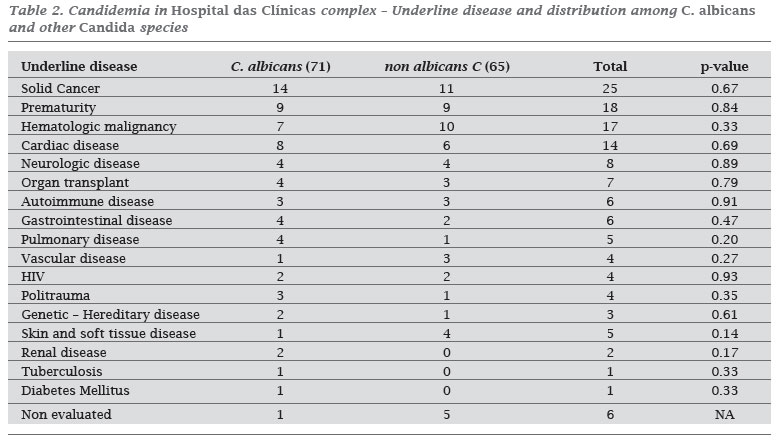
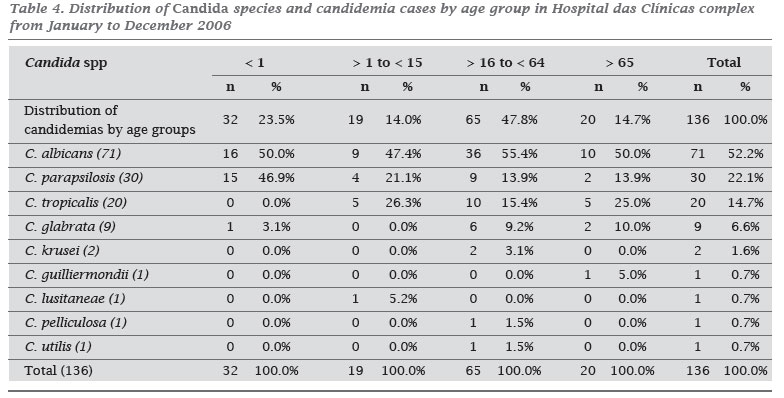
INTRODUCTION: Although the spectrum of fungi causing bloodstream fungal infections continues to expand, Candida spp. remains responsible for the majority of these cases. OBJECTIVE: The purpose of this study was to characterize the candidemia epidemiology, species distribution and antifungal susceptibility patterns at a Brazilian tertiary teaching public hospital with 2,500 beds. METHODS: Records from the microbiology laboratory were used to identify patients with positive blood cultures during 2006. The in vitro activity of amphotericin B, caspofungin, itraconazole, fluconazole, voricanozole, and posaconazole were determined using the Etest method. RESULTS: One hundred and thirty-six cases of candidemia were identified and 100 strains were available for antifungal susceptibility testing. The overall incidence of candidemia was 1.87 cases/1.000 admissions and 0.27 cases/1.000 patient-days. Among the patients, 58.1% were male, and the median age was 40 years old. C. albicans was the most common species (52.2%), followed by C. parapsilosis (22.1%), C. tropicalis (14.8%), and C. glabrata (6.6%). All strains were susceptible to amphotericin B with a MIC90 of 0.5 µg/mL. Overall susceptibility for voriconozole, fluconazole, and caspofungin was > 97% with a MIC90 of 0.064, 4.0 and 1.0 µg/mL, respectively. For itraconazole the susceptibility rate was 81% with a MIC90 of 0.5 µg/mL. Posaconazole also demonstrated good in vitro activity with a MIC90 of 0.25 µg/mL. CONCLUSION: This is the first antifungal susceptibility report in our institution
candidemia; Candida spp; susceptibility testing; Candida epidemiology; Candida Etest; antifungal susceptibility profile; Candida antifungal agents; azoles