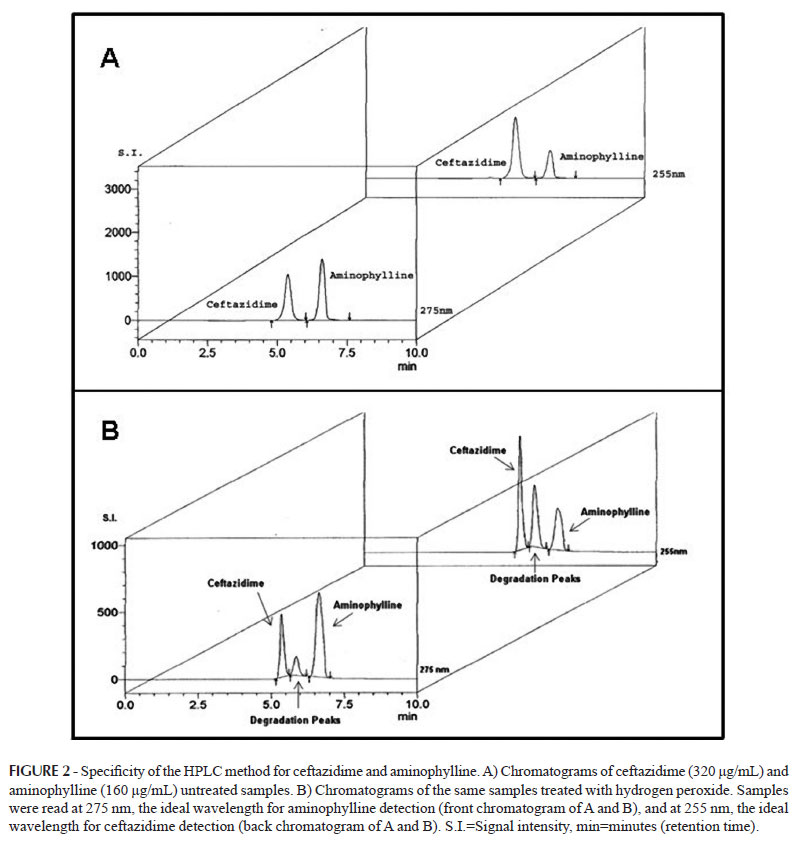
Ceftazidime is a broad spectrum antibiotic administered mainly by the parenteral route, and it is especially effective against Pseudomonas aeruginosa. The period of time in which serum levels exceed the Minimum Inhibitory Concentration (MIC) is an important pharmacodynamic parameter for its efficacy. One of the forms to extend this period is to administer the antibiotic by continuous infusion, after prior dilution in a Parenteral Solution (PS). The present work assessed the stability of ceftazidime in 5% glucose PS for 24 hours, combined or not with aminophylline, through High Performance Liquid Chromatography (HPLC). The physicochemical evaluation was accompanied by in vitro antimicrobial activity compared MIC test in the 24-hour period. Escherichia coli and Pseudomonas aeruginosa were the microorganisms chosen for the MIC comparison. The HPLC analysis confirmed ceftazidime and aminophylline individual stability on PS, while the MIC values were slightly higher than the mean described in the literature. When both drugs were associated in the same PS, the ceftazidime concentration by HPLC decreased 25% after 24 hours. Not only did the MIC values show high loss of antibiotic activity within the same period, but also altered MIC values immediately after the preparation, which was not detected by HPLC. Our results indicate that this drug combination is not compatible, even if used right away, and that PS might not be the best vehicle for ceftazidime, emphasizing the importance of the MIC evaluation for drug interactions.
Drug; Injectables; Parenteral solutions; Ceftazidime; Aminophylline; High Performance Liquid Chromatography; Minimum inhibitory concentration (MIC)