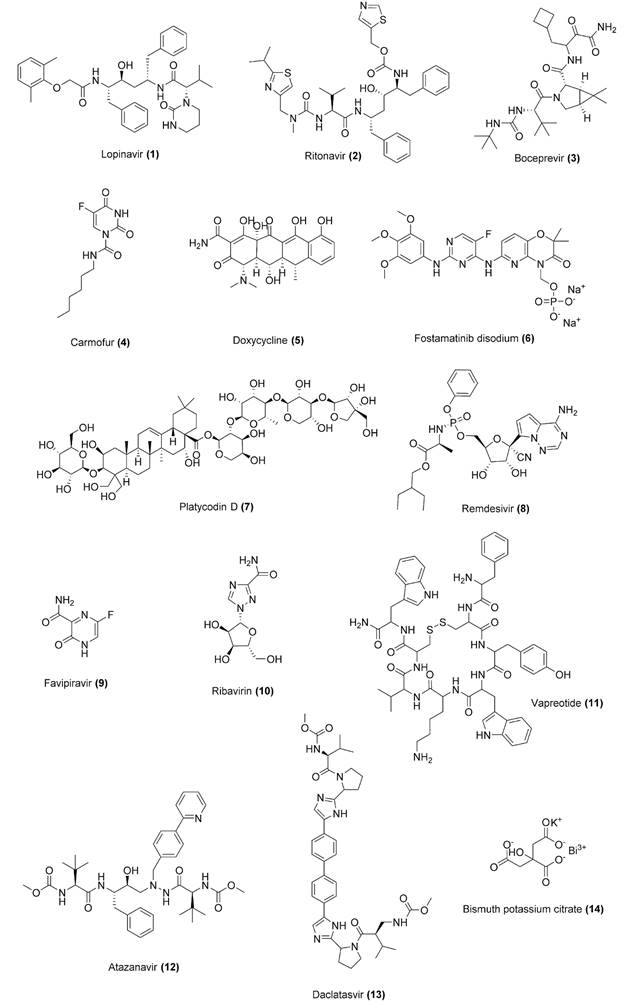
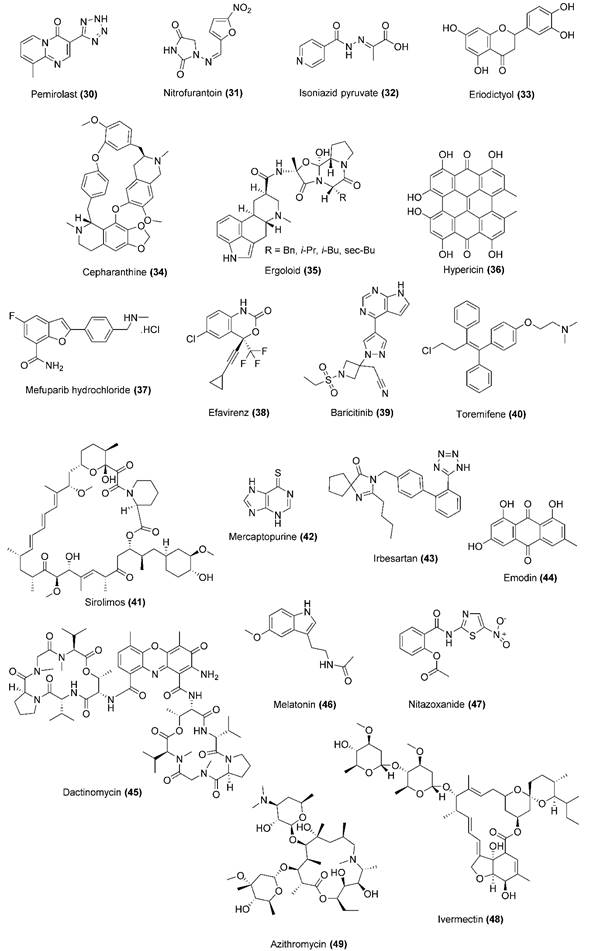
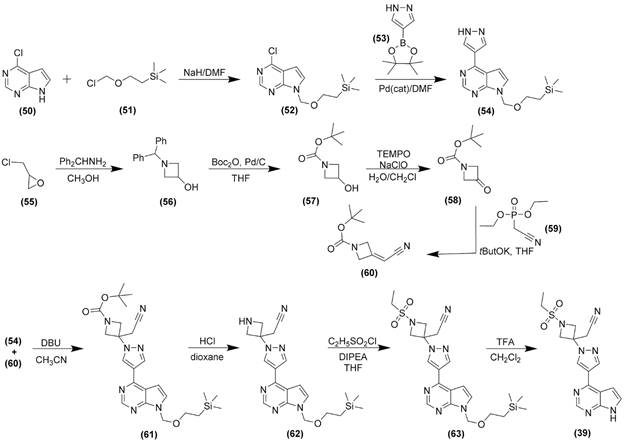
| Repurposed drug |
Study characteristics |
Estimated enrolment |
Dosage |
Characteristics of the study and conclusion |
Clinical trial and recruitment status |
| Remdesivir (8) |
Phase III, double-blind, randomised, placebo-controlled, multicentre |
308 patients |
200 mg Intravenously on the first day plus 100 mg once daily, for more nine consecutive days |
Adult patients with mild and moderate COVID-19, no published results |
NCT04252664 (suspended by epidemic control in China with no eligible patients) |
| 237 patients |
Adult patients with severe COVID-19, remdesivir (8) was not associated with clinical benefits in severe forms. However, an observed numerical reduction in time to clinical improvement in patients treated earlier, but still requires confirmation |
215215. Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, et al. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020; 395(10236): 1569-78.; NCT04257656 (terminated by epidemic control in China with no eligible patients) |
| 1063 patients |
Remdesivir (8) treated patients with lower respiratory tract infection have a shorter time to recovery compared to placebo group (11 vs 15 days) |
216216. Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, et al. Remdesivir for the treatment of Covid-19 - Preliminary report. N Engl J Med [Internet]. 2020 [cited 2020 Aug 8]. Available from: https://doi.org/10.1056/NEJMoa2007764.
https://doi.org/10.1056/NEJMoa2007764...
; NCT04280705 |
| Chloroquine (20) |
Multicentre |
> 100 patients |
500 mg per day, for 10 days |
Compilation of various clinical studies that are in course, authors conclude that the dosage used could be sufficient |
217
217. Gao J, Tian Z, Yang X. Breakthrough: chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci Trends. 2020; 14(1): 72-3.
|
| Phase IIb Double-blind, randomised |
81 patients |
600 mg twice daily for 10 days and 450 mg twice on the first day, once daily for more 4 days |
Higher dosage of chloroquine (20) has toxic effects and increased lethality, with any clinical benefit |
218
218. Borba MGS, Val FFA, Sampaio VS, Alexandre MAA, Melo GC, Brito M, et al. Effect of high vs low doses of chloroquine diphosphate as adjunctive therapy for patients hospitalized with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. JAMA Netw Open. 2020; 3(4): e208857.
|
| Hydroxychloroquine (26) |
non-randomised, non-double-blind, non-placebo-controlled |
36 patients |
600 mg of hydroxychloroquine (26) daily for 10 days |
Hydroxychloroquine (26) significantly reduces viral load despite small sample size |
219
219. Gautret P, Lagier J-C, Parola P, Hoang VT, Meddeb L, Mailhe M, et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents. 2020; 56(1): 105949.
|
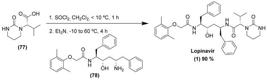
| Ritonavir (2)/lopinavir (1) |
Randomised, controlled, open-label |
199 patients |
400 mg/100 mg twice a day for 14 days |
No benefit was observed with lopinavir (1)-ritonavir (2) treatment compared to standard care in severe Covid-19 patients |
220
220. Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, et al. A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. N Engl J Med. 2020; 382(19): 1787-99.
|
| Ritonavir (2)/lopinavir (1) plus interferon |
Phase II, randomised, controlled, open-label |
127 participants |
400 mg/100 mg twice a day for 14 days; Interferon Beta-1B plus 0.25 mg subcutaneous injection alternate day for three days |
Completed with no published results until this moment |
NCT04276688 |

 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail
 Thumbnail
Thumbnail