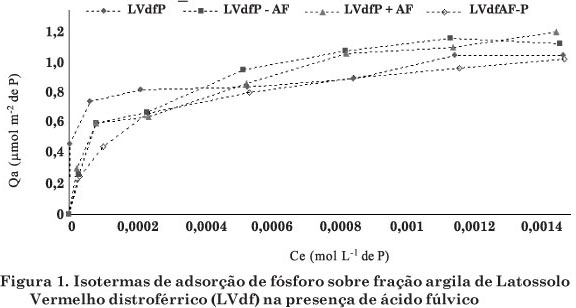
Os ácidos orgânicos podem ser adsorvidos aos coloides do solo, favorecendo maior disponibilidade de P às plantas. Por meio de isotermas, avaliou-se a influência de ácido fúlvico (AF) sobre a adsorção de P na fração argila de dois Latossolos Vermelhos (LVdf e LVd), caulinita natural pouco cristalizada (KGa2), goethita (Gt) e hematita (Hm) sintéticas. As isotermas de adsorção de P ajustadas pelo modelo de Langmuir na presença de AF foram adequadas somente para a região de baixa concentração de P. De maneira geral, a presença de AF incubado nas frações argila estudadas foi capaz de reduzir a adsorção de P. Na fração argila do LVdf, a adsorção de P em baixas e altas concentrações desse nutriente teve pequena influência da sequência de incubação de P com AF, enquanto para a fração argila do LVd as sequências de incubação com P e AF influenciaram a adsorção de P. A redução da adsorção de P sobre a fração argila da KGa2 em baixas concentrações de P nas isotermas não dependeu da sequência de incubação de P e AF, e sim da presença do AF. Na região de altas concentrações de P, a sequência de incubação de P e AF influenciou a adsorção desse elemento. A adsorção de P sobre Gt foi dependente da sequência de incubação com AF, e a incubação prévia do AF promoveu redução da adsorção de P. A sequência de incubação do P e AF na Hm influenciou a adsorção de P quando em baixas e altas concentrações nas isotermas. Os menores valores de adsorção de P ocorreram com a incubação de P juntamente ao AF ou incubação de AF precedida da do P.
isoterma; fosfato; Langmuir