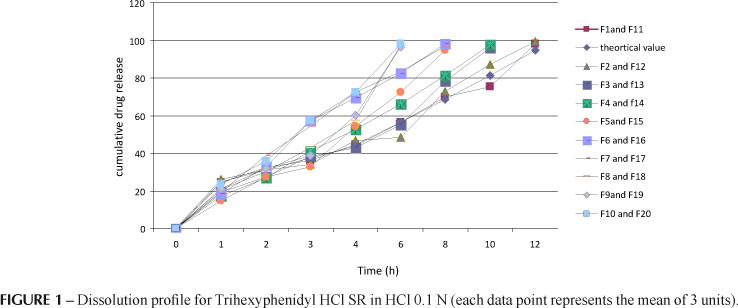
The purpose of this research study was to establish ziprasidone HCl NR 40 mg and trihexyphenidyl HCl SR 4mg in the form of bi-layer sustained release floating tablets. The tablets were prepared using sodium HPMC K4M / HPMC K15M as bio-adhesive polymers and sodium bicarbonate acting as a floating layer. Tablets were evaluated based on different parameters such as thickness, hardness, friability, weight variation, in vitro dissolution studies, content of active ingredient and IR studies. The physico-chemical properties of the finished product complied with the specifications. In vitro release from the formulation was studied as per the USP XXIII dissolution procedure. The formulations gave a normal release effect followed by sustained release for 12 h which indicates bimodal release of ziprasidone HCl from the matrix tablets. The data obtained was fitted to Peppas models. Analysis of n values of the Korsmeyer equation indicated that the drug release involved non-diffusional mechanisms. By the present study, it can be concluded that bi-layer tablets of ziprasidone HCl and trihexyphenidyl HCl will be a useful strategy for extending the metabolism and improving the bioavailability of Ziprasidone HCl and Trihexyphenidyl HCl.
Bi-layer tablet; Ziprasidone HCl; Trihexyphenidyl HCl; Sustained release