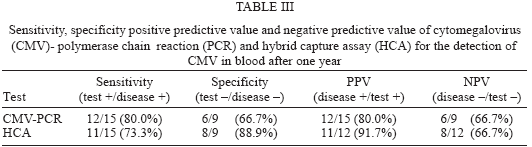
The clinical value of an in-house cytomegalovirus nested polymerase chain reaction (CMV-PCR) and a commercial molecular assay hybrid capture CMV DNA assay (HCA) was evaluated in monitoring a group of renal transplant patients for six months follow up. In this study, the sensitivity, specificity, positive predictive value, and negative predictive value of nested CMV DNA PCR assay and HCA at the beginning of the study were 70, 42.9, 46.7, 66.7, and 60, 78.6, 66.7, and 73.3% respectively. After six months, they were 80, 66.7, 80, 66.7 for CMV PCR and 73.3, 88.9, 91.7, 66.7% for HCA respectively. These results indicate that in monitoring and predicting CMV infections in renal transplant recipients, not only qualitative but also quantitative assays must be used together in order to decide the preemptive strategies.
cytomegalovirus; polymerase chain reaction; hybridization