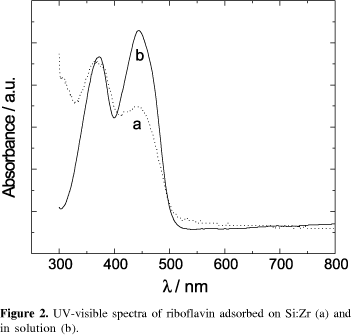
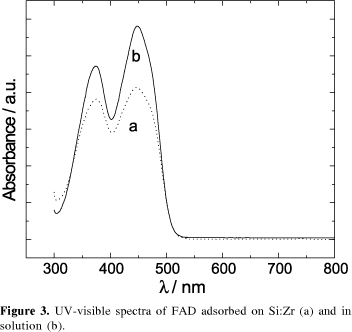
Modified carbon paste electrodes were prepared by the adsorption of flavin on a silica gel modified with zirconium oxide (Si:Zr:flavin). The electrochemical behavior of immobilized riboflavin, FMN and FAD was investigated. The formal potentials, Eº´, for the flavins in these electrodes were found to be -0.46 V vs. SCE, this being similar to their values obtained in solution. Their Eº´ values remained pH dependent with certain invariance for riboflavin between 4 and 5. The composition of the supporting electrolyte did not affect the Eº´ with exception of Ca2+, but in buffer systems and especially in phosphate a good rate of electron transfer was observed. The differences verified in the electrochemical behavior and UV-visible absorption spectra among the immobilized flavins are discussed in terms of different kinds of interaction.
flavin; carbon paste electrode; silica gel; zirconium oxide