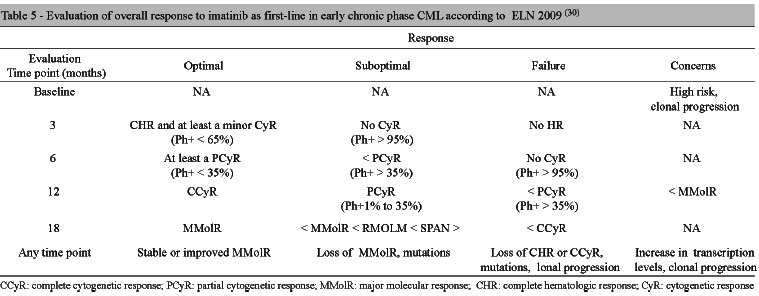
Imatinib mesylate was the first BCR-ABL-target agent approved for the treatment of chronic myeloid leukemia. Although most patients respond well to imatinib therapy, the literature shows that one third develops resistance or intolerance. The timing of second-line treatment after failure of initial treatment may have a significant impact on long-term outcome. Thus, appropriate monitoring to identify resistance and/or intolerance is crucial to early intervention with second generation tyrosine kinase inhibitors and attainment of better results
Leukemia, myelogenous, chronic, BCR-ABL positive; Drug resistance; Drug monitoring, neoplasm; Receptor Protein-Tyrosine Kinases; Pyrimidines; Antineoplastic agents; Monitoring; Prognosis