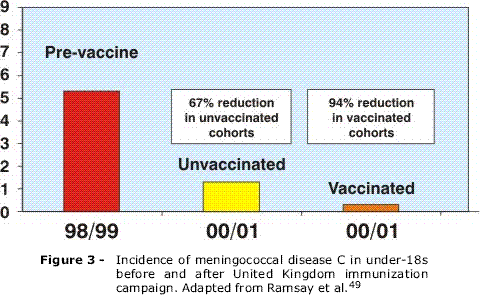
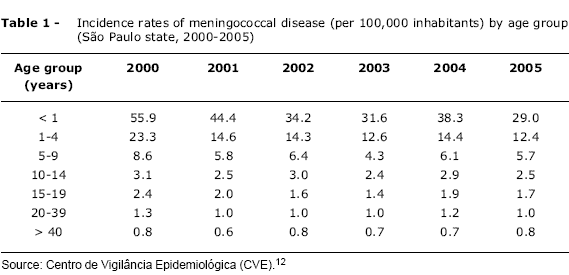
OBJECTIVE: Meningococcal disease continues to be a serious public health concern, being associated with high morbidity and mortality rates worldwide, particularly in Brazil. In addition to discussing recent changes in the global epidemiology of meningococcal disease, we also analyze the development and impact of new conjugate vaccines on the prevention of meningococcal disease, with emphasis on the different immunization strategies implemented with these vaccines. SOURCES OF DATA: MEDLINE databases were searched from 1996 to 2006, with emphasis on review articles, clinical trials and epidemiological studies. Information was also sought on the Centers for Disease Control and Prevention, Brazilian Ministry of Health and Centro de Vigilância Epidemiológica do Estado de São Paulo websites. SUMMARY OF THE FINDINGS: Five serogroups (A, B, C, W135 and Y) are responsible for virtually all cases of the disease worldwide, with marked regional and temporal differences. The new meningococcal serogroup C conjugate vaccines (MCC) offer unmistakable advantages over polysaccharide vaccines. MCC vaccines generate a more efficient and long-lasting antibody response, inducing immunologic memory and reduction of nasopharyngeal carriage. The immediate results of introducing these vaccines into immunization programs have been encouraging, with a dramatic reduction in the incidence of serogroup C disease, not only in vaccinated, but also in unvaccinated individuals (herd immunity). However, concerns have arisen regarding the long-term effectiveness of these vaccines, especially for infants vaccinated in the routine schedule. CONCLUSIONS: The reported waning of efficacy more than 1 year after routine infant immunization supports alternative schedules incorporating a booster dose of MCC vaccine given at 12-18 months of age, in order to maintain long-term protection. The recent licensure of the tetravalent meningococcal conjugate vaccine represents, at last, a real possibility of a broader protection against meningococcal disease, although the need to develop an effective vaccine against serogroup B remains.
Neisseria meningitidis; meningococcal disease; conjugate meningococcal vaccines