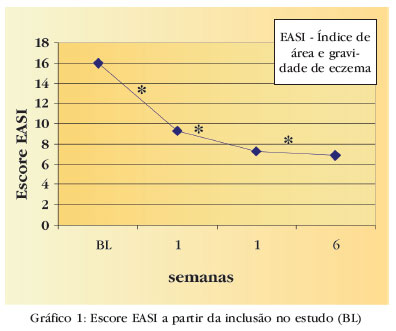
FUNDAMENTOS - Tacrolimo pomada é eficaz no tratamento da dermatite atópica. OBJETIVOS - Avaliar a eficácia e segurança do tacrolimo pomada 0,03% (Protopic®) no tratamento de pacientes pediátricos com dermatite atópica. MÉTODOS - Estudo multicêntrico, aberto e não comparativo. Incluídos 174 pacientes (dois a 10 anos)com dermatite atópica. Utilizou-se tacrolimo duas vezes ao dia, por seis semanas. O critério primário de eficácia foi a melhora clínica > a 90% avaliada pelo médico (Escala de Avaliação Global da Resposta Clínica). Outros critérios de eficácia foram a redução no índice de área e gravidade do eczema (EASI), a redução do percentual de superfície corporal afetada (%BSA) e a avaliação do prurido pelo paciente (escala analógica visual). Segurança foi avaliada pela ocorrência de eventos adversos relatados pelos pais e pacientes ou pelos investigadores. RESULTADOS - Trinta e três por cento dos pacientes apresentaram melhora clínica >90%. Quando avaliado o escore EASI, houve redução de 45,5% (primeira semana) e 61,8% (sexta semana) quando comparado com o basal (p<0,001). Em relação a %BSA, houve redução de 30,4% e 55,5%, na primeira e na sexta semana. Houve redução do prurido em relação ao basal (p<0,001).Os eventos adversos mais comuns foram - queimação e prurido localizados. CONCLUSÃO - Tacrolimo pomada 0,03% é terapia segura e efetiva no tratamento da dermatite atópica leve a grave em pacientes pediátricos.
Criança; Dermatite; Dermatite atópica; Eczema; Tacrolimo