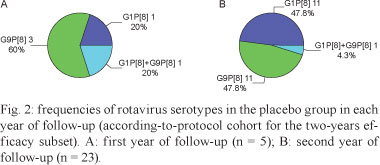
In a large Phase III trial conducted in 10 Latin American countries, the safety and efficacy of the live attenuated monovalent rotavirus vaccine RIX4414 was evaluated in 15,183 healthy infants followed up during the first two years of life. Belém was the only site in Brazil included in this multicentre trial. The study in Belém included a subset of 653 infants who were followed up until 24 months of age for protection against severe rotavirus gastroenteritis. These subjects were randomly assigned in a 1:1 ratio to receive two doses of vaccine (n = 328) or two doses of placebo (n = 325) at approximately two and four months of age. Of the 653 enrolled infants, 23 dropped out during the study period. For the combined two-year period, the efficacy of RIX4414 was 72.3% [95% confidence interval (CI) 37.5-89.1%] against severe rotavirus-related gastroenteritis, reaching a protection rate of 81.8% (95% CI 36.4-96.6%) against circulating wild-type G9 rotavirus strains. It is concluded that two doses of RIX4414 are highly efficacious against severe rotavirus gastroenteritis in Belém during the first two years of life and provide high protection against the worldwide emergence and spread of G9P[8] strains.
rotavirus; gastroenteritis; vaccine efficacy; G9P[8] strains