ABSTRACT
Ducrosia anethifolia has been recommended as a remedy for neurological disorders. However, the anticonvulsant effects of D. anethifolia essential oil (DAEO) and its major constituent α-pinene have not yet been clarified.
Methods:
A rat model of pentylenetetrazole (PTZ)-induced convulsions was used. Oxidant and antioxidant parameters were assayed in the temporal lobe.
Results:
The data showed that DAEO (50, 100 and 200 mg/kg, i.p.) and α-pinene (0.2 and 0.4 mg/kg i.p.) delayed the initiation time, and reduced the duration of myoclonic and tonic-clonic seizures following PTZ injection. The PTZ produced oxidative stress so that malondialdehyde and hydrogen peroxide levels were increased and catalase and peroxidase activity decreased. Pretreatment with DAEO and α-pinene significantly inhibited the above-mentioned enzymatic changes in PTZ-treated animals.
Conclusion:
The results suggest that α-pinene, at teast in part, was responsible for the induction of the anticonvulsant and antioxidant effects of DAEO in rats.
Keywords:
Pentytenetetrazote; seizures; oxidative stress
RESUMO
A Ducrosia anethifolia tem sido recomendada como remédio para os distúrbios neurológicos. No entanto, os efeitos anticonvulsivantes do óleo essencial de Ducrosia anethifolia (DAEO) e do seu principal constituinte atfa-pineno (α-pineno) ainda não foram clarificados.
Métodos:
Foi utilizado um modelo de rato de convulsões induzidas por pentilenotetrazol (PTZ). Os parâmetros oxidante e antioxidante foram ensaiados no lobo temporal do cérebro.
Resultados:
Os dados mostraram que DAEO (50, 100 e 200 mg / kg, i.p.) e α-pineno (0,2 e 0,4 mg / kg i.p.) retardaram o tempo de iniciação e reduziram a duração das crises mioclônicas e tônico-clônicas após a injeção de PTZ. O PTZ produziu estresse oxidativo, de modo que os níveis de malondialdeído (MDA) e de peróxido de hidrogênio aumentaram e a atividade da catalase e da peroxidase diminuiu. O pré-tratamento com DAEO e α-pineno inibiu significativamente as alterações enzimáticas mencionadas em animais tratados com PTZ.
Conclusão:
O resultado sugere que α-pineno, peto menos em parte, é responsável peta indução dos efeitos anticonvulsivantes e antioxidantes da DAEO em ratos.
Palavras-chave:
Ducrosia anethifolia; α-pinene; Pentilenotetrazol; Crise; Estresse oxidativo
Epilepsy is one of the oldest conditions known to man and is the third most common neurological disorder after stroke and Alzheimer's disease. Approximately 1% of the world's population suffers from epilepsy11. Löscher W. Current status and future directions in the pharmacotherapy of epilepsy. Trends Pharmacol Sci. 2002 Mar;23(3):113-8. https://doi.org/10.1016/S0165-6147(00)01974-X
https://doi.org/10.1016/S0165-6147(00)01...
. Anti-epileptic drugs are usually the first choice of treatment for epilepsy but approximately one-third of people with epilepsy do not respond to the drugs. Anti-epileptic drugs do not cure epilepsy, but can prevent seizures from occurring. Recently, it has been proposed that seizures and status epilepticus may be associated with oxidative stress22. Barros L, Ferreira MJ, Queiros B, Ferreira IC, Baptista P. Total phenols, ascorbic acid, β-carotene and lycopene in Portuguese wild edible mushrooms and their antioxidant activities. Food Chem. 2007;103(2):413-9. https://doi.org/10.1016/j.foodchem.2006.07.038
https://doi.org/10.1016/j.foodchem.2006....
. Oxidative stress, due to the increase in the activity of glutamatergic transmitters, plays a crucial role in the induction of neuronal cell death33. Rauca C, Zerbe R, Jantze H. Formation of free hydroxyl radicals after pentylenetetrazol-induced seizure and kindling. Brain Res. 1999 Nov;847(2):347-51. https://doi.org/10.1016/S0006-8993(99)02084-3
https://doi.org/10.1016/S0006-8993(99)02...
. Since the brain utilizes the greatest amount of oxygen compared with other organs, it is particularly at risk of oxidative stress44. Mariani E, Polidori MC, Cherubini A, Mecocci P. Oxidative stress in brain aging, neurodegenerative and vascular diseases: an overview. J Chromatogr B Analyt Technol Biomed Life Sci. 2005 Nov;827(1):65-75. https://doi.org/10.1016/j.jchromb.2005.04.023
https://doi.org/10.1016/j.jchromb.2005.0...
.
Experimental models of epilepsy have been developed to find the basic mechanisms of epileptic seizures and new therapeutic approaches. The chemical kindling induced by the pentylenetetrazole (PTZ) is one of the most-widely used models for the induction of convulsions in animals.
Medicinal plants have recently become a major target in the search for new drugs and have led to compounds to treat epilepsy accompanied by oxidative stress55. Golechha M, Bhatia J, Ojha S, Arya DS. Hydroalcoholic extract of Emblica officinalis protects against kainic acid-induced status epilepticus in rats: evidence for an antioxidant, anti-inflammatory, and neuroprotective intervention. Pharm Biol. 2011 Nov;49(11):1128-36. https://doi.org/10.3109/13880209.2011.571264
https://doi.org/10.3109/13880209.2011.57...
,66. Mehla J, Reeta KH, Gupta P, Gupta YK. Protective effect of curcumin against seizures and cognitive impairment in a pentylenetetrazole-kindled epileptic rat model. Life Sci. 2010 Nov;87(19-22):596-603. https://doi.org/10.1016/j.lfs.2010.09.006
https://doi.org/10.1016/j.lfs.2010.09.00...
. Ducrosia anethifolia Boiss, known in Persian as Moshgak, Roshgak, and Moshkbu, belongs to the Apiaceae family. It is one of the three species of Iranian Ducrosia growing wild in southeastern Iran, in the mountainous regions of the Kerman province77. Mozaffarian V. A dictionary of Iranian plant names: Latin, English, Persian. City: Farhang Mo'aser; 1996.. In Iranian traditional Medicine, the whole herb—especially its aerial parts—has been used as an analgesic for headache, backache, as well as for the treatment of colic, and colds. It is also used to relax the body and mind, allowing a restful sleep88. Haghi G, Safaei A, Safari J. Extraction and determination of the main components of the essential oil of Ducrosia anethifolia by GC and GC/MS. Iran J Pharm Res. 2004;3(suppl 2):90-91.. Furthermore, antianxiolytic effects of D. anethifolia essential oil (DAEO) have been reported99. Hajhashemi V, Rabbani M, Ghanadi A, Davari E. Evaluation of antianxiety and sedative effects of essential oil of Ducrosia anethifolia in mice. Clinics (São Paulo). 2010;65(10):1037-42. https://doi.org/10.1590/S1807-59322010001000020
https://doi.org/10.1590/S1807-5932201000...
. The antioxidant, antimicrobial, antimycobacterial, antifungal, and central nervous system depressant effects of this plant and other species of Ducrosia have been reported in pharmacological and biological studies1010. Stavri M, Mathew KT, Bucar F, Gibbons S. Pangelin, an antimycobacterial coumarin from Ducrosia anethifolia. Planta Med. 2003 Oct;69(10):956-9. https://doi.org/10.1055/s-2003-45109
https://doi.org/10.1055/s-2003-45109...
. Phytochemical studies of DAEO revealed that aliphatic aldehydes and other monoterpene hydrocarbons such as limonene, citronellal, terpinolene, myrcene, α-pinene, pulegone, p-cymene and coumarins such as pangelin are the main components of D. anethifolia aerial parts1111. Mostafavi A, Afzali D, Mirtadzadini S. Chemical composition of the essential oil of Ducrosia anethifolia (DC.) Boiss. from Kerman Province in Iran. J Essent Oil Res. 2008;20(6):509-12. https://doi.org/10.1080/10412905.2008.9700073
https://doi.org/10.1080/10412905.2008.97...
. High performance liquid chromatography (HPLC) analysis of DAEO indicated the presence of terpenoids such as α-pinene as one of the major components. Terpenes constitute the major portion of the essential oils and, somehow, are responsible for the medicinal plant's pharmacological activities such as antinociceptive, anti-inflammatory and anticonvulsant effects1212. Guilhon CC, Raymundo LJ, Alviano DS, Blank AF, Arrigoni-Blank MF, Matheus ME et al. Characterisation of the anti-inflammatory and antinociceptive activities and the mechanism of the action of Lippia gracilis essential oil. J Ethnopharmacol. 2011 May;135(2):406-13. https://doi.org/10.1016/j.jep.2011.03.032
https://doi.org/10.1016/j.jep.2011.03.03...
.
It has been reported that α-pinene has anticonvulsant and antioxidant properties1313. Miyazawa M, Yamafuji C. Inhibition of acetylcholinesterase activity by bicyclic monoterpenoids. J Agric Food Chem. 2005 Mar;53(5):1765-8. https://doi.org/10.1021/jf040019b
https://doi.org/10.1021/jf040019b...
. However, there is no scientific information to validate the anticonvulsant activity of this plant in experimental animals. Therefore, the present study was designed to determine the possible effects of DAEO, and its major component α-pinene, on PTZ-induced seizure and brain oxidative stress in male rats.
METHODS
Animals
Adult male Wistar rats weighting 200-250g were prepared from the Animal House of Shahid Bahonar University of Kerman. The animals were housed in a room with photoperiod control (a 12-hour light/dark cycle) and temperature (22 ± 2°C). Food and water was available ad libitum. All experimental procedures were approved by the Animal Research Ethics Committee of the Kerman Neuroscience Research Center, Kerman, Iran (EC/95).
Drugs
Pentylenetetrazole, α-pinene and diazepam were purchased from Sigma-Aldrich Co. The drugs were dissolved in a saline solution (0.9%) and injected intraperitoneally (i.p.) in a volume of 1 ml/kg of the rat's body weight.
Plant material
Fresh aerial parts (leaves and flowers) of D. anethifolia were collected, in July, from the Lalehzar mountainous area in Kerman province, Iran, at an altitude of 2,800 m. The voucher specimens were deposited at the herbarium of Shahid Bahonar University of Kerman (Code number: 1371). The material was dried at room temperature and used for distillation. The essential oil was isolated by hydrodistillation of the fresh aerial parts for 4 hours, and then dried over anhydrous sodium sulfate 14 and stored in a refrigerator (4°C).
Acute toxicity
Seven rats were treated with the DAEO (500 mg/kg, i.p.) and the mortality and morbidity were determined.
PTZ-induced seizures
Pentylenetetrazole (80 mg/kg, i.p.) was injected to induce convulsions in rats. Diazepam (2 mg/kg, i.p.) and DAEO (25, 50, 100 and 200 mg/kg, i.p.) and α-pinene (0.2 and 0.4 mg/kg, i.p.) were administered 30 minutes before receiving PTZ. The seizure parameters were precisely monitored for 40 minutes after each PTZ injection in all groups. The following parameters were measured using a stopwatch in seconds, and behaviors were recorded with a CD camera.
The resultant seizures were classified according to the modified Racine scale1414. Racine RJ. Modification of seizure activity by electrical stimulation. II. Motor seizure. Electroencephalogr Clin Neurophysiol. 1972 Mar;32(3):281-94. https://doi.org/10.1016/0013-4694(72)90177-0
https://doi.org/10.1016/0013-4694(72)901...
as follows:
-
Stage 0: no response.
-
Stage 1: ear and facial twitching.
-
Stage 2: myoclonic jerks without rearing.
-
Stage 3: myoclonic jerks, rearing.
-
Stage 4: turning over onto side position, tonic-clonic seizures.
-
Stage 5: turning over onto back position, generalized tonic-clonic seizures.
-
Latency: the time between PTZ injection and the onset of seizures1515. Akamatsu N, Fueta Y, Endo Y, Tamagawa A, Yuhi T, Uozumi T et al., editors. The therapeutic effects of high-frequency transcranial magnetic stimulation on pentylenetetrazol-induced status epilepticus in rats. Int Congr Ser. 2005 Mar;1278423-6. https://doi.org/10.1016/j.ics.2004.11.130
https://doi.org/10.1016/j.ics.2004.11.13... . -
Duration: the time interval from the onset to termination of seizures or death of the animal.
-
Percent of death: the number of rats that died after PTZ injection among the rats of a particular group.
-
Protection percentage: the number of rats that responded to the test1616. Abbasnejad M, Keramat B, Mahani E, Rezaeezade-Roukerd M. Effect of hydro-methanolic extract of sour orange flowers, Citrus aurantium, on pentylentetrazole induced seizure in male rats. Majallah-i Danishgah-i Ulum-i Pizishki-i Babul. 2012;14(5):20-8.. P%=1-(nt/Nt) (nc/NC) ×100.
Biochemical measurements
After behavioral assessment, the animals were euthanized under deep anesthesia, and the temporal lobes of the brains were dissected and stored at −80°C until the day of assay.
Brain lipid peroxidation
Lipid peroxidation products such as malondialdehyde (MDA) are considered to be reliable indicators of oxidative damage1717. Hodges DM, DeLong JM, Forney CF, Prange RK. Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta. 1999;207(4):604-11. https://doi.org/10.1007/s004250050524
https://doi.org/10.1007/s004250050524...
. Temporal lobe tissue (0.5 g) was homogenized in 10 mg of 0.1% trichloroacetic acid; the homogenate was centrifuged at 15,000 rpm for 15 minutes to 1.0 mg aliquot of the supernatant; and 4.0 mg of 0.5% thiobarbituric acid in 20% trichloroacetic acid was added. The mixture was heated at 95°C for 30 minutes and then cooled in an ice bath. After centrifugation (10,000 rpm for 10 minutes), the absorbance of the supernatant was recorded at 532 nm (Biochrom WPA Biowave II UV/Visible Spectrophotometer). The thiobarbituric acid reactive substances content was calculated according to its extinction coefficient of 155mM−1cm−1 and expressed in units (U). One ‘U’ is defined as μmol of MDA formed min−1mg−1 protein.
Hydrogen peroxide
Hydrogen peroxide (H2O2) was determined by the method described by Velikova et al., (2000). Temporal lobe tissue (0.5 g) was finely ground with trichloroacetic acid (5 ml of 0.1 % w/v) and centrifuged at 10,000 × g for 15 minutes. Phosphate buffer (0.5 ml, pH 7.0) and 1 ml potassium iodide were added to the 0.5 ml supernatant. Its absorbance was recorded at 390 nm after overtaxing using a UV visible spectrophotometer.
Total soluble proteins
Total proteins were estimated using the Bradford method and bovine serum albumin was used as the standard.
Antioxidant enzymes activities
Temporal lobe tissue (0.5 g) was finely ground under chilled conditions in 3 ml of phosphate buffer (50 mM with pH 7.5) for the extraction of antioxidant enzymes. Centrifugation of the mixture was performed at 10,000 × g for 10 minutes at 4°C. The supernatant was recentrifuged at 15,000 × g for 10 minutes and the resultant extract stored at −20°C for determination of the activity of antioxidant enzymes.
Evaluation of catalase activity
The activity of catalase (CAT) was estimated by monitoring the decrease in absorbance of H2O2 within 30 seconds at 240 nm. The assay solution contained 50 mM potassium phosphate buffer (pH 7.0) and 15 mM H2O2 and 100 μl enzyme extract1818. Dhindsa RS, Matowe W. Drought tolerance in two mosses: correlated with enzymatic defence against lipid peroxidation. J Exp Bot. 1981;32(1):79-91. https://doi.org/10.1093/jxb/32.1.79
https://doi.org/10.1093/jxb/32.1.79...
.
Evaluation of peroxidase activity
Peroxidase (POD) activity was assayed according to the method of Plewa et al.1919. Plewa MJ, Smith SR, Wagner ED. Diethyldithiocarbamate suppresses the plant activation of aromatic amines into mutagens by inhibiting tobacco cell peroxidase. Mutat Res. 1991 Mar;247(1):57-64. https://doi.org/10.1016/0027-5107(91)90033-K
https://doi.org/10.1016/0027-5107(91)900...
, based on the amount of tetraguaiacol absorbed after formation, by oxidation, of guaiacol catalyzed by this enzyme in 3 minutes at a wavelength of 470 nm using an extinction coefficient of tetraguaiacol, ε = 26.6 mM−1cm−1.
HPLC analysis
The obtained essential oil was analyzed using HPLC (Agilent Technologies, 1200 Infinity series, USA) equipped with a 1260 Infinity Quaternary Pump and a 1260 Infinity Variable Wavelength Detector. An Agilent 1260 Infinity Manual Injector fitted with a 20 μL sample loop was used to introduce the samples. The analytes were separated on a Restek Ultra C18 (250 mm × 4.6 mm, 5μm) column (USA). Chromatograms were processed by an Agilent HPLC Chem Station (Rev. B.04.03).
Statistical analysis
The data are expressed as mean ± SEM. Comparison between groups was made by analysis of variance followed by the Tukey test. Differences between experimental groups of each point with p < 0.05 were considered statistically significant.
RESULTS
Acute toxicity
The essential oil of D. anethifolia has shown no mortality up to a dose of 500 mg/kg. However, we used doses of 25, 50, 100 and 200 mg/kg in this study.
Anticonvulsant activity assessment
Effect of DAEO on PTZ-induced seizures
The essential oil showed dose-dependent effects against PTZ-induced seizures. It could significantly reduce the number of convulsing animals. Pretreatment with DAEO (50, 100 and 200 mg/kg) and α-pinene (0.2 and 0.4 mg/kg) significantly reduced mortality rate and attenuated PTZ-induced seizures (Table).
The effect of Ducrosia anethifolia essential oil and α-pinene on pentylenetetrazole (PTZ) induced seizures in rats.
Effect of DAEO and α-pinene on the onset of seizure
The DAEO (50, 100 and 200 mg/kg, i.p.) significantly delayed the onset of PTZ-induced seizures. However, diazepam and α-pinene had no significant effects on the onset of seizure (Figure 1).
The effect of Ducrosia anethifolia essential oil (DAEO) and α-pinene on the onset of seizure of pentylenetetrazole (PTZ)-induced convulsion in rats.
Histograms represent mean ± SEM for seven animals. ***p < 0.001, **p < 0.01, versus PTZ group by analysis of variance with Tukey's post hoc test.
Effect of DAEO and α-pinene on the duration of seizure
The essential oil at doses of 50, 100 and 200 mg/ kg, α-pinene (0.2 and 0.4 mg/kg) and diazepam could significantly alter the duration of seizures in PTZ-treated rats. However, 25 mg/kg of DAEO had no effect on the duration of seizures (Figure 2).
The effect of Ducrosia anethifolia essential oil (DAEO) and α-pinene on the duration of PTZ-induced seizure in Rats.
Histograms represent mean ± SEM (n=7). ****p < 0.001 versus PTZ-treated group. The data were analyzed by one-way analysis of variance with Tukey's post hoc test.
Biochemical measurements
MDA levels
The PTZ injection significantly increased brain temporal lobe MDA levels, which were significantly attenuated by DAEO (50, 100, 200 mg/kg) and α-pinene (0.2 and 0.4 mg/kg) (Figure 3).
The effect of Ducrosia anethifolia essential oil (DAEO) and α-pinene on the temporal lobe MDA levels in the PTZ seizure models.
Data represent means ± SEM (n=7), ***p < 0.001 and *p < 0.05 compared with nontreated normal rats. +++p < 0.001 versus PTZ-injected group.
H2O2 levels
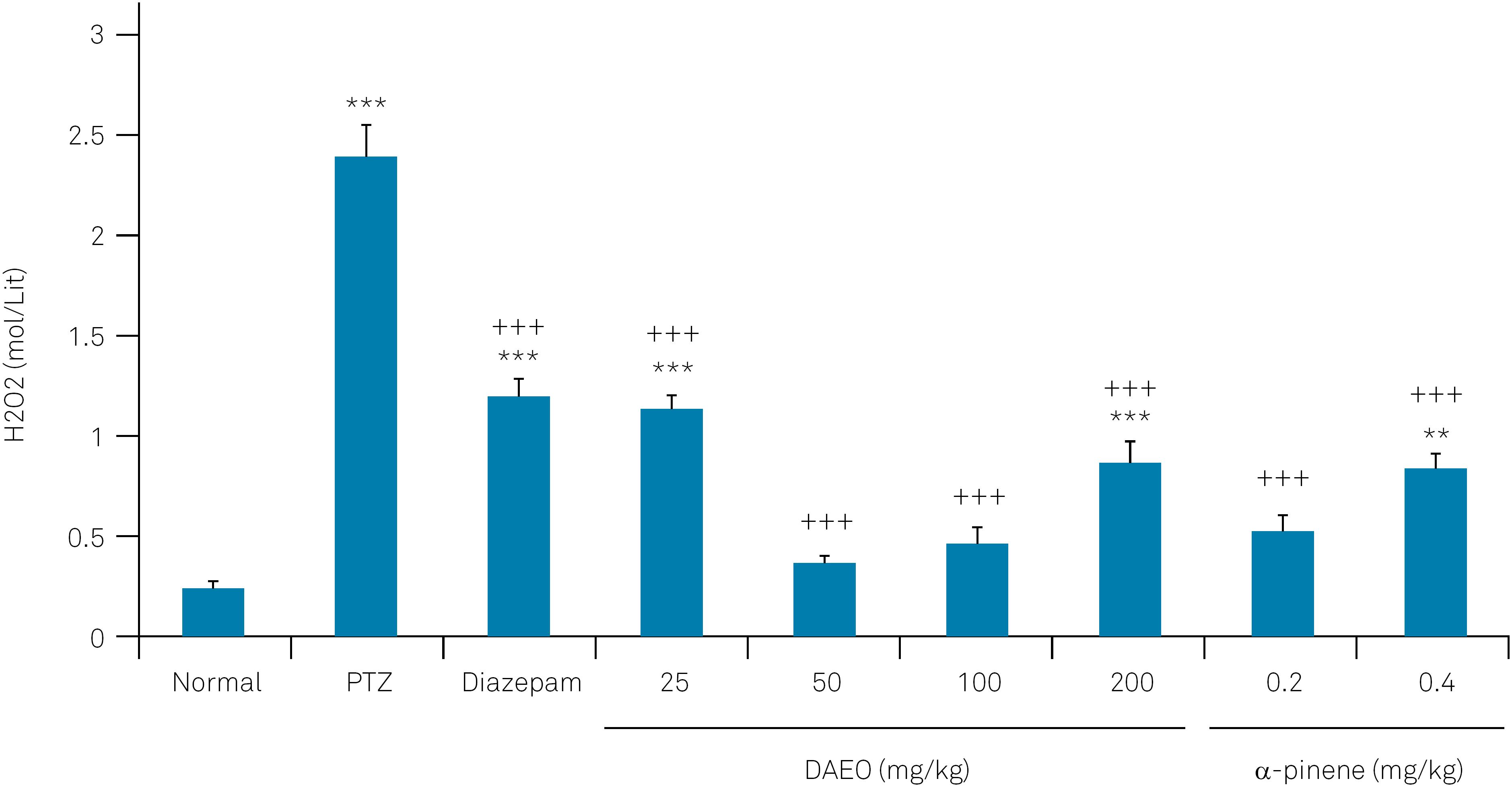
The PTZ-treated rats showed a significant increase in H2O2 levels in the temporal lobe. Alternatively, DAEO, α-pinene and diazepam significantly decreased PTZ-induced H2O2 production (Figure 4).
The effect of Ducrosia anethifolia essential oil (DAEO) and α-pinene on the temporal lobe H2O2 levels in the rat PTZ seizure models.
Data represent means ± SEM (n=7), ***p < 0.001 and **p < 0.01 compared with the control nontreated groups. +++p < 0.001 compared with PTZ-treated animals.
The effect of DAEO and α-pinene on brain CAT and POD activities in PTZ-treated animals
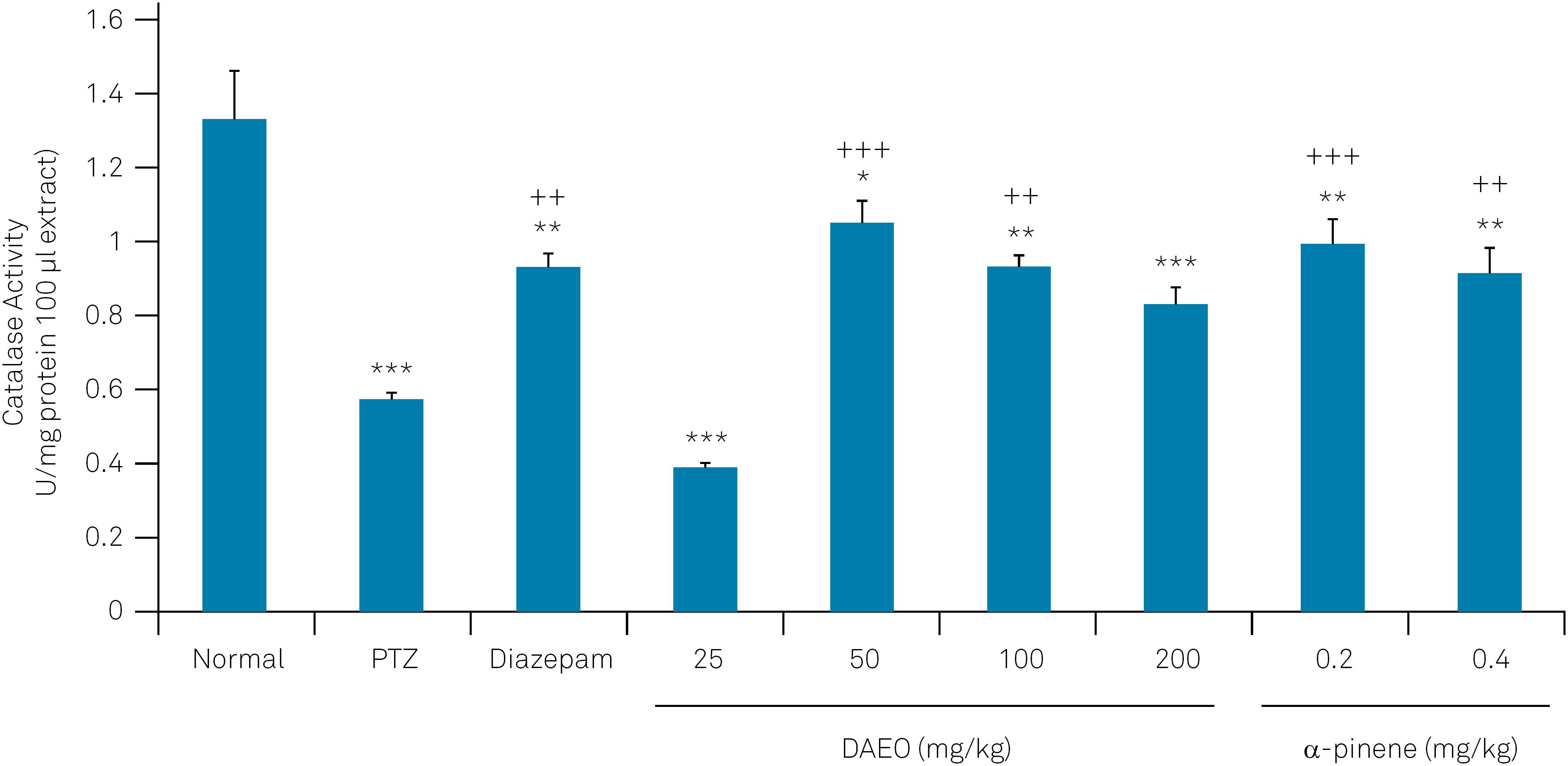
The brain CAT and POD activities were significantly decreased following PTZ administration. However, DAEO (50,100 and 200 mg/kg), α-pinene (0.2 and 0.4 mg/kg) and diazepam could prevent the effect of PTZ on CAT and POD activities (Figure 5 and 6).
The effect of Ducrosia anethifolia essential oil (DAEO) and α-pinene on catalase activity in the temporal lobe of the brain in the PTZ seizure model.
Data represent means ± SEM (n=7), ****p < 0.001, **p < 0.01 and *p < 0.05 compared with the control non-treated groups. +++p < 0.001 and ++p < 0.01 compared with PTZ-treated animals.
The effect of Ducrosia anethifolia essential oil (DAEO) and α-pinene on peroxidase activity in the temporal lobe in the PTZ seizure model.
Data represent means ± SEM (n=7), ****p < 0.001, **p < 0.01 compared with the control untreated groups. +++p < 0.001 and ++p < 0.01 compared with PTZ-treated animals.
HPLC analysis
According to the obtained HPLC spectrum of essential oil of D. anethifolia, there was a major peak following retention times (min): 6.950 (Figure 7). The peak for the reference standard, α-pinene, appeared at the retention time (min) of 6.866.
DISCUSSION
In the present work, the effects of DAEO and α-pinene were studied. Ducrosia anethifolia essential oil and α-pinene were initially evaluated in a behavioral study that gave a good indication of the reduction of seizures. Additionally, the results showed that DAEO and α-pinene were able to significantly decrease the oxidative stress factors after seizures induced by PTZ.
The PTZ method is a valid model of convulsion for the study of generalized myoclonic (absence) seizures2020. De Deyn PP, D'Hooge R, Marescau B, Pei YQ. Chemical models of epilepsy with some reference to their applicability in the development of anticonvulsants. Epilepsy Res. 1992 Jul;12(2):87-110. https://doi.org/10.1016/0920-1211(92)90030-W
https://doi.org/10.1016/0920-1211(92)900...
,2121. Eraković V, Župan G, Varljen J, Simonić A. Pentylenetetrazol-induced seizures and kindling: changes in free fatty acids, superoxide dismutase, and glutathione peroxidase activity. Neurochem Int. 2003 Jan;42(2):173-8. https://doi.org/10.1016/S0197-0186(02)00070-0
https://doi.org/10.1016/S0197-0186(02)00...
,2222. Kandratavicius L, Balista PA, Lopes-Aguiar C, Ruggiero RN, Umeoka EH, Garcia-Cairasco N et al. Animal models of epilepsy: use and limitations. Neuropsychiatr Dis Treat. 2014 Sep;10:1693-705. https://doi.org/10.2147/NDT.S50371
https://doi.org/10.2147/NDT.S50371...
. It has been demonstrated that oxidative stress resulting from free radicals plays a critical role in the genesis of epilepsy and in post-seizure neuronal death. The brain is particularly susceptible to oxidative stress damage44. Mariani E, Polidori MC, Cherubini A, Mecocci P. Oxidative stress in brain aging, neurodegenerative and vascular diseases: an overview. J Chromatogr B Analyt Technol Biomed Life Sci. 2005 Nov;827(1):65-75. https://doi.org/10.1016/j.jchromb.2005.04.023
https://doi.org/10.1016/j.jchromb.2005.0...
,2323. Uttara B, Singh AV, Zamboni P, Mahajan RT. Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol. 2009 Mar;7(1):65-74. https://doi.org/10.2174/157015909787602823
https://doi.org/10.2174/1570159097876028...
,2424. Bouayed J, Rammal H, Soulimani R. Oxidative stress and anxiety: relationship and cellular pathways. Oxid Med Cell Longev. 2009 Apr-Jun;2(2):63-7. https://doi.org/10.4161/oxim.2.2.7944
https://doi.org/10.4161/oxim.2.2.7944...
. Traditionally, medicinal plants with antioxidant properties have been candidates for preventing oxidative damage and epilepsy2525. Noda Y, Anzai K, Mori A, Kohno M, Shinmei M, Packer L. Hydroxyl and superoxide anion radical scavenging activities of natural source antioxidants using the computerized JES-FR30 ESR spectrometer system. Biochem Mol Biol Int. 1997 Jun;42(1):35-44. https://doi.org/10.1080/15216549700202411
https://doi.org/10.1080/1521654970020241...
. The phytochemical and HPLC analysis by Hajhashemi et al.99. Hajhashemi V, Rabbani M, Ghanadi A, Davari E. Evaluation of antianxiety and sedative effects of essential oil of Ducrosia anethifolia in mice. Clinics (São Paulo). 2010;65(10):1037-42. https://doi.org/10.1590/S1807-59322010001000020
https://doi.org/10.1590/S1807-5932201000...
showed that DAEO had a wide spectrum of bioactive compounds, and terpenoids were its major components99. Hajhashemi V, Rabbani M, Ghanadi A, Davari E. Evaluation of antianxiety and sedative effects of essential oil of Ducrosia anethifolia in mice. Clinics (São Paulo). 2010;65(10):1037-42. https://doi.org/10.1590/S1807-59322010001000020
https://doi.org/10.1590/S1807-5932201000...
. The antinociceptive, anticonvulsant and anti-inflammatory properties of monoterpenes, such as α-pinene, carvacrol, γ-terpineol, citronellol and linalool have been reported2626. Liapi C, Anifandis G, Chinou I, Kourounakis AP, Theodosopoulos S, Galanopoulou P. Antinociceptive properties of 1,8-Cineole and beta-pinene, from the essential oil of Eucalyptus camaldulensis leaves, in rodents. Planta Med. 2007 Oct;73(12):1247-54. https://doi.org/10.1055/s-2007-990224
https://doi.org/10.1055/s-2007-990224...
,2727. Guimarães AG, Quintans JS, Quintans LJ Jr. Monoterpenes with analgesic activity—a systematic review. Phytother Res. 2013 Jan;27(1):1-15. https://doi.org/10.1002/ptr.4686
https://doi.org/10.1002/ptr.4686...
. Pentylenetetrazole induces convulsion by inhibiting GABA receptors-chloride channel complexes. It appears that the inhibitory effect of DAEO against PTZ-induced seizure may occur through the rise of the convulsion threshold in the brain via the stimulation of GABA receptors2828. Kasture VS, Chopde CT, Deshmukh VK. Anticonvulsive activity of Albizzia lebbeck, Hibiscus rosa sinesis and Butea monosperma in experimental animals. J Ethnopharmacol. 2000 Jul;71(1-2):65-75. https://doi.org/10.1016/S0378-8741(99)00192-0
https://doi.org/10.1016/S0378-8741(99)00...
. The α-pinenes, as major components of DAEO, have a promoting effect on GABAA receptors and increase the postsynaptic GABA-dependent chloride flows, as well as being a potent inhibitor of acetylcholinesterase1313. Miyazawa M, Yamafuji C. Inhibition of acetylcholinesterase activity by bicyclic monoterpenoids. J Agric Food Chem. 2005 Mar;53(5):1765-8. https://doi.org/10.1021/jf040019b
https://doi.org/10.1021/jf040019b...
. The major inhibitory neurotransmitter in the brain is GABA and the inhibition of its neurotransmission has been thought to be a critical factor in epilepsy2929. Gale K. GABA and epilepsy: basic concepts from preclinical research. Epilepsia. 1992;33 Suppl 5:S3-12.. The standard antiepileptic drugs, phenobarbital and diazepam, can induce their antiepileptic effects by enhancing GABA neurotransmission. Glutamate and glutamatergic receptors are located in both central and peripheral nervous systems and may be responsible for most of the excitatory neurotransmission.
In addition to GABA dysregulation, it has been indicated that excitatory amino acids are also involved in the initiation and propagation of seizures3030. Melo MS, Sena LC, Barreto FJ, Bonjardim LR, Almeida JR, Lima JT et al. Antinociceptive effect of citronellal in mice. Pharm Biol. 2010 Apr;48(4):411-6. https://doi.org/10.3109/13880200903150419
https://doi.org/10.3109/1388020090315041...
,3131. Quintans-Júnior LJ, Melo MS, Sousa DP, Araújo AA, Onofre AC, Gelain DP et al. Antinociceptive effects of citronellal in formalin-, capsaicin-, and glutamate-induced orofacial nociception in rodents and its action on nerve excitability. J Orofac Pain. 2010;24(3):305-12.. Citronellal, citronellol, myrcene and β-pinene, the DAEO monoterpenes, have NMDA receptor antagonist activities and can protect neurons against overstimulation3030. Melo MS, Sena LC, Barreto FJ, Bonjardim LR, Almeida JR, Lima JT et al. Antinociceptive effect of citronellal in mice. Pharm Biol. 2010 Apr;48(4):411-6. https://doi.org/10.3109/13880200903150419
https://doi.org/10.3109/1388020090315041...
,3232. Quintans-Júnior LJ, Melo MS, Sousa DP, Araujo AA, Onofre AC, Gelain DP et al. Antinociceptive effects of citronellal in formalin-, capsaicin-, and glutamate-induced orofacial nociception in rodents and its action on nerve excitability. J Orofac Pain. 2010;24(3):305-12. Duplicata da 31. Activation of NMDA receptors generally increases intracellular calcium influx, which raises neuronal excitation and excitability mainly via stimulation of cAMP-dependent signaling molecules including adenylyl cyclases and protein kinase A3333. Crump FT, Dillman KS, Craig AM. cAMP-dependent protein kinase mediates activity-regulated synaptic targeting of NMDA receptors. J Neurosci. 2001 Jul;21(14):5079-88. https://doi.org/10.1523/JNEUROSCI.21-14-05079.2001
https://doi.org/10.1523/JNEUROSCI.21-14-...
. Especially, it has been reported that down-regulation of the cAMP-response element-binding protein is correlated with the suppression of epileptic seizures3434. Zhu X, Han X, Blendy JA, Porter BE. Decreased CREB levels suppress epilepsy. Neurobiol Dis. 2012 Jan;45(1):253-63. https://doi.org/10.1016/j.nbd.2011.08.009
https://doi.org/10.1016/j.nbd.2011.08.00...
. It has been reported that linalool, a DAEO constituent compound, exerts a considerable anticonvulsant activity in a rat model of PTZ-kindling via modulation of glutamatergic currents3535. Elisabetsky E, Brum LF, Souza DO. Anticonvulsant properties of linalool in glutamate-related seizure models. Phytomedicine. 1999 May;6(2):107-13. https://doi.org/10.1016/S0944-7113(99)80044-0
https://doi.org/10.1016/S0944-7113(99)80...
. In addition, linalool inhibits adenylate cyclase in chick retinas3636. Sampaio LF, Maia JG, Parijós AM, Souza RZ, Barata LE. Linalool from rosewood (Aniba rosaeodora Ducke) oil inhibits adenylate cyclase in the retina, contributing to understanding its biological activity. Phytother Res. 2012 Jan;26(1):73-7. https://doi.org/10.1002/ptr.3518
https://doi.org/10.1002/ptr.3518...
. Thus, DAEO anticonvulsant capacity, at least in part, is mediated by modulation of intracellular second messengers such as calcium and glutamate. However, additional studies are still required to clarify this important issue in more details.
In the present study, PTZ-induced seizures could increase the levels of oxidative stress indicators such as MDA and H2O2, and decrease the activities of antioxidant enzymes, CAT and POD3737. Gaweł S, Wardas M, Niedworok E, Wardas P. Malondialdehyde (MDA) as a lipid peroxidation marker. Wiadomosci lekarskie (Warsaw, Poland: 1960). 2004;57(9-10):453-5.,3838. Ho E, Karimi Galougahi K, Liu CC, Bhindi R, Figtree GA. Biological markers of oxidative stress: applications to cardiovascular research and practice. Redox Biol. 2013 Oct;1(1):483-91. https://doi.org/10.1016/j.redox.2013.07.006
https://doi.org/10.1016/j.redox.2013.07....
. It has been demonstrated that the use of free radical scavengers in the treatment of epilepsy provides an important perspective that will be the driving force for future drug design of novel antiepileptics3939. Azam F, Prasad MV, Thangavel N. Targeting oxidative stress component in the therapeutics of epilepsy. Curr Top Med Chem. 2012;12(9):994-1007. https://doi.org/10.2174/156802612800229224
https://doi.org/10.2174/1568026128002292...
. Pretreatment with DAEO and α-pinene could prevent the seizures and thus decrease oxidative stress. The data showed a dose-dependent effect of DAEO against seizure-induced oxidative stress in experimental models of seizures.
Potential antioxidant therapy that includes either natural antioxidants or agents is capable of augmenting the functions of these enzymes4040. Bast A, Haenen GR, Doelman CJ. Oxidants and antioxidants: state of the art. Am J Med. 1991 Sep;91(3 3C):2S-13S. https://doi.org/10.1016/0002-9343(91)90278-6
https://doi.org/10.1016/0002-9343(91)902...
. Earlier reports have shown that the natural drugs like DAEO have antioxidant properties because of the presence of α-pinene, citronellal, γ-terpinene, myrcene and limonene4141. Ciftci O, Ozdemir I, Tanyildizi S, Yildiz S, Oguzturk H. Antioxidative effects of curcumin, β-myrcene and 1,8-cineole against 2,3,7,8-tetrachlorodibenzo-p-dioxin-induced oxidative stress in rats liver. Toxicol Ind Health. 2011 Jun;27(5):447-53. https://doi.org/10.1177/0748233710388452
https://doi.org/10.1177/0748233710388452...
.
Taken together, the data suggest that DAEO and α-pinene have antiepileptic activities. This effect may be due to their antioxidant properties and possible activation of GABAA receptors. Our experiment contributes to our knowledge of the pharmacology of D. anethifolia (Boiss).
-
Support: Shahid Bahonar University and Kerman Neuroscience Research Center (KNRC/94).
References
-
1Löscher W. Current status and future directions in the pharmacotherapy of epilepsy. Trends Pharmacol Sci. 2002 Mar;23(3):113-8. https://doi.org/10.1016/S0165-6147(00)01974-X
» https://doi.org/10.1016/S0165-6147(00)01974-X -
2Barros L, Ferreira MJ, Queiros B, Ferreira IC, Baptista P. Total phenols, ascorbic acid, β-carotene and lycopene in Portuguese wild edible mushrooms and their antioxidant activities. Food Chem. 2007;103(2):413-9. https://doi.org/10.1016/j.foodchem.2006.07.038
» https://doi.org/10.1016/j.foodchem.2006.07.038 -
3Rauca C, Zerbe R, Jantze H. Formation of free hydroxyl radicals after pentylenetetrazol-induced seizure and kindling. Brain Res. 1999 Nov;847(2):347-51. https://doi.org/10.1016/S0006-8993(99)02084-3
» https://doi.org/10.1016/S0006-8993(99)02084-3 -
4Mariani E, Polidori MC, Cherubini A, Mecocci P. Oxidative stress in brain aging, neurodegenerative and vascular diseases: an overview. J Chromatogr B Analyt Technol Biomed Life Sci. 2005 Nov;827(1):65-75. https://doi.org/10.1016/j.jchromb.2005.04.023
» https://doi.org/10.1016/j.jchromb.2005.04.023 -
5Golechha M, Bhatia J, Ojha S, Arya DS. Hydroalcoholic extract of Emblica officinalis protects against kainic acid-induced status epilepticus in rats: evidence for an antioxidant, anti-inflammatory, and neuroprotective intervention. Pharm Biol. 2011 Nov;49(11):1128-36. https://doi.org/10.3109/13880209.2011.571264
» https://doi.org/10.3109/13880209.2011.571264 -
6Mehla J, Reeta KH, Gupta P, Gupta YK. Protective effect of curcumin against seizures and cognitive impairment in a pentylenetetrazole-kindled epileptic rat model. Life Sci. 2010 Nov;87(19-22):596-603. https://doi.org/10.1016/j.lfs.2010.09.006
» https://doi.org/10.1016/j.lfs.2010.09.006 -
7Mozaffarian V. A dictionary of Iranian plant names: Latin, English, Persian. City: Farhang Mo'aser; 1996.
-
8Haghi G, Safaei A, Safari J. Extraction and determination of the main components of the essential oil of Ducrosia anethifolia by GC and GC/MS. Iran J Pharm Res. 2004;3(suppl 2):90-91.
-
9Hajhashemi V, Rabbani M, Ghanadi A, Davari E. Evaluation of antianxiety and sedative effects of essential oil of Ducrosia anethifolia in mice. Clinics (São Paulo). 2010;65(10):1037-42. https://doi.org/10.1590/S1807-59322010001000020
» https://doi.org/10.1590/S1807-59322010001000020 -
10Stavri M, Mathew KT, Bucar F, Gibbons S. Pangelin, an antimycobacterial coumarin from Ducrosia anethifolia. Planta Med. 2003 Oct;69(10):956-9. https://doi.org/10.1055/s-2003-45109
» https://doi.org/10.1055/s-2003-45109 -
11Mostafavi A, Afzali D, Mirtadzadini S. Chemical composition of the essential oil of Ducrosia anethifolia (DC.) Boiss. from Kerman Province in Iran. J Essent Oil Res. 2008;20(6):509-12. https://doi.org/10.1080/10412905.2008.9700073
» https://doi.org/10.1080/10412905.2008.9700073 -
12Guilhon CC, Raymundo LJ, Alviano DS, Blank AF, Arrigoni-Blank MF, Matheus ME et al. Characterisation of the anti-inflammatory and antinociceptive activities and the mechanism of the action of Lippia gracilis essential oil. J Ethnopharmacol. 2011 May;135(2):406-13. https://doi.org/10.1016/j.jep.2011.03.032
» https://doi.org/10.1016/j.jep.2011.03.032 -
13Miyazawa M, Yamafuji C. Inhibition of acetylcholinesterase activity by bicyclic monoterpenoids. J Agric Food Chem. 2005 Mar;53(5):1765-8. https://doi.org/10.1021/jf040019b
» https://doi.org/10.1021/jf040019b -
14Racine RJ. Modification of seizure activity by electrical stimulation. II. Motor seizure. Electroencephalogr Clin Neurophysiol. 1972 Mar;32(3):281-94. https://doi.org/10.1016/0013-4694(72)90177-0
» https://doi.org/10.1016/0013-4694(72)90177-0 -
15Akamatsu N, Fueta Y, Endo Y, Tamagawa A, Yuhi T, Uozumi T et al., editors. The therapeutic effects of high-frequency transcranial magnetic stimulation on pentylenetetrazol-induced status epilepticus in rats. Int Congr Ser. 2005 Mar;1278423-6. https://doi.org/10.1016/j.ics.2004.11.130
» https://doi.org/10.1016/j.ics.2004.11.130 -
16Abbasnejad M, Keramat B, Mahani E, Rezaeezade-Roukerd M. Effect of hydro-methanolic extract of sour orange flowers, Citrus aurantium, on pentylentetrazole induced seizure in male rats. Majallah-i Danishgah-i Ulum-i Pizishki-i Babul. 2012;14(5):20-8.
-
17Hodges DM, DeLong JM, Forney CF, Prange RK. Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta. 1999;207(4):604-11. https://doi.org/10.1007/s004250050524
» https://doi.org/10.1007/s004250050524 -
18Dhindsa RS, Matowe W. Drought tolerance in two mosses: correlated with enzymatic defence against lipid peroxidation. J Exp Bot. 1981;32(1):79-91. https://doi.org/10.1093/jxb/32.1.79
» https://doi.org/10.1093/jxb/32.1.79 -
19Plewa MJ, Smith SR, Wagner ED. Diethyldithiocarbamate suppresses the plant activation of aromatic amines into mutagens by inhibiting tobacco cell peroxidase. Mutat Res. 1991 Mar;247(1):57-64. https://doi.org/10.1016/0027-5107(91)90033-K
» https://doi.org/10.1016/0027-5107(91)90033-K -
20De Deyn PP, D'Hooge R, Marescau B, Pei YQ. Chemical models of epilepsy with some reference to their applicability in the development of anticonvulsants. Epilepsy Res. 1992 Jul;12(2):87-110. https://doi.org/10.1016/0920-1211(92)90030-W
» https://doi.org/10.1016/0920-1211(92)90030-W -
21Eraković V, Župan G, Varljen J, Simonić A. Pentylenetetrazol-induced seizures and kindling: changes in free fatty acids, superoxide dismutase, and glutathione peroxidase activity. Neurochem Int. 2003 Jan;42(2):173-8. https://doi.org/10.1016/S0197-0186(02)00070-0
» https://doi.org/10.1016/S0197-0186(02)00070-0 -
22Kandratavicius L, Balista PA, Lopes-Aguiar C, Ruggiero RN, Umeoka EH, Garcia-Cairasco N et al. Animal models of epilepsy: use and limitations. Neuropsychiatr Dis Treat. 2014 Sep;10:1693-705. https://doi.org/10.2147/NDT.S50371
» https://doi.org/10.2147/NDT.S50371 -
23Uttara B, Singh AV, Zamboni P, Mahajan RT. Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol. 2009 Mar;7(1):65-74. https://doi.org/10.2174/157015909787602823
» https://doi.org/10.2174/157015909787602823 -
24Bouayed J, Rammal H, Soulimani R. Oxidative stress and anxiety: relationship and cellular pathways. Oxid Med Cell Longev. 2009 Apr-Jun;2(2):63-7. https://doi.org/10.4161/oxim.2.2.7944
» https://doi.org/10.4161/oxim.2.2.7944 -
25Noda Y, Anzai K, Mori A, Kohno M, Shinmei M, Packer L. Hydroxyl and superoxide anion radical scavenging activities of natural source antioxidants using the computerized JES-FR30 ESR spectrometer system. Biochem Mol Biol Int. 1997 Jun;42(1):35-44. https://doi.org/10.1080/15216549700202411
» https://doi.org/10.1080/15216549700202411 -
26Liapi C, Anifandis G, Chinou I, Kourounakis AP, Theodosopoulos S, Galanopoulou P. Antinociceptive properties of 1,8-Cineole and beta-pinene, from the essential oil of Eucalyptus camaldulensis leaves, in rodents. Planta Med. 2007 Oct;73(12):1247-54. https://doi.org/10.1055/s-2007-990224
» https://doi.org/10.1055/s-2007-990224 -
27Guimarães AG, Quintans JS, Quintans LJ Jr. Monoterpenes with analgesic activity—a systematic review. Phytother Res. 2013 Jan;27(1):1-15. https://doi.org/10.1002/ptr.4686
» https://doi.org/10.1002/ptr.4686 -
28Kasture VS, Chopde CT, Deshmukh VK. Anticonvulsive activity of Albizzia lebbeck, Hibiscus rosa sinesis and Butea monosperma in experimental animals. J Ethnopharmacol. 2000 Jul;71(1-2):65-75. https://doi.org/10.1016/S0378-8741(99)00192-0
» https://doi.org/10.1016/S0378-8741(99)00192-0 -
29Gale K. GABA and epilepsy: basic concepts from preclinical research. Epilepsia. 1992;33 Suppl 5:S3-12.
-
30Melo MS, Sena LC, Barreto FJ, Bonjardim LR, Almeida JR, Lima JT et al. Antinociceptive effect of citronellal in mice. Pharm Biol. 2010 Apr;48(4):411-6. https://doi.org/10.3109/13880200903150419
» https://doi.org/10.3109/13880200903150419 -
31Quintans-Júnior LJ, Melo MS, Sousa DP, Araújo AA, Onofre AC, Gelain DP et al. Antinociceptive effects of citronellal in formalin-, capsaicin-, and glutamate-induced orofacial nociception in rodents and its action on nerve excitability. J Orofac Pain. 2010;24(3):305-12.
-
32Quintans-Júnior LJ, Melo MS, Sousa DP, Araujo AA, Onofre AC, Gelain DP et al. Antinociceptive effects of citronellal in formalin-, capsaicin-, and glutamate-induced orofacial nociception in rodents and its action on nerve excitability. J Orofac Pain. 2010;24(3):305-12. Duplicata da 31
-
33Crump FT, Dillman KS, Craig AM. cAMP-dependent protein kinase mediates activity-regulated synaptic targeting of NMDA receptors. J Neurosci. 2001 Jul;21(14):5079-88. https://doi.org/10.1523/JNEUROSCI.21-14-05079.2001
» https://doi.org/10.1523/JNEUROSCI.21-14-05079.2001 -
34Zhu X, Han X, Blendy JA, Porter BE. Decreased CREB levels suppress epilepsy. Neurobiol Dis. 2012 Jan;45(1):253-63. https://doi.org/10.1016/j.nbd.2011.08.009
» https://doi.org/10.1016/j.nbd.2011.08.009 -
35Elisabetsky E, Brum LF, Souza DO. Anticonvulsant properties of linalool in glutamate-related seizure models. Phytomedicine. 1999 May;6(2):107-13. https://doi.org/10.1016/S0944-7113(99)80044-0
» https://doi.org/10.1016/S0944-7113(99)80044-0 -
36Sampaio LF, Maia JG, Parijós AM, Souza RZ, Barata LE. Linalool from rosewood (Aniba rosaeodora Ducke) oil inhibits adenylate cyclase in the retina, contributing to understanding its biological activity. Phytother Res. 2012 Jan;26(1):73-7. https://doi.org/10.1002/ptr.3518
» https://doi.org/10.1002/ptr.3518 -
37Gaweł S, Wardas M, Niedworok E, Wardas P. Malondialdehyde (MDA) as a lipid peroxidation marker. Wiadomosci lekarskie (Warsaw, Poland: 1960). 2004;57(9-10):453-5.
-
38Ho E, Karimi Galougahi K, Liu CC, Bhindi R, Figtree GA. Biological markers of oxidative stress: applications to cardiovascular research and practice. Redox Biol. 2013 Oct;1(1):483-91. https://doi.org/10.1016/j.redox.2013.07.006
» https://doi.org/10.1016/j.redox.2013.07.006 -
39Azam F, Prasad MV, Thangavel N. Targeting oxidative stress component in the therapeutics of epilepsy. Curr Top Med Chem. 2012;12(9):994-1007. https://doi.org/10.2174/156802612800229224
» https://doi.org/10.2174/156802612800229224 -
40Bast A, Haenen GR, Doelman CJ. Oxidants and antioxidants: state of the art. Am J Med. 1991 Sep;91(3 3C):2S-13S. https://doi.org/10.1016/0002-9343(91)90278-6
» https://doi.org/10.1016/0002-9343(91)90278-6 -
41Ciftci O, Ozdemir I, Tanyildizi S, Yildiz S, Oguzturk H. Antioxidative effects of curcumin, β-myrcene and 1,8-cineole against 2,3,7,8-tetrachlorodibenzo-p-dioxin-induced oxidative stress in rats liver. Toxicol Ind Health. 2011 Jun;27(5):447-53. https://doi.org/10.1177/0748233710388452
» https://doi.org/10.1177/0748233710388452
Publication Dates
-
Publication in this collection
Feb 2019
History
-
Received
29 Nov 2017 -
Reviewed
28 Sept 2018 -
Accepted
16 Oct 2018