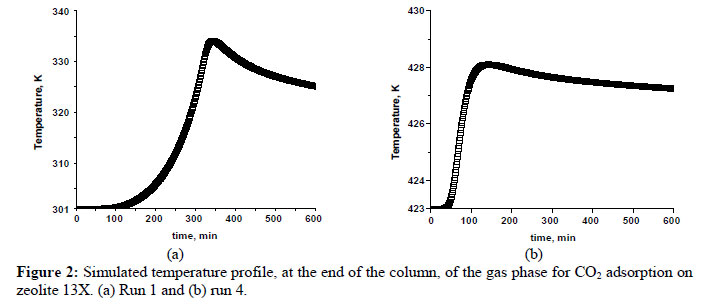
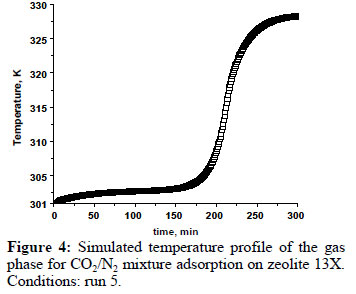
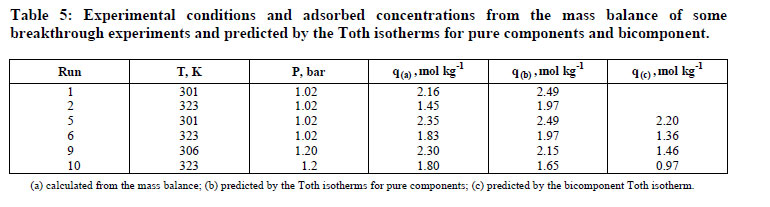
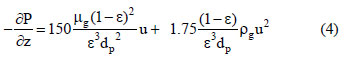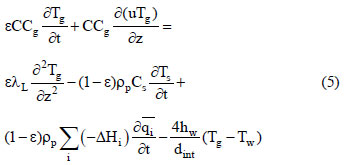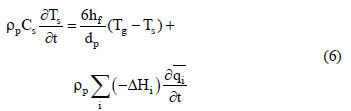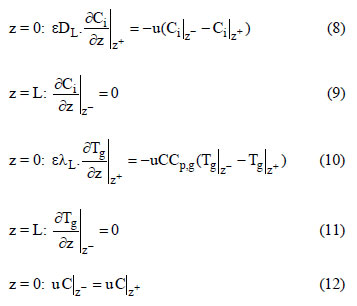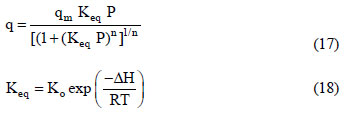In this study, the fixed - bed adsorption of carbon dioxide and a carbon dioxide - nitrogen mixture on zeolite 13X was investigated. The adsorption equilibrium and breakthrough curves were determined at different temperatures - 301 - 306 K, 323 K, 373 K and 423 K. A model based on the LDF approximation for the mass transfer, considering the energy and momentum balances, was used to describe the adsorption kinetics of carbon dioxide and a carbon dioxide - nitrogen mixture. The model acceptably reproduced all of the breakthrough curves and can be considered as adequate for designing a PSA cycle to separate carbon dioxidenitrogen mixtures.
Adsorption; Carbon dioxide; Nitrogen; Zeolite 13X; Modeling