Abstracts
The use of other inducers as substitutes for pectin was studied aiming to reduce the production costs of pectic enzymes. The effects of sugar-cane juice on the production of pectin lyase (PL) and polygalacturonase (PG) by Penicillium griseoroseum were investigated. The fungus was cultured in a mineral medium (pH 6.3) in a rotary shaker (150 rpm) for 48 h at 25oC. Culture media were supplemented with yeast extract and sucrose or sugar-cane juice. Sugar-cane juice added singly to the medium promoted higher PL activity and mycelial dry weight when compared to pectin and the use of sugar-cane juice and yeast extract yielded levels of PG activity that were similar to those obtained with sucrose-yeast extract or pectin. The results indicated that, even at low concentrations, sugar-cane juice was capable of inducing pectin lyase and polygalacturonase with no cellulase activity in P. griseoroseum.
pectin lyase; polygalacturonase; pectinases; Penicillium griseoroseum; sugar-cane juice
Na produção de enzimas pécticas estudou-se a utilização de outros indutores além de pectina visando a redução de custos. Os efeitos de caldo de cana na produção de pectina liase (PL) e poligalacturonase (PG) por Penicillium griseoroseum foram investigados. O fungo foi cultivado em meio mineral (pH 6,3) em um agitador rotatório (150 rpm) por 48 h a 2oC. O meio de cultura foi suplementado com extrato de levedura e sacarose ou caldo de cana. A adição de caldo de cana ao meio de cultivo promoveu maior atividade de PL e peso do micélio seco quando comparado com a adição de pectina. Na presença de caldo de cana e extrato de levedura observou-se atividade de PG semelhante ao obtido na presença de sacarose e extrato de levedura ou pectina. Os resultados indicaram que, mesmo em baixas concentrações, caldo de cana foi capaz de induzir pectina liase e poligalacturonase, sem atividade de celulase, em Penicillium griseoroseum.
pectina liase; poligalacturonase; pectinases; Penicillium griseoroseum; caldo de cana
SUGAR-CANE JUICE INDUCES PECTIN LYASE AND POLYGALACTURONASE IN PENICILLIUM GRISEOROSEUM
Rosana Cristina Minussi, Juliana Rocha Lopes Soares-Ramos, Jorge Luiz Cavalcante Coelho, Daison Olzany Silva* * Corresponding author. Mailing address: Departamento de Microbiologia, Núcleo de Biotecnologia Aplicada à Agropecuária BIOAGRO, Universidade Federal de Viçosa, CEP: 36571-000, Viçosa, MG, Brazil. Fax: (+5531) 899-2864
Departamento de Microbiologia, Núcleo de Biotecnologia Aplicada à Agropecuária, BIOAGRO, Universidade Federal de Viçosa, Viçosa, MG, Brazil
Submitted: February 07, 1997; Returned to authors for corrections: March 24, 1998;
Approved: August 20, 1998.
ABSTRACT
The use of other inducers as substitutes for pectin was studied aiming to reduce the production costs of pectic enzymes. The effects of sugar-cane juice on the production of pectin lyase (PL) and polygalacturonase (PG) by Penicillium griseoroseum were investigated. The fungus was cultured in a mineral medium (pH 6.3) in a rotary shaker (150 rpm) for 48 h at 25oC. Culture media were supplemented with yeast extract and sucrose or sugar-cane juice. Sugar-cane juice added singly to the medium promoted higher PL activity and mycelial dry weight when compared to pectin and the use of sugar-cane juice and yeast extract yielded levels of PG activity that were similar to those obtained with sucrose-yeast extract or pectin. The results indicated that, even at low concentrations, sugar-cane juice was capable of inducing pectin lyase and polygalacturonase with no cellulase activity in P. griseoroseum.
Key words: pectin lyase, polygalacturonase, pectinases, Penicillium griseoroseum, sugar-cane juice.
INTRODUCTION Pectic enzymes have been tested for retting processes based on the ability of certain pectolytic microorganisms to liberate cellulose fibers from the stems of fibrous plants (15).
Among pectic enzymes, pectin lyase (EC 4.2.2.3) is the most interesting, as it is the only one capable of depolimerize pectins without altering their esterification level (16) and to cleave a-1,4 bonds of highly esterified pectin without the previous action of other enzymes (1). Most commercial enzymatic complexes used today contain cellulases that attack cellulose fibers making them less resistant to industrial processing.
Other inducers than pectin have been studied aiming to reduce the production costs of pectinases. Penicillium griseoroseum synthesizes low amounts of cellulases (6) and produces pectin lyase in media containing sucrose and yeast extract (YE) (3), methylxanthines (13), or tea extract (4,12). Sugar-cane has a low pectin concentration (around 0.2%) but is rich in sucrose (approximately 16%). The aim of this work was to investigate the effects of sugar-cane juice on the production of pectin lyase (PL) and polygalacturonase (PG) by P. griseoroseum, since it is inexpensive and readily available in Brazil.
MATERIALS AND METHODS
Microorganism and cultivation conditions
Penicillium griseoroseum was originally obtained from seeds of forest trees at the Departamento de Fitopatologia, Universidade Federal de Viçosa, Viçosa, Minas Gerais, Brazil. Inoculum was produced by culturing Penicillium griseoroseum on oatmeal-agar for 9 days at 25
oC. Mineral medium (50 ml, pH 6.3, consisted of (g/L): KH
2PO
4, 8.0; K
2HPO
4, 2.48; MgSO
44.7H
2O, 1.1 and (NH
4)
2SO
4, 1.0) was supplemented with 0.03% yeast extract (YE) and sucrose (0.1 to 2.0%) or sugar-cane juice to a final concentration of sucrose ranging from 0.1 to 2.0% (the sucrose content in sugar-cane juice being assumed to be around of 16%). Sugar-cane juice, 0.4% tea extract (TE) and 0.4% citric pectin were also tested in the absence of YE. The cultures were inoculated with 5x104 spores per ml and incubated on a rotary shaker (150 rpm) for 48 h at 25
oC (5). All experiments were carried out in triplicate.
Enzymatic analysis and mycelial growth
Cultures were harvested by filtering through a 400 mesh sieve (37 µm pore size) and growth was determined as mycelial dry weight (MDW) according to Calam (7). PL and PG activities were determined spectrophotometrically (A
235and A
540), in the culture filtrate, according to Albersheim (2) and by the 3,5-dinitrosalicylic acid method (11) using galacturonic acid as standard, respectively. PL activity unit (U/ml) was defined as nmoles of unsaturated uronides produced per min per ml of culture filtrate (13). PG activity unit (U/ml) was defined as nmoles of galacturonic acid produced per min per ml of culture filtrate (14). Cellulase activity was estimated as described and modified by Halliwell (9), filter paper (1x7 cm) was incubated with 2.0 ml of culture filtrate at 40
oC for 1 hour and the glucose formed was measured by 3,5-dinitrosalicylic acid method (11) using glucose as a standard. RESULTS AND DISCUSSION The effect of different substrates on the production of PL and PG by
Penicillium griseoroseum can be seen in
Fig. 1. The fungus did not produce a significant amount of PL in the presence of sucrose as the sole carbon source. However, when the medium was supplemented with sucrose and YE or other inducers like pectin, sugar-cane juice (with or without YE) , or tea extract, a significant PL activity was detected (
Fig. 1A). Maximum PL activity was achieved when the culture was grown in the mineral medium containing sugar-cane juice and YE (72.16 U/ml). In this case, PL activity corresponded to 2.64 and 1.96 times that obtained with pectin and sucrose-YE, respectively. Sugar-cane juice added singly to the medium promoted higher PL activity and MDW when compared to pectin (
Figs. 1A and 1C). However, this did not occur for PG (
Fig. 1B). Maximum PG activity was obtained with pectin and YE. The use of sugar-cane juice and YE yielded levels of PG activity that were similar to those obtained with sucrose-YE or pectin, while TE could induce only low levels of enzyme activity (
Fig. 1B).
Figure 1. Pectin lyase (PL) activity (A), polygalacturonase (PG) activity (B), and mycelial dry weight (MDW) (C) of
P. griseoroseum cultured on 0.4% sucrose (S), 0.4% sucrose and 0.03% YE (S+YE), 0.4% pectin (P), 0.4% pectin and 0.03% YE (P+YE), 0.4% tea extract (TE), 0.4% sugar-cane juice (SC), 0.4% sugar-cane juice and 0.03% YE (SC+YE).
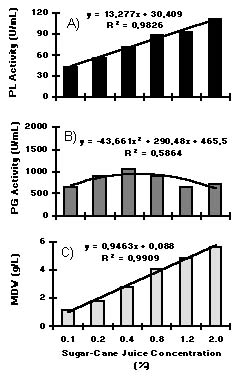
Sucrose at concentrations ranging from 0.1 to 2.0% with 0.03% YE promoted an increase in MDW, though PL and PG activity remained approximately constant (the statistical analysis demonstrated that there was no significant difference (P>0.05)) (Fig. 2). The same was not true for sugar-cane juice (Fig. 3). Increasing concentrations of sugar-cane juice (0.1-2.0%), in the presence of 0.03% YE, promoted higher PL activity and MDW. The maximum value for PL activity corresponded to 2.9 times that obtained with sucrose and YE added to the culture medium at equivalent sucrose concentrations (Figs. 2A and 3A). PG activity increased as the concentrations of sugar-cane juice were in the range of 0.1 to 0.4% (Fig. 3B).
Figure 2. Pectin lyase (PL) activity (A), polygalacturonase (PG) activity (B), and mycelial dry weight (MDW) (C) of
P. griseoroseum cultured on 0.03% YE and increasing concentrations of sucrose (0.1-2.0%).
Pectin lyase (PL) activity (A), polygalacturonase (PG) activity (B), and mycelial dry weight (MDW) (C) of P. griseoroseum cultured on 0.03% YE and increasing concentrations of sugar-cane juice (0.1-2.0% as sucrose concentrations).
Sugar-cane juice induced high PL production even in the medium without YE, though with lower activity (Fig. 4A). The higher the concentrations of sugar-cane juice in the medium (up to 0.4%), the higher the amounts of PG synthesized by the fungus (Fig. 4B). MDW increases were directly proportional to the concentration of sugar-cane juice (Fig. 4C).
Pectin lyase (PL) activity (A), polygalacturonase (PG) activity (B), and mycelial dry weight (MDW) (C) of P. griseoroseum cultured on sugar-cane juice (0.1-2.0% as sucrose concentrations).
The results obtained in this study indicate that sugar-cane juice induced PL and PG synthesis in P. griseoroseum either in the presence or absence of YE. Differently from sucrose, increasing concentrations of sugar-cane juice promoted increases in PL and PG activities with or without the addition of YE to the medium. Sugar-cane juice and YE seem to have different effects on the induction of PL and PG. A synergistic effect of these compounds on PL and PG activities could be observed when they were added simultaneously to the culture medium. The results indicate the existence of different induction mechanisms for PL and PG in P. griseoroseum cultured on sugar-cane juice. Increasing concentrations of sugar-cane juice promoted linear increases in PL activity (linear function), contrarily to that observed for PG activity (polynomial function) (Figs. 3 and 4). The PG synthesis could be partially repressed when sugar-cane juice at sucrose concentrations above 0.4% was added to the culture medium (catabolite repression) (14).
According to Leme and Borges (10), sugar-cane contains 11-18% sucrose, 0.15-0.25% pectic substances, gums and mucilages, 0.3-0.6% nitrogenous compounds (aminoacids, xanthinic compounds), 5.5% cellulose, among other constituents. P. griseoroseum is capable of producing PL when grown in media containing sucrose and YE (3), methylxanthines (13), or tea extract (4,12). When 0.1-2.0% sugar-cane juice (as sucrose concentrations) were added to 50 ml of mineral medium, the amount of pectic substances present were very low (0.00125-0.025%, considering 0.2% pectic substances in sugar-cane juice). Baracat et al. (4) showed that the addition of 0.0001-0.005% of citric pectin to the culture medium promoted no significant induction of PL by P. griseoroseum. Minussi et al. (12) reported that best PL activity was obtained when P. griseoroseum was cultured in 0.35% pectin and 0.05% sucrose in comparison to the treatment with 0.4% pectin added singly to the medium. Media containing sugar-cane juice are much richer and therefore are more suitable for higher mycelial mass production. Thus, the authors suggest a synergistic effect brought about by the simultaneous addition of pectin substances, sucrose, and xanthinic compounds present in sugar-cane juice, allied with the high mycelial mass production, for the high PL production by P. griseoroseum cultured in sugar-cane juice.
To verify the existence of cellulases, P. griseoroseum was grown at 25
oC for 144 h on 0.4% sugar-cane juice and 0.03% YE. MDW and the activities of PL, PG, and cellulase were determined. No cellulase activity was detected in the culture filtrate during 144 h, which are in according with Brumano
et al. (6). PL activity had a slight increase after 120 h of cultivation. MDW were approximately constant after 48h of growth, while PG activity had a decrease (Figure 5). Geöcze
et al. (8) also observed that there was a fall in PG production when
Penicillium expansum was cultured in citrus pectin supplemented with YE, reaching maximal enzyme activity after 36 to 48 h of culture. These authors suggered that the amino acids present in the yeast extract may be repressing PG synthesis.
Our results indicated that, even at low concentrations, sugar-cane juice added singly to the medium was capable of inducing PL and PG in P. griseoroseum. Sugar-cane juice, when compared to other pectinase inducers, seems to be an economical alternative for the industrial production of PL and PG.
ACKNOWLEDGEMENTS
The authors wish to thank the Brazilian Agencies, FINEP and CNPq, for the financial support.
RESUMO
Caldo de cana induz pectina liase e poligalacturonase em Penicillium griseoroseum
Na produção de enzimas pécticas estudou-se a utilização de outros indutores além de pectina visando a redução de custos. Os efeitos de caldo de cana na produção de pectina liase (PL) e poligalacturonase (PG) por Penicillium griseoroseum foram investigados. O fungo foi cultivado em meio mineral (pH 6,3) em um agitador rotatório (150 rpm) por 48 h a 25
oC. O meio de cultura foi suplementado com extrato de levedura e sacarose ou caldo de cana. A adição de caldo de cana ao meio de cultivo promoveu maior atividade de PL e peso do micélio seco quando comparado com a adição de pectina. Na presença de caldo de cana e extrato de levedura observou-se atividade de PG semelhante ao obtido na presença de sacarose e extrato de levedura ou pectina. Os resultados indicaram que, mesmo em baixas concentrações, caldo de cana foi capaz de induzir pectina liase e poligalacturonase, sem atividade de celulase, em
Penicillium griseoroseum.
Palavras-chave: pectina liase, poligalacturonase, pectinases, Penicillium griseoroseum, caldo de cana
REFERENCES
1. Alaña, A.; Llama, M.J.; Serra, J.L. Purification and some properties of the pectin lyase from
Penicillium italicum.
FEBS Lett., 280: 335-340, 1991.
2. Albersheim, P. Pectin lyase from fungi. In: Neufeuld, E.F.; Ginsburg, V. (eds). Methods in enzymology. Academic Press, New York, 1966, p. 628-635.
3. Baracat-Pereira, M.C.; Coelho, J.L.C.; Silva, D.O. Production of pectin lyase by Penicillium griseoroseum cultured on sucrose and yeast extract for degumming of natural fibers. Lett. Appl. Microbiol., 18: 127-129, 1994.
4. Baracat-Pereira, M.C.; Minussi, R.C.; Coelho, J.L.C.; Silva, D.O. Tea extract as an inexpensive inducer of pectin lyase in Penicillium griseoroseum cultured on sucrose. J. Ind. Microbiol. & Biotechnol. 18: 308-311, 1997.
5. Brumano, M.H.N.; Coelho, J.L.C.; Araújo, E.F.; Silva, D.O. Production of pectin lyase by Penicillium griseoroseum as a function of inoculum and culture conditions. World J. Microbiol. Biotechnol., 9: 225-228, 1993.
6. Brumano, M.H.N.; Coelho, J.L.C.; Araújo, E.F.; Silva, D.O. Pectin lyase of Penicillium griseoroseum related to degumming of ramie. Rev. Microbiol. 24: 175-178, 1993.
7. Calam, C.T. The evaluation of mycelial growth. In: Norris, J.R.; Ribbons D.W. (ed). Methods in microbiology. Academic Press, London, 1969, p. 567-591.
8. Geöcze, M.L.A.; Coelho, J.L.C.; Araújo, E.F.; Silva, D.O. Effect of yeast extract and medium pH on polygalacturonase production by Penicillium expansum. Rev. Microbiol. 26: 165-168, 1995.
9. Halliwell, G. Cellulase. In: Bergmeyer, H.U. (ed). Methods of enzymatic analysis. England, 1974, p. 1134-1142.
10. Leme, Jr, J.; Borges, J.M. Açúcar de cana. Imprensa Universitária/UFV, Viçosa, 1965.
11. Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analyt. Chem., 31: 426-431,1959.
12. Minussi, R.C.; Coelho, J.L.C.; Baracat-Pereira, M.C.; Silva, D.O. Pectin lyase production by Penicillium griseoroseum: effect of tea extract, caffeine, yeast extract, and pectin. Biotechnol. Lett. 18: 1283-1286, 1996.
13. Minussi, R.C.; Baracat-Pereira, M.C.; Coelho, J.L.C.; Silva, D.O. Methylxanthines as inducers of pectin lyase in Penicillium griseoroseum cultured on sucrose. Lett. Appl. Microbiol. 24: 369-372, 1997.
14. Piccoli-Valle, R.H.; Baracat-Pereira, M.C.; Silva, D.O. Catabolite repression of inductive polygalacturonase synthesis in Penicillium expansum by sucrose. J. Basic Microbiol. 35: 189-193, 1995.
15. Rombouts, F.M.; Pilnik, W. Pectic enzymes. In: Rose A. H. (ed). Economic microbiology. Microbial enzymes and bioconversions. Academic Press, London, 1980, p. 227-282.
16. Spagna, G.; Pifferi, P.G.; Gilioli, E. Immobilization of a pectinlyase from
Aspergillus niger for application in food technology.
Enzyme Microb. Technol. 17: 729-738, 1995.
-
1Alaña, A.; Llama, M.J.; Serra, J.L. Purification and some properties of the pectin lyase from Penicillium italicum FEBS Lett., 280: 335-340, 1991.
-
2Albersheim, P. Pectin lyase from fungi. In: Neufeuld, E.F.; Ginsburg, V. (eds). Methods in enzymology Academic Press, New York, 1966, p. 628-635.
-
3Baracat-Pereira, M.C.; Coelho, J.L.C.; Silva, D.O. Production of pectin lyase by Penicillium griseoroseum cultured on sucrose and yeast extract for degumming of natural fibers. Lett. Appl. Microbiol., 18: 127-129, 1994.
-
5Brumano, M.H.N.; Coelho, J.L.C.; Araújo, E.F.; Silva, D.O. Production of pectin lyase by Penicillium griseoroseum as a function of inoculum and culture conditions. World J. Microbiol. Biotechnol., 9: 225-228, 1993.
-
6Brumano, M.H.N.; Coelho, J.L.C.; Araújo, E.F.; Silva, D.O. Pectin lyase of Penicillium griseoroseum related to degumming of ramie. Rev. Microbiol. 24: 175-178, 1993.
-
7Calam, C.T. The evaluation of mycelial growth. In: Norris, J.R.; Ribbons D.W. (ed). Methods in microbiology Academic Press, London, 1969, p. 567-591.
-
8Geöcze, M.L.A.; Coelho, J.L.C.; Araújo, E.F.; Silva, D.O. Effect of yeast extract and medium pH on polygalacturonase production by Penicillium expansum Rev. Microbiol. 26: 165-168, 1995.
-
9Halliwell, G. Cellulase. In: Bergmeyer, H.U. (ed). Methods of enzymatic analysis England, 1974, p. 1134-1142.
-
10Leme, Jr, J.; Borges, J.M. Açúcar de cana Imprensa Universitária/UFV, Viçosa, 1965.
-
11Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analyt. Chem., 31: 426-431,1959.
-
12Minussi, R.C.; Coelho, J.L.C.; Baracat-Pereira, M.C.; Silva, D.O. Pectin lyase production by Penicillium griseoroseum: effect of tea extract, caffeine, yeast extract, and pectin. Biotechnol. Lett. 18: 1283-1286, 1996.
-
13Minussi, R.C.; Baracat-Pereira, M.C.; Coelho, J.L.C.; Silva, D.O. Methylxanthines as inducers of pectin lyase in Penicillium griseoroseum cultured on sucrose. Lett. Appl. Microbiol. 24: 369-372, 1997.
-
14Piccoli-Valle, R.H.; Baracat-Pereira, M.C.; Silva, D.O. Catabolite repression of inductive polygalacturonase synthesis in Penicillium expansum by sucrose. J. Basic Microbiol. 35: 189-193, 1995.
-
15Rombouts, F.M.; Pilnik, W. Pectic enzymes. In: Rose A. H. (ed). Economic microbiology. Microbial enzymes and bioconversions Academic Press, London, 1980, p. 227-282.
-
16Spagna, G.; Pifferi, P.G.; Gilioli, E. Immobilization of a pectinlyase from Aspergillus niger for application in food technology. Enzyme Microb. Technol. 17: 729-738, 1995.
Publication Dates
-
Publication in this collection
27 May 1999 -
Date of issue
Oct 1998
History
-
Accepted
20 Aug 1998 -
Received
07 Feb 1997 -
Reviewed
24 Mar 1998