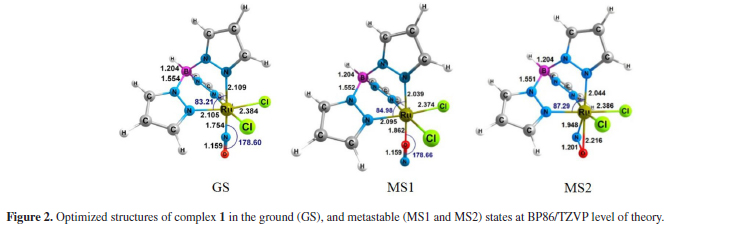
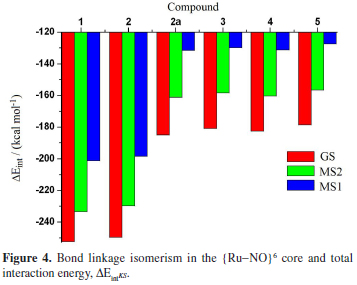
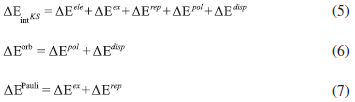The lability of NO+ group in [TpRuCl2(NO)]q (Tp = BL(pyrazol-1-yl)3) complexes was evaluated at the light of energy decomposition analysis (Su-Li EDA). The electronic effects of different pseudoaxial substituents (L = H, pyrazolyl anion, pyrazole, isoxazole and isothiazole) on the nature of Ru-NO bonding were evaluated considering complexes in ground (GS) and in metastable (MS1 and MS2) states. {Ru-NO}6 bond nature in [TpRuCl2(NO)]q (Tp = BL(pyrazol-1-yl)3) complexes is in essence covalent, but with a still significant electrostatic character. The nature of pseudoaxial substituents has a direct effect on the magnitude of {Ru-NO}6 bonds.
scorpionate ligands; ruthenium nitrosyl complexes; Su-Li EDA; NBO