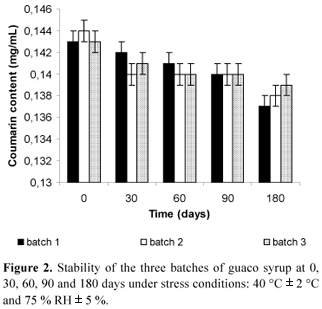
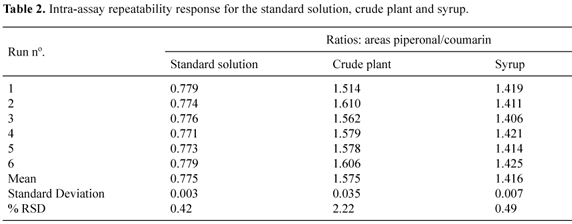
This work describes a full validation of a capillary gas chromatography analytical methodology using internal standardization for the quantification of coumarin (1,2-benzopyrone) in guaco (Mikania glomerata Spreng - Asteraceae) products: syrup, plant and its extract, including the stability study of the phytomedicine. For the analysis, it was used an HP-5 capillary column (30 m x 0.32 mm x 0.25 µm), hydrogen at a flow rate of 1.8 mL/min and the increasing temperature gradient was: 100 ºC to 250 ºC, 15 ºC/min. The temperature of injector (split 1:20) and detector were kept at 250 ºC and 270 ºC, respectively. The retention times of the standards for the above conditions were 2.86 minutes for 1, 2, 3, 4-tetramethylbenzene, 4.45 min for piperonal (internal standards), and 5.36 minutes for coumarin. After extraction procedure, the recovery of coumarin determined for plant raw material was 101.6 %, while for syrup it was 100.8 %. Detection and quantification limits were 0.5 µg/mL and 1.5 µg/mL, respectively. Precision was determined for all samples and the results were lower than 2.5 %. The total amount of coumarin in plant raw material, its extract and syrup were 0.38% w/w, 1.33 mg/mL and 0.143 mg/mL, respectively.
Mikania glomerata; Asteraceae; coumarin; gas chromatography; method validation; syrup