regarding the length, weight, sex
and length at first sexual maturity, in order to manage an effectively exploitable
fishery population. Small sized immature individuals of H. affinis
were not captured during this study. The immature individuals frequently occur in
the oceanic region and only the adults migrate to the coastal waters for breeding.
However, the effect of the selectivity of the fishing gear employed cannot be ruled
out (Araújo and Chellappa, 2002Araújo, AS. and Chellappa, S., 2002. Estratégia reprodutiva do peixe
voador, . Hirundichthys affinis Günther (Osteichthyes,
Exocoetidae)Revista Brasileira de Zoologia, vol. 19, no. 3, p. 691-702.
http://dx.doi.org/10.1590/S0101-81752002000300006.
http://dx.doi.org/10.1590/S0101-81752002...
). The
absence of individuals less than 19 cm and greater than 24.5cm of the same species,
in the Caribbean coastal waters, was due to the same reason (Khokiattiwong et al., 2000Khokiattiwong, S., Mahon, R. and HUnte, W., 2000. Seasonal abundance
and reproduction of the fourwing flyingfish, , of Barbados.
Hirundichthys affinisEnvironmental Biology of Fishes, vol.
59, no. 1, p. 43-60. http://dx.doi.org/10.1023/A:1007647918255.
http://dx.doi.org/10.1023/A:100764791825...
).
The females of H. affinis in this study were larger and heavier than
the males. The females are usually heavier than the males due to their gonads which
tend to have a greater mass in relation to the testicles (Araújo and Chellappa, 2002Araújo, AS. and Chellappa, S., 2002. Estratégia reprodutiva do peixe
voador, . Hirundichthys affinis Günther (Osteichthyes,
Exocoetidae)Revista Brasileira de Zoologia, vol. 19, no. 3, p. 691-702.
http://dx.doi.org/10.1590/S0101-81752002000300006.
http://dx.doi.org/10.1590/S0101-81752002...
; Murua et al., 2003Murua, H., Kraus, G., Saborido-Rey, F., Witthames, PR., Thorsen, A.
and Junquera, S., 2003. Procedures to Estimate Fecundity of Marine Fish Species
in Relation to their Reproductive Strategy. Journal of Northwest Atlantic
Fishery Science, vol. 33, p. 33-54.
http://dx.doi.org/10.2960/J.v33.a3.
http://dx.doi.org/10.2960/J.v33.a3...
). For the same species in the Caribbean, it was
observed that the average length of males and females was not different when fish
were immature, but later the females grew faster than males and became larger and
heavier (Khokiattiwong et al., 2000Khokiattiwong, S., Mahon, R. and HUnte, W., 2000. Seasonal abundance
and reproduction of the fourwing flyingfish, , of Barbados.
Hirundichthys affinisEnvironmental Biology of Fishes, vol.
59, no. 1, p. 43-60. http://dx.doi.org/10.1023/A:1007647918255.
http://dx.doi.org/10.1023/A:100764791825...
). The
present study registered a predominance of females which could be due to the
selectivity of the fishing gear used with the interest of capturing the bigger
individuals. However, females were observed more in the months of June and July, and
this period corresponds to the peak of the reproductive activity of
H. affinis in Rio Grande do Norte, Brazil
(Araújo and Chellappa, 2002Araújo, AS. and Chellappa, S., 2002. Estratégia reprodutiva do peixe
voador, . Hirundichthys affinis Günther (Osteichthyes,
Exocoetidae)Revista Brasileira de Zoologia, vol. 19, no. 3, p. 691-702.
http://dx.doi.org/10.1590/S0101-81752002000300006.
http://dx.doi.org/10.1590/S0101-81752002...
; Araújo et al., 2011Araújo, AS., Oliveira, MR., Campos, CEC., Yamamoto, ME. and
Chellappa, S., 2011. Características morfométricas-merísticas, peso-comprimento
e maturação gonadal do peixe voador, Hirundichythys affinis
(Günther, 1866). Biota Amazônia., vol. 1, no. 2, p. 33-40.).
It is possible to determine the type of growth of a species through the allometric
coefficient (b), which is isometric when b = 3,
positive allometry when b> 3 and negative allometry when
b <3 (Jobling,
2002Jobling, M., 2002. Environmental factors and rates of development
and growth. In HART, PJ. and REYNOLDS, JD. (Eds.). Handbook of fish biology and
fisheries. Oxford: Blackwell Publishing Ltd. p. 97-122. Fish Biology, vol. 1.
http://dx.doi.org/10.1002/9780470693803.ch5.
http://dx.doi.org/10.1002/9780470693803....
). Isometric growth indicates that the body increases in all dimensions
in the same proportion during growth, whereas positive allometry indicates that the
body becomes more rotound as it increases in length, and negative allometry
indicates a slimmer body (Jobling, 2002Jobling, M., 2002. Environmental factors and rates of development
and growth. In HART, PJ. and REYNOLDS, JD. (Eds.). Handbook of fish biology and
fisheries. Oxford: Blackwell Publishing Ltd. p. 97-122. Fish Biology, vol. 1.
http://dx.doi.org/10.1002/9780470693803.ch5.
http://dx.doi.org/10.1002/9780470693803....
). In
this study, the allometric coefficient of the length-weight relationship of males
and females of H. affinis was estimated to be
lower than 3 indicating negatively allometric growth. These results are in agreement
with a study conducted for the same species in the oceanic waters of Northeastern
Brazil (Lessa and Bezerra Junior, 2004Lessa, RP. and Bezerra JUNIOr, JL., 2004. Dinâmica de populações e
avaliação dos estoques dos recursos pesqueiros do nordeste do Brasil: peixe
voador, . In Hirundichthys affinisLESSA, RT., NÓBREGA, MF. and
BEZERRA JUNIOR, JL. Dinâmica de populações e avaliação dos estoques dos recursos
pesqueiros da região nordeste do Brasil. Recife: Programa REVIZEE; Relatório
Executo. p. 39-50. vol. 2.). For
this same species in Barbados, the values of b=2.98 for males,
b= 3.03 for females and b=3.01 for grouped sex
(Khokiattiwong et al., 2000Khokiattiwong, S., Mahon, R. and HUnte, W., 2000. Seasonal abundance
and reproduction of the fourwing flyingfish, , of Barbados.
Hirundichthys affinisEnvironmental Biology of Fishes, vol.
59, no. 1, p. 43-60. http://dx.doi.org/10.1023/A:1007647918255.
http://dx.doi.org/10.1023/A:100764791825...
). The
parameters of length-weight relationship in fish could be influenced by
environmental conditions, gonadal maturity, sex, condition factor, season,
population and variations between species (Froese,
2006Froese, R., 2006. Cube law, condition factor and weight-length
relationship: history, meta-analysis and recommendations. Journal of Applied
Ichthyology, vol. 22, no. 4, p. 241-253.
http://dx.doi.org/10.1111/j.1439-0426.2006.00805.x.
http://dx.doi.org/10.1111/j.1439-0426.20...
). In this study, the gonadal development of females could have
possibly influenced the body mass.
For rational management of fishery stocks which are subjected to exploitation, it is
important to know the size at first gonadal maturation (L50), since it
provides information for determining the minimum size at capture and mesh dimensions
of the fishing gear. In this study, H. affinis presented a mean
length of 27 cm at first maturity, which is larger than that registered in the
Caribbeans, where they attained maturity below 18 cm (Khokiattiwong et al., 2000Khokiattiwong, S., Mahon, R. and HUnte, W., 2000. Seasonal abundance
and reproduction of the fourwing flyingfish, , of Barbados.
Hirundichthys affinisEnvironmental Biology of Fishes, vol.
59, no. 1, p. 43-60. http://dx.doi.org/10.1023/A:1007647918255.
http://dx.doi.org/10.1023/A:100764791825...
). In the oceanic waters of
Northeastern Brazil, the minimum size of capture which was registered for H.
affinis is 22.7 cm (Lessa and Bezerra
Junior, 2004Lessa, RP. and Bezerra JUNIOr, JL., 2004. Dinâmica de populações e
avaliação dos estoques dos recursos pesqueiros do nordeste do Brasil: peixe
voador, . In Hirundichthys affinisLESSA, RT., NÓBREGA, MF. and
BEZERRA JUNIOR, JL. Dinâmica de populações e avaliação dos estoques dos recursos
pesqueiros da região nordeste do Brasil. Recife: Programa REVIZEE; Relatório
Executo. p. 39-50. vol. 2.). The size at first maturity is not fixed and may vary
between individuals of the same species, whose populations are subject to different
environmental conditions (Wootton, 1990WOOTTON, RJ., 1990. Ecology of Teleost Fishes. 1st ed. Londres:
Chapman & Hall. 404 p.).
The length of first sexual maturation may be directly affected through changes in
the quantity of energy reserves available for gonad development (Morgan, 2004Morgan, MJ., 2004. The relationship between fish condition and the
probability of being mature in American plaice (). Hippoglossoides
platessoidesICES Journal of Marine Science, vol. 61, no. 1, p.
64-70. http://dx.doi.org/10.1016/j.icesjms.2003.09.001.
http://dx.doi.org/10.1016/j.icesjms.2003...
).
The macroscopic analyses of gonads of H. affinis showed four stages
each of ovarian and testicular development. For the same species in Barbados, five
ovarian and testicular maturation stages were observed, which included the immature
stage (Khokiattiwong et al., 2000Khokiattiwong, S., Mahon, R. and HUnte, W., 2000. Seasonal abundance
and reproduction of the fourwing flyingfish, , of Barbados.
Hirundichthys affinisEnvironmental Biology of Fishes, vol.
59, no. 1, p. 43-60. http://dx.doi.org/10.1023/A:1007647918255.
http://dx.doi.org/10.1023/A:100764791825...
). During
this study, immature individuals were not captured since the migratory stocks were
in the gonad maturation process. Possibly the younger fish were in the oceanic
region. Additionally, the fishermen tend to capture the bigger individuals for
commercial purposes. The macroscopic description of gonads shows the stages in
reproductive cycle, which could be useful to understand the gross reproductive
biology of this species.
The histological observations of this study register for the first time the presence
of hydrated oocyte phase in the mature ovaries and oocytes in atresia and
post-ovulatory follicle phase in the spent ovaries for H. afinnis.
The maturation of the oocyte ends in ovulation, with the rupture of the follicle,
liberating the oocytes in the lumen of the ovary (Lowerre-Barbieri et al., 2011Lowerre-Barbieri, SK., Ganias, K., Saborido-Rey, F., Murua, H. and
Hunter, JR., 2011. Reproductive Timing in Marine Fishes: Variability, Temporal
Scales, and Methods. Marine and Coastal Fisheries, vol. 3, no. 1, p. 71-91.
http://dx.doi.org/10.1080/19425120.2011.556932.
http://dx.doi.org/10.1080/19425120.2011....
). The ruptured follicles are known as
post-ovulatory follicles (POFs) and they remain in the ovary till they are
reabsorbed (Hunter and Macewicz, 1985Hunter, JR. and Macewicz, BJ., 1985. Measurement of spawning
frequency in multiple spawning fishes. NOAA Technical Report NMFS, vol. 36, p.
79-94.). The
females which are capable of spawning are those which appear during the reproductive
cycle with vitelogenic oocytes or with histological indicators of imminent spawning
(late migration of germinal vesicle, germinal vesicle breakdown and hydration) or
recent spawning (POFs of recent collapse). This study registered the presence of
H. affinis in the coastal waters with hydrated oocytes and
POFs, which suggests that the females use this area as spawning grounds. This
species migrates from the oceanic waters to the coastal waters of Caiçara do Norte
in Northeastern Brazil to spawn and complete its reproductive cycle.
This study describes for the first time the histological analyses of spermatogenesis
in the testes of H. affinis in the coastal waters of Rio Grande do
Norte, Brazil. Observing the distribution of spermatogonia in the germinal
compartment of testes H. affinis shows restricted distribution in
which the spermatogonia are distributed near the tunica albuginea (Grier, 1981Grier, HJ., 1981. Cellular organization of the testis and
spermatogenesis in fishes. American Zoologist, vol. 21, p.
345-357.). This type of organization is
found in teleost orders such as Atheriniformes, Cyprinodontiformes and Beloniformes
(Parenti and Grier, 2004Parenti, LR. and Grier, HJ., 2004. Evolution and phylogeny of gonad
morphology in bony fishes. Integrative and Comparative Biology, vol. 44, no. 5,
p. 333-348. http://dx.doi.org/10.1093/icb/44.5.333.
PMid:21676719
http://dx.doi.org/10.1093/icb/44.5.333...
).
This study registered an average of 9092 vitellogenic oocytes for H.
affinis, although a lower value has been registered in an earlier study
(Araújo and Chellappa, 2002Araújo, AS. and Chellappa, S., 2002. Estratégia reprodutiva do peixe
voador, . Hirundichthys affinis Günther (Osteichthyes,
Exocoetidae)Revista Brasileira de Zoologia, vol. 19, no. 3, p. 691-702.
http://dx.doi.org/10.1590/S0101-81752002000300006.
http://dx.doi.org/10.1590/S0101-81752002...
). Fecundity
is a specific reproductive tactics and is adapted to the conditions of the life
cycle of the species, varying with the growth, population density, body size, food
availability, and rate of mortality (Witthames et
al., 1995Witthames, PR., Greer Walker, M., Dinis, MT. and Whiting, CL., 1995.
The geographical variation in the potential annual fecundity od dover sole, .
Solea solea, from European shelf waters during
1991Netherlands Journal of Sea Research, vol. 34, no. 1-3, p. 45-58.
http://dx.doi.org/10.1016/0077-7579(95)90013-6.
http://dx.doi.org/10.1016/0077-7579(95)9...
; Murua et al.,
2003Murua, H., Kraus, G., Saborido-Rey, F., Witthames, PR., Thorsen, A.
and Junquera, S., 2003. Procedures to Estimate Fecundity of Marine Fish Species
in Relation to their Reproductive Strategy. Journal of Northwest Atlantic
Fishery Science, vol. 33, p. 33-54.
http://dx.doi.org/10.2960/J.v33.a3.
http://dx.doi.org/10.2960/J.v33.a3...
).
Spawning of H. affinis occurrs during the months of March to July.
For the same species, the reproductive period registered was from May to June in
Northeastern Brazil (El-Deyr, 1998El Deyr, ACA., 1998. Biologia reprodutiva do peixe voador
Hirundichtys affinis da região de Caiçara-RN. Recife: Universidade Federal Rural
de Pernambuco. 92 p. Dissertação de Mestrado em Oceanografia.; Araújo and Chellappa, 2002Araújo, AS. and Chellappa, S., 2002. Estratégia reprodutiva do peixe
voador, . Hirundichthys affinis Günther (Osteichthyes,
Exocoetidae)Revista Brasileira de Zoologia, vol. 19, no. 3, p. 691-702.
http://dx.doi.org/10.1590/S0101-81752002000300006.
http://dx.doi.org/10.1590/S0101-81752002...
; Araújo et al., 2011Araújo, AS., Oliveira, MR., Campos, CEC., Yamamoto, ME. and
Chellappa, S., 2011. Características morfométricas-merísticas, peso-comprimento
e maturação gonadal do peixe voador, Hirundichythys affinis
(Günther, 1866). Biota Amazônia., vol. 1, no. 2, p. 33-40.), and in Barbados was from
December to June, with a peak from March to June (Khokiattiwong et al., 2000Khokiattiwong, S., Mahon, R. and HUnte, W., 2000. Seasonal abundance
and reproduction of the fourwing flyingfish, , of Barbados.
Hirundichthys affinisEnvironmental Biology of Fishes, vol.
59, no. 1, p. 43-60. http://dx.doi.org/10.1023/A:1007647918255.
http://dx.doi.org/10.1023/A:100764791825...
). Breeding season may be influenced by local
climatic conditions. Information pertaining to the reproductive period of H.
affinis is important for the conservation of natural stocks of this
epipelagic fishery resource. The microscopic descriptions of the stages of gonad
maturation indicate that the study area is an important spawning ground of
H. affinis. This information could be used for adequate
management of exploitable fishery stocks of this commercially and ecologically
important fish.
Acknowledgements
This study was supported by the by the National Council for Scientific and Technological Development of Brazil (CNPq) in the form of Research grants and by the Post-Graduate Federal Agency CAPES/MEC, Brazil.
-
(With 6 figures)
References
- Araújo, AS. and Chellappa, S., 2002. Estratégia reprodutiva do peixe voador, . Hirundichthys affinis Günther (Osteichthyes, Exocoetidae)Revista Brasileira de Zoologia, vol. 19, no. 3, p. 691-702. http://dx.doi.org/10.1590/S0101-81752002000300006.
» http://dx.doi.org/10.1590/S0101-81752002000300006 - Araújo, AS., Oliveira, MR., Campos, CEC., Yamamoto, ME. and Chellappa, S., 2011. Características morfométricas-merísticas, peso-comprimento e maturação gonadal do peixe voador, Hirundichythys affinis (Günther, 1866). Biota Amazônia., vol. 1, no. 2, p. 33-40.
- Chen, Y. and Paloheimo, JE., 1994. Estimating fish length and age at 50% maturity using a logistic type model. Aquatic Sciences, vol. 56, no. 3, p. 206-219. http://dx.doi.org/10.1007/BF00879965.
» http://dx.doi.org/10.1007/BF00879965 - El Deyr, ACA., 1998. Biologia reprodutiva do peixe voador Hirundichtys affinis da região de Caiçara-RN. Recife: Universidade Federal Rural de Pernambuco. 92 p. Dissertação de Mestrado em Oceanografia.
- Froese, R., 2006. Cube law, condition factor and weight-length relationship: history, meta-analysis and recommendations. Journal of Applied Ichthyology, vol. 22, no. 4, p. 241-253. http://dx.doi.org/10.1111/j.1439-0426.2006.00805.x.
» http://dx.doi.org/10.1111/j.1439-0426.2006.00805.x - Grier, HJ. and Taylor, RG., 1998. Testicular maturation and regression in the common snook.Journal of Fish Biology, vol. 53, no. 3, p. 521-542. http://dx.doi.org/10.1111/j.1095-8649.1998.tb00999.x.
» http://dx.doi.org/10.1111/j.1095-8649.1998.tb00999.x - Grier, HJ., 1981. Cellular organization of the testis and spermatogenesis in fishes. American Zoologist, vol. 21, p. 345-357.
- Gomes, C., DALES, RBG. and OXENFORD, HA., 1998. The application of RAPD markers in stock discrimination of the four-wing flyingfish, in the central western Atlantic. Hirundichthys affinisMolecular Ecology, vol. 7, no. 8, p. 1029-1039. http://dx.doi.org/10.1046/j.1365-294x.1998.00427.x.
» http://dx.doi.org/10.1046/j.1365-294x.1998.00427.x - Hayes, DB., BRODZIAK, JKT. and O’GORMAN, JB., 1995. Efficiency and bias of estimators and sampling designs for determining length-weight relationships of fish. Canadian Journal of Animal Science, vol. 52, p. 84-92.
- Hunter, JR. and Goldberg, SR., 1980. Spawning incidence and batch fecundity in northern anchovy, Engraulis mordax.Fish Bulletin, vol. 77, p. 641-652.
- Hunter, JR. and Macewicz, BJ., 1985. Measurement of spawning frequency in multiple spawning fishes. NOAA Technical Report NMFS, vol. 36, p. 79-94.
- Jobling, M., 2002. Environmental factors and rates of development and growth. In HART, PJ. and REYNOLDS, JD. (Eds.). Handbook of fish biology and fisheries. Oxford: Blackwell Publishing Ltd. p. 97-122. Fish Biology, vol. 1. http://dx.doi.org/10.1002/9780470693803.ch5.
» http://dx.doi.org/10.1002/9780470693803.ch5 - King, MG., 1997. Fisheries biology, assesment and management. Oxford: Osney Mead. 341 p. Fishing news books.
- Khokiattiwong, S., Mahon, R. and HUnte, W., 2000. Seasonal abundance and reproduction of the fourwing flyingfish, , of Barbados. Hirundichthys affinisEnvironmental Biology of Fishes, vol. 59, no. 1, p. 43-60. http://dx.doi.org/10.1023/A:1007647918255.
» http://dx.doi.org/10.1023/A:1007647918255 - Le Cren, ED., 1951. The length-weight relationship and seasonal cycle in gonadal weight and condition in the perch (). Perca fluviatilisJournal of Animal Ecology, vol. 20, no. 2, p. 201-219. http://dx.doi.org/10.2307/1540.
» http://dx.doi.org/10.2307/1540 - Lessa, RP. and Bezerra JUNIOr, JL., 2004. Dinâmica de populações e avaliação dos estoques dos recursos pesqueiros do nordeste do Brasil: peixe voador, . In Hirundichthys affinisLESSA, RT., NÓBREGA, MF. and BEZERRA JUNIOR, JL. Dinâmica de populações e avaliação dos estoques dos recursos pesqueiros da região nordeste do Brasil. Recife: Programa REVIZEE; Relatório Executo. p. 39-50. vol. 2.
- Lowerre-Barbieri, SK., Ganias, K., Saborido-Rey, F., Murua, H. and Hunter, JR., 2011. Reproductive Timing in Marine Fishes: Variability, Temporal Scales, and Methods. Marine and Coastal Fisheries, vol. 3, no. 1, p. 71-91. http://dx.doi.org/10.1080/19425120.2011.556932.
» http://dx.doi.org/10.1080/19425120.2011.556932 - Mackie, MC. and Lewis, PD., 2001. Assessment of gonad staging systems and other methods used in the study of the reproductive biology of the narrow-barred Spanish mackerel, Scomberomorus commerson, in Western Australia. Western Australia: Fisheries Department. p. 1-32. Fisheries Research Report, vol. 136.
- Martini, EE. and Fountain, R., 1981. Ovarian cycling frequency and batch fecundity in the queenfish, : attributes representative of serial spawning fishes. Seriphus politusFish Bulletin, vol. 79, no. 3, p. 547-559.
- Monteiro, A., Vaske JUNIOR, T., LESSA, RP. and El-DEYR, ACA., 1998. Exocoetidae (Beloniformes) off north-eastern Brazil. Cybium, vol. 22, no. 4, p. 395-403.
- Morgan, MJ., 2004. The relationship between fish condition and the probability of being mature in American plaice (). Hippoglossoides platessoidesICES Journal of Marine Science, vol. 61, no. 1, p. 64-70. http://dx.doi.org/10.1016/j.icesjms.2003.09.001.
» http://dx.doi.org/10.1016/j.icesjms.2003.09.001 - Moreno, T., Castro, JJ. and Socorro, J., 2005. Reproductive biology of the sand smelt Cuvier, 1829 (Pisces:Atherinidae) in the central-east Atlantic. Atherina presbyterFisheries Research, vol. 72, no. 1, p. 121-131. http://dx.doi.org/10.1016/j.fishres.2004.06.016.
» http://dx.doi.org/10.1016/j.fishres.2004.06.016 - Murua, H., Kraus, G., Saborido-Rey, F., Witthames, PR., Thorsen, A. and Junquera, S., 2003. Procedures to Estimate Fecundity of Marine Fish Species in Relation to their Reproductive Strategy. Journal of Northwest Atlantic Fishery Science, vol. 33, p. 33-54. http://dx.doi.org/10.2960/J.v33.a3.
» http://dx.doi.org/10.2960/J.v33.a3 - Oxenford, HA., Mahon, R. and Hunte, W., 1995. Distribution and relative abundance of flyingfish (Exocoetidae) in the eastern Caribbean. I. Adults. Marine Ecology Progress Series, vol. 117, p. 11-23. http://dx.doi.org/10.3354/meps117011.
» http://dx.doi.org/10.3354/meps117011 - Parenti, LR. and Grier, HJ., 2004. Evolution and phylogeny of gonad morphology in bony fishes. Integrative and Comparative Biology, vol. 44, no. 5, p. 333-348. http://dx.doi.org/10.1093/icb/44.5.333. PMid:21676719
» http://dx.doi.org/10.1093/icb/44.5.333 - Parin, NV., 1960. Flying fish (Exocoetidae) of the northwestern part of the Pacific Ocean. Trudy Instituta Okeanologie Akademii Nauk SSS, vol. 31, p. 205-285.
- Parin, NV., 2002. Exocoetidae: flyingfishes. In CARPENTER, KE. (Ed.). The living marine resources of the western Central Atlantic. Rome: FAO Species Identification Guide for Fishery Purposes and American Society of Ichthyologists and Herepetologists Special Publication no. 5. p. 1116-1134. Bony fishes part 1 (), vol. 2.
- Sokal, RR. and Rohlf, FJ., 1987. Introduction to biostatistics. 2nd ed. New York: Freeman. 363 p.
- Schulz, RW., de França, LR., Lareyre, JJ., Le Gac, F., Chiarini-Garcia, H., Nobrega, RH. and Miura, T., 2010. Spermatogenesis in fish. General and Comparative Endocrinology, vol. 165, no. 3, p. 390-411. http://dx.doi.org/10.1016/j.ygcen.2009.02.013. PMid:19348807
» http://dx.doi.org/10.1016/j.ygcen.2009.02.013 - Wallace, RA. and Selman, K., 1981. Cellular and dynamic aspects of oocyte growth in teleosts.American Zoologist, vol. 21, p. 325-343.
- West, G., 1990. Methods of assessing ovarian development in fishes: a R eview.Australian Journal of Marine and Freshwater Research, vol. 41, no. 2, p. 199-222. http://dx.doi.org/10.1071/MF9900199.
» http://dx.doi.org/10.1071/MF9900199 - Witthames, PR., Greer Walker, M., Dinis, MT. and Whiting, CL., 1995. The geographical variation in the potential annual fecundity od dover sole, . Solea solea, from European shelf waters during 1991Netherlands Journal of Sea Research, vol. 34, no. 1-3, p. 45-58. http://dx.doi.org/10.1016/0077-7579(95)90013-6.
» http://dx.doi.org/10.1016/0077-7579(95)90013-6 - WOOTTON, RJ., EVANS, GW. and MILLS, LA., 1978. Annual cycle in female three-spined sticklebacks ( L.) from an upland and lowland population. Gasterosteus aculeatusJournal of Fish Biology, vol. 12, no. 4, p. 331-343. http://dx.doi.org/10.1111/j.1095-8649.1978.tb04178.x.
» http://dx.doi.org/10.1111/j.1095-8649.1978.tb04178.x - WOOTTON, RJ., 1990. Ecology of Teleost Fishes. 1st ed. Londres: Chapman & Hall. 404 p.
- Yoshida, HO., 1964. Skipjack tuna spawning in the Marquesas Islands and Tuamotu Archipelago. Fish Bulletin, vol. 65, p. 479-488.
Publication Dates
-
Publication in this collection
Jan-Mar 2015
History
-
Received
04 July 2013 -
Accepted
20 Aug 2013