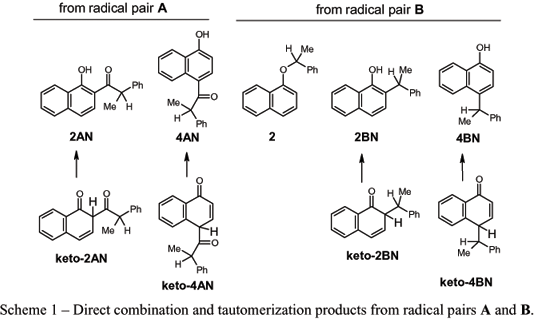
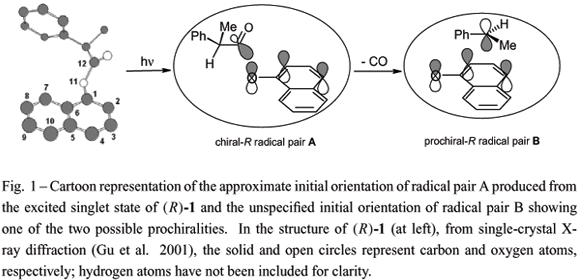
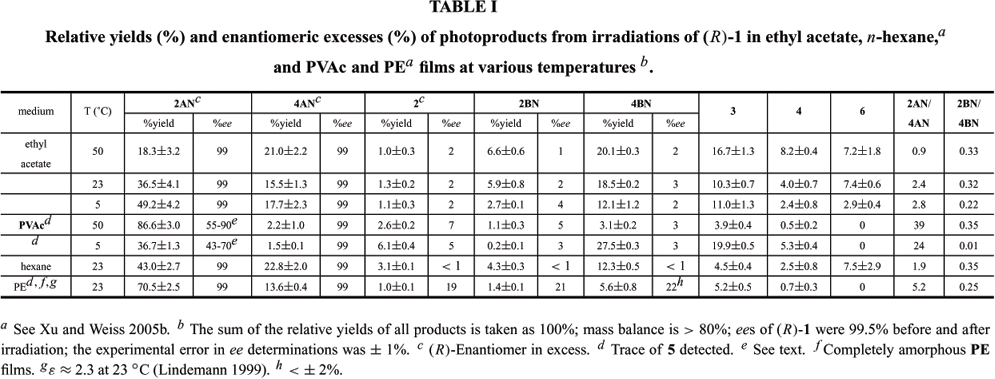
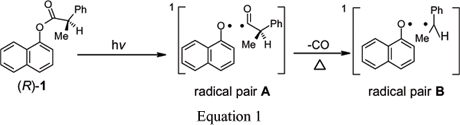
Both the regio- and stereo-chemistries of the photoreactions of 1-naphthyl (R)-2-phenylpropanoate have been investigated in poly(vinyl acetate) films in their glassy (at 5ºC) and melted (at 50ºC) states and in ethyl acetate. These results are compared with those from irradiations in polyethylene films and in n-hexane. The regioselectivity of the intermediate 1-naphthoxy/(R)-2-phenylpropanoyl radical pair combinations is much higher in both the melt and glassy states of poly(vinyl acetate) films than that in the melt state of completely amorphous polyethylene films, but the stereoselectivity of intermediate prochiral 1-naphthoxy/1-phenylethyl radical pair combinations is much lower in poly(vinyl acetate). The results emphasize the need to control the ratio between the rates of radical tumbling and translation, as well as the ratio between the rates of in-cage motions and cage-escape, if high stereo- and regio-selectivities of combination products are to be achieved. A mechanistic picture of how the radicals of the intermediate pairs are affected by and interact with the various media is advanced.
radical pair; stereoselectivity; polymer relaxation; photo-Fries