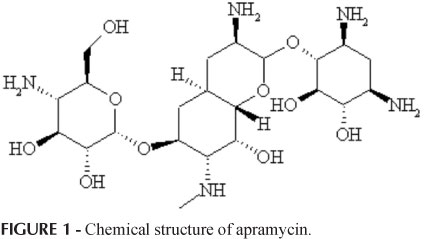
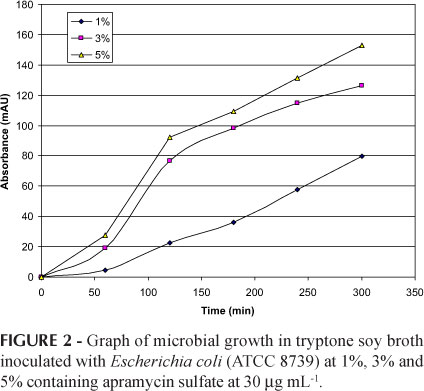
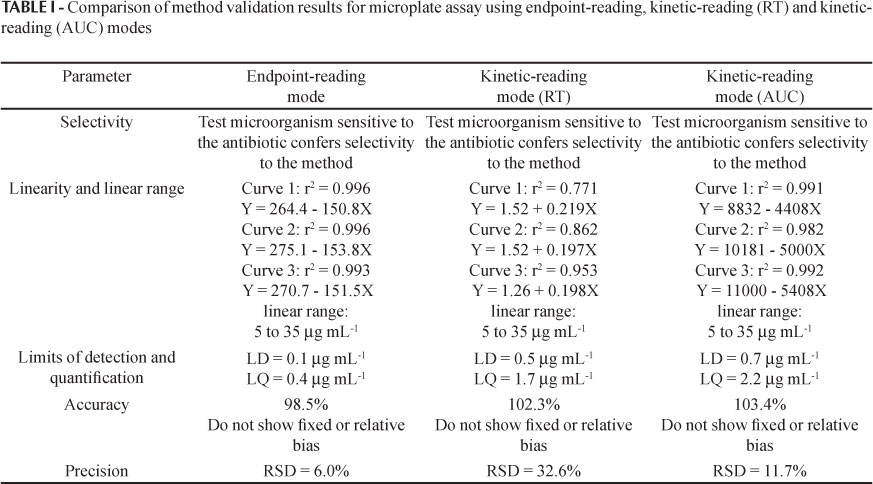
The aim of this study was to determine the optimal experimental conditions to develop a methodology for microbiological assay of apramycin employing microplate and kinetic reading mode, and to validate the developed method, through evaluation of parameters of selectivity, linearity, linear range, limits of detection and quantification, accuracy and precision. The turbidimetric assay principle is simple: the test solution is added to a suspension of test microorganism in culture media, the mixture is incubated under appropriate conditions and the microbial growth is measured by photometric reading. Microplate with kinetic reading mode employed in antibiotic assay is of considerable interest since it allows reduction of material and analysis time and enables a large number of samples to be analyzed simultaneously, with automated reading and calculating. Established conditions considered the standard-curve of apramycin at concentrations from 5.0 to 35.0 μg mL-1, and tryptic soy broth inoculated with 5% Escherichia coli (ATCC 8739) suspension. Satisfactory results were obtained with 2 hours of incubation. The developed method showed appropriate selectivity, linearity in the range from 5.0 to 35.0 μg mL-1, limits of detection and quantification of 0.1 and 0.4 μg mL-1, respectively, as well as satisfactory accuracy (recuperation = 98.5%) and precision (RSD = 6.0%). Microplate assay combined the characteristics of microbiological (evaluation of antibiotic activity against sensitive test microorganism) and physico-chemical (operationally straightforward and faster results) assays.
Antibiotics; Apramycin; Apramycin sulfate; Turbidimetric assay