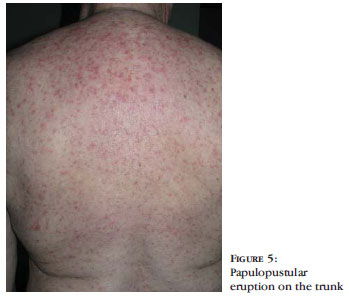
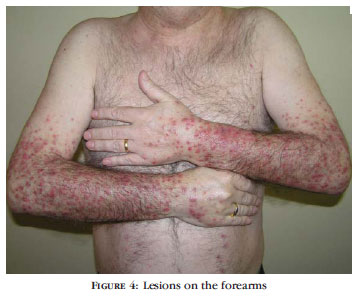
BACKGROUND: Cetuximab and erlotinib, epidermal growth factor receptor inhibitors, often cause peculiar adverse cutaneous reactions. OBJECTIVES: Our aim was to evaluate adverse cutaneous reactions and their management in patients undergoing treatment with cetuximab and erlotinib. PATIENTS AND METHODS: Between March/2005 and September/2009, we observed 14 patients with a mean age of 59.6 years undergoing treatment with cetuximab (7) or erlotinib (7), due to lung(10) or colorectal cancer (4). We evaluated the interval between introduction of the drug and onset of symptoms, treatment response, and the clinical pattern of evolution of the cutaneous reaction retrospectively. RESULTS: Twelve patients presented papular-pustular eruption typically affecting the face, chest and back, which appeared in average 13.5 days after starting the drug treatment. The patients underwent oral treatment with minocycline or doxycycline and topical treatment with metronidazole, benzoyl peroxide and/or corticosteroids. All patients showed improvement of the lesions. Five patients presented periungual pyogenic granulomas, which were associated with paronychia in 4 cases, after an average of 8 weeks of treatment. There was improvement of the lesions with topical treatment (antibiotics, corticosteroids and antiseptics). Xerosis was observed in some patients. Other less frequent adverse side effects such as telangiectasia and angiomas, hair and eyelash alterations, and eruptive melanocytic nevi were also described. Treatment with epidermal growth factor receptor inhibitor was maintained in most patients. CONCLUSION: The increasing use of these targeted therapies requires knowledge of their adverse cutaneous side effects to ensure timely intervention in order to allow the continuation of the therapy
Drug eruptions; Drug toxicity; Epidermal growth factor; Receptor, epidermal growth factor