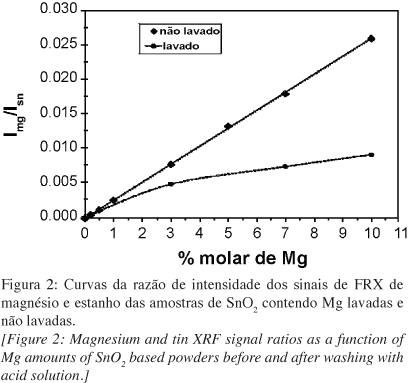
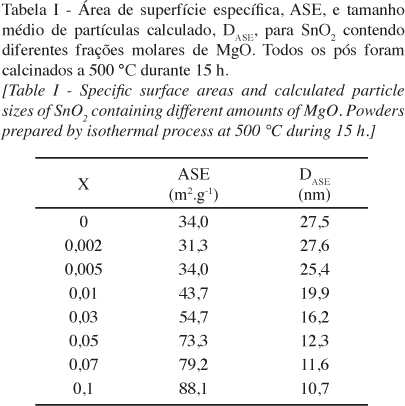
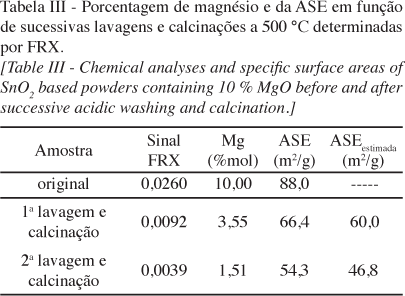
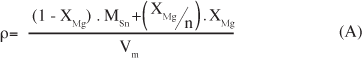Um dos maiores desafios na síntese de nanopartículas é a instabilidade da estrutura relativa ao aumento de energia advindo da grande área de superfície específica desta classe de materiais. O conhecimento dos mecanismos pelos quais a estabilidade pode ser alcançada é de fundamental importância para o controle de seu crescimento e para potenciais aplicações. Fenômenos de segregação superficial em sistemas bi-componentes envolvem diminuição de energia do sistema devido ao excesso de superfície - conhecido como fenômeno de tensoatividade. Apesar de muito bem conhecidos em tecnologias de emulsões e em fenômenos que envolvem molhabilidade, o uso destes conceitos para estabilização da superfície sólido-vapor e controle de nanopartículas é menos expressivo. Embora a segregação seja conhecida para vários sistemas de óxidos nanoparticulados, sua quantificação visando o controle é limitada pela dificuldade de obtenção de resultados confiáveis. Neste trabalho foi realizada a quantificação da segregação de MgO na superfície de nanopartículas de SnO2 através da análise química dos pós antes e após lavagem em meio ácido para lixiviação do MgO superficial. Foi demonstrado que a solubilidade do MgO no bulk do SnO2 é preferencial em concentrações inferiores a 5% em mol de MgO, existindo, no entanto, uma distribuição do MgO ao longo da superfície e do bulk, gerando um efeito tenso-ativo e restringindo o crescimento das partículas de SnO2. Para concentrações maiores que 5% em mol, a maior parte do MgO foi detectada na superfície das nanopartículas de SnO2, sendo o controle tenso-ativo mais expressivo. Apesar dos resultados serem restritos para o sistema SnO2-MgO, esta técnica pode ser extrapolada para o controle de quaisquer sistemas onde o elemento segregado é solúvel em condições específicas.
segregação; SnO2; nanopartículas; tensoatividade