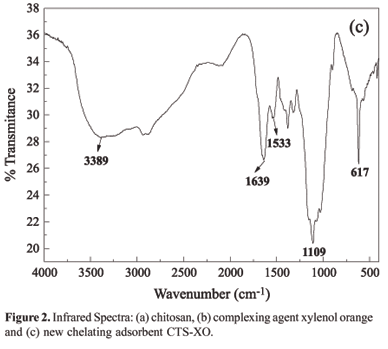
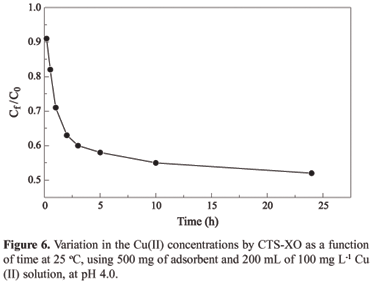
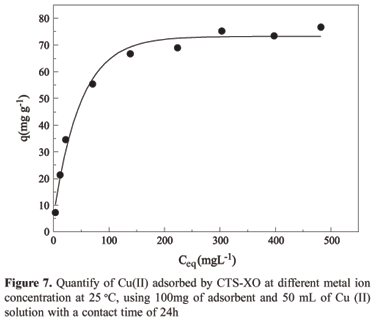
The aim of this study was to impregnate the chelating agent 3,3-bis-N,N,bis-(carboxymethyl)aminomethyl-o-cresolsulfonephthalein in chitosan and to investigate the adsorption of Cu(II) ions. The chemical modification was confirmed by FTIR spectrometry, thermogravimetric analysis (TGA) and energy dispersive x-ray spectroscopy (EDX). The adsorption studies were carried out with Cu(II) ions in a batch process and were shown to be dependent on pH. The adsorption kinetics was tested using three models: pseudo first-order, pseudo second order and intraparticle diffusion. The experimental kinetics data were best fitted with the pseudo second-order model (R² = 0.999), which provided a rate constant, k2, of 1.21 x 10-3 g mg-1 min-1. The adsorption rate depended on the concentration of Cu(II) ions on the adsorbent surface and on the quantity of Cu(II) ions adsorbed at equilibrium. The Langmuir isotherm model provided the best fit for the equilibrium data in the concentration range investigated, with the maximum adsorption capacity being 81.0 mg of Cu(II) per gram of adsorbent, as obtained from the linear equation of the isotherm. Desorption tests revealed that around 90% of the adsorbed metal was removed, using EDTA solution as the eluent. This result suggests that the polymeric matrix can be reused.
Chitosan; impregnation; adsorption; xylenol orange; Cu(II)