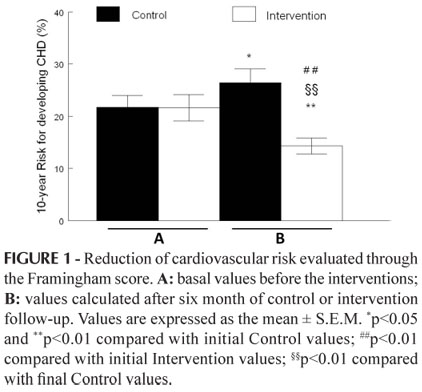
The objective of this study was to determine the impact of a pharmaceutical care (PC) program in a sample of public outpatients with metabolic syndrome (MS) who were being treated in Brazil's health system; the patients were randomized into PC or standard care. The pharmacotherapy follow-up (PF) was performed in a total of 120 patients with type 2 diabetes for 6 months. Adherence to treatment (measured with the Morisky test), negative outcomes associated with medication (NOM) and anthropometric and biochemical parameters were measured before and after PF. The Framingham scoring method was used to estimate changes in 10-year coronary heart disease risk scores in all patients. Ninety-six of 120 patients had characteristics of MS and were randomized into two groups (G): the control group (CG: 36) and the intervention group (IG: 38). Among the MS patients, 100% were taking a glucose-lowering drug; many were also taking anti-hypertensive drugs (CG: 72%; IG: 73%), and some patients were also taking hypolipemic drugs (CG: 12.0%; IG: 14.7%). Only 20.7% of the IG patients were considered adherent to their prescribed drugs. In the CG, an increase of coronary heart disease (CHD) risk (22±2 to 26±3; p<0.05) was observed, while in the IG, there was a reduction in CHD risk (22±2 to 14±2%; p<0.01). The PC program administered to patients with MS monitored through the primary healthcare services of the Brazilian public health system improved patient health, resulting in clinical improvements and a decrease in cardiovascular risk in IG patients over a period of ten years.
Pharmaceutical service; Metabolic syndrome; Drug therapy follow-up; Medicines