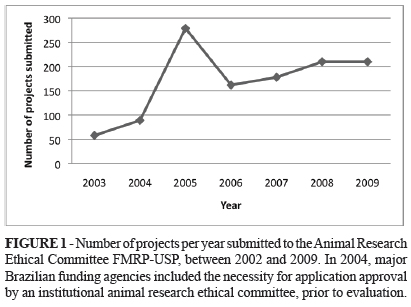
PURPOSE: This study describes in Brazil and in the global biomedical community the time course of the development of animal research welfare guidelines. METHODS: The database of the Ethics Committee of the Faculty of Medicine of Ribeirao Preto (EC/FMRP-USP), Brazil, was surveyed since its inception in 2002 as the regulations became more stringent to provide better protection of animal research welfare at this institution. Medline database was evaluated to identify the number of publications in the period between 1968 and 2008 that used research animals and were in compliance with established ethics guidelines. RESULTS: The EC/FMRP-USP evaluated 979 projects up until 2009. Most of the applications came from Department of Physiology and the most frequently requested species was the rat. In 2004, national research funding agencies started to request prior approval from institutional review ethics committees prior to application review and this requirement became federal law in Brazil in 2008. The analysis of international publications revealed a relative reduction in studies involving research animals (18% in 1968 to 7.5% in 2008). CONCLUSIONS: The present work showed that in the last four decades major changes occurred in the guidelines dictating use of research animals occurred and they are being adopted by developing countries. Moreover, animal welfare concern in the scientific community preceded the introduction of journal guidelines for this purpose. Furthermore, in Brazil it was anticipated that laws were needed to protect animal research welfare from being not upheld.
Ethics; Animal Experimentation; Database