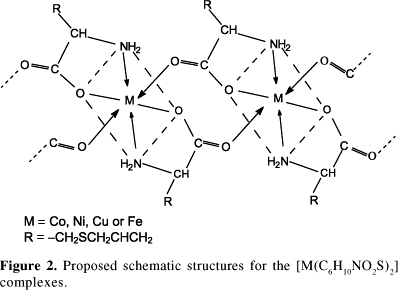
The solid complexes [Co(C6H10NO2S)2], [Ni(C6H10NO2S)2], [Cu(C6H10NO2S)2] and [Fe(C6H10NO2S)2] were obtained from the reaction of cobalt(II), nickel(II), copper(II) and iron(II) salts with the potassium salt of the amino acid deoxyalliin (S-allyl-L-cysteine). Electronic absorption spectra of the complexes are typical of octahedral structures. Infrared spectroscopy confirms the ligand coordination to the metal ions through (COO-) and (NH2) groups. EPR spectrum of the Cu(II) complex indicates a slight distortion of its octahedral symmetry. Mössbauer parameters permitted to identify the presence of iron(II) and iron(III) species in the same sample, both of octahedral geometry. Thermal decomposition of the complexes lead to the formation of CoO, NiO, CuO and Fe2O3 as final products. The compounds show poor solubility in water and in the common organic solvents.
deoxyalliin; cobalt; nickel; copper; iron