Abstracts
Lymphocytes activated by mitogenic lectins display changes in transmembrane potential, an elevation in the cytoplasmic Ca2+ concentrations, proliferation and/or activation induced cell death. Low concentrations of ouabain (an inhibitor of Na+,K+-ATPase) suppress mitogen-induced proliferation and increases cell death. To understand the mechanisms involved, a number of parameters were analyzed using fluorescent probes and flow cytometry. The addition of 100nM ouabain to cultures of peripheral blood lymphocytes activated with 5µg/ml phytohemagglutinin (PHA) did not modify the increased expression of the Fas receptor or its ligand FasL induced by the mitogen. However, treatment with ouabain potentiated apoptosis induced by an anti-Fas agonist antibody. A synergy between ouabain and PHA was also observed with regard to plasma membrane depolarization. PHA per se did not induce dissipation of mitochondrial membrane potential but when cells were also exposed to ouabain a marked depolarization could be observed, and this was a late event. It is possible that the inhibitory effect of ouabain on activated peripheral blood lymphocytes involves the potentiation of some of the steps of the apoptotic process and reflects an exacerbation of the mechanism of activation-induced cell death.
lymphocytes; ouabain; PHA; apoptosis; membrane depolarization
Quando linfócitos são ativados por lectinas mitogênicas apresentam mudanças do potencial de membrana, elevação das concentrações citoplasmáticas de cálcio, proliferação e/ou morte celular induzida por ativação (AICD). Concentrações baixas de ouabaína (um inibidor da Na,K-ATPase) suprimem a proliferação induzida por mitógenos e aumentam a morte celular. Para entender os mecanismos envolvidos, uma série de parâmetros foram avaliados usando sondas fluorescentes e citometria de fluxo. A adição de 100nM de ouabaína para culturas de linfócitos de sangue periférico ativadas por fitohemaglutinina (PHA) não modificou o aumento de expressão do receptor Fas ou de seu ligante FasL induzida pelo mitógeno. No entanto, o tratamento com ouabaína potenciou a apoptose induzida por um anticorpo anti-Fas funcionando como agonista. Um sinergismo entre ouabaína e PHA também foi observado com relação à despolarização da membrana plasmática. Com relação à membrana mitocondrial, PHA por si só não produziu despolarização, mas quando as células foram também expostas à ouabaina uma dissipação do potencial foi observado, mas isso foi um evento tardio. É possível que o efeito inibitório da ouabaína em linfócitos de sangue periférico ativados envolva a potencialização de alguns aspectos do processo apoptótico e reflita uma exacerbação do mecanismo de AICD.
linfócitos; ouabaína; PHA; apoptose; despolarização de membrana
BIOMEDICAL AND MEDICAL SCIENCES
Ouabain exacerbates activation-induced cell death in human peripheral blood lymphocytes
Mabel B. Esteves; Luis F. Marques-Santos; Ottília R. Affonso-Mitidieri; Vivian M. Rumjanek* * Member Academia Brasileira de Ciências
Laboratório de Imunologia Tumoral, Instituto de Bioquímica Médica, Universidade Federal do Rio de Janeiro, 21941-590 Rio de Janeiro, RJ, Brasil
Correspondence Correspondence to Vivian M. Rumjanek E-mail: vivian@bioqmed.ufrj.br
ABSTRACT
Lymphocytes activated by mitogenic lectins display changes in transmembrane potential, an elevation in the cytoplasmic Ca2+ concentrations, proliferation and/or activation induced cell death. Low concentrations of ouabain (an inhibitor of Na+,K+-ATPase) suppress mitogen-induced proliferation and increases cell death. To understand the mechanisms involved, a number of parameters were analyzed using fluorescent probes and flow cytometry. The addition of 100nM ouabain to cultures of peripheral blood lymphocytes activated with 5µg/ml phytohemagglutinin (PHA) did not modify the increased expression of the Fas receptor or its ligand FasL induced by the mitogen. However, treatment with ouabain potentiated apoptosis induced by an anti-Fas agonist antibody. A synergy between ouabain and PHA was also observed with regard to plasma membrane depolarization. PHA per se did not induce dissipation of mitochondrial membrane potential but when cells were also exposed to ouabain a marked depolarization could be observed, and this was a late event. It is possible that the inhibitory effect of ouabain on activated peripheral blood lymphocytes involves the potentiation of some of the steps of the apoptotic process and reflects an exacerbation of the mechanism of activation-induced cell death.
Key words: lymphocytes, ouabain, PHA, apoptosis, membrane depolarization.
RESUMO
Quando linfócitos são ativados por lectinas mitogênicas apresentam mudanças do potencial de membrana, elevação das concentrações citoplasmáticas de cálcio, proliferação e/ou morte celular induzida por ativação (AICD). Concentrações baixas de ouabaína (um inibidor da Na,K-ATPase) suprimem a proliferação induzida por mitógenos e aumentam a morte celular. Para entender os mecanismos envolvidos, uma série de parâmetros foram avaliados usando sondas fluorescentes e citometria de fluxo. A adição de 100nM de ouabaína para culturas de linfócitos de sangue periférico ativadas por fitohemaglutinina (PHA) não modificou o aumento de expressão do receptor Fas ou de seu ligante FasL induzida pelo mitógeno. No entanto, o tratamento com ouabaína potenciou a apoptose induzida por um anticorpo anti-Fas funcionando como agonista. Um sinergismo entre ouabaína e PHA também foi observado com relação à despolarização da membrana plasmática. Com relação à membrana mitocondrial, PHA por si só não produziu despolarização, mas quando as células foram também expostas à ouabaina uma dissipação do potencial foi observado, mas isso foi um evento tardio. É possível que o efeito inibitório da ouabaína em linfócitos de sangue periférico ativados envolva a potencialização de alguns aspectos do processo apoptótico e reflita uma exacerbação do mecanismo de AICD.
Palavras-chave: linfócitos, ouabaína, PHA, apoptose, despolarização de membrana.
INTRODUCTION
Ouabain is a cardiotonic steroid that has been widely used, as well as other cardiac glycosides, in the clinic. The known mode of action of ouabain is through the inhibition of the Na+,K+ pump. The Na+,K+-ATPase is associated to the plasma membrane and is responsible for the maintenance, in the interior of the cells, of elevated levels of K+ and low concentration of Na+ typical of the majority of mammalian cells. The pump uses the energy of hydrolysis of one ATP molecule to transport to the exterior of the cell three ions of Na+ in exchange of the entry of two K+ ions (Skou 1990).
Quastel and Kaplan (1968) were the first to describe the inhibition by ouabain of lymphocyte proliferation induced by PHA. Since then, a number of workers, including ourselves, has described the inhibitory effects of ouabain on lymphocyte proliferation induced by various stimuli such as mitogens (Szamel et al. 1981, Moraes et al. 1989, Olej et al. 1998), phorbol ester (Olej et al. 1994, Brodie et al.1995), CD3 (Szamel et al. 1995), Interleukin-2 (Redondo et al. 1986) and calcium ionophore (Jensen et al. 1977).
Very early signals such as the expression of c-myc (Olej et al. 1998) and CD69 (Pires et al. 1997) are increased in activated lymphocytes exposed to ouabain suggesting that these lymphocytes are originally responsive to stimulation. One explanation for this lack of proliferation in parallel with an apparent response to the stimulus could result from the fact that activated lymphocytes were following the apoptotic pathway as opposed to the proliferative pathway. Anner et al. (1994) showed that peripheral blood lymphocytes underwent cell death when exposed to the endogenous isoform of ouabain produced in the hypothalamus. Similarly, our group also described apoptosis in activated PBL exposed to commercial ouabain (Olej et al. 1998). Similarly, it has been reported that Jurkat T cells (a human lymphocyte cell line) underwent apoptosis after exposure to the drug (Orlov et al. 1999).
Lymphocytes are subjected to cell death checkpoints at many points during their lifespan. The process of activation-induced cell death (AICD) is a physiologic mechanism occurring at the end of immune responses essential to maintain the homeostasis of the immune system. Overexpression of the molecules Fas receptor and FasL is seen in stimulated lymphocytes and activation of Fas receptor by its ligand triggers the death program (Nagata and Goldstein 1995, Martin and Green 1995). Lack of this mechanism leads to autoimmunity and to the failure of limiting the expansion of an immune response by eliminating effector cells that are not longer needed (Lynch et al. 1995). The process of AICD involves reduction in cell size; activation of the caspase machinery via caspase 8 which in turn activates caspase 3 to initiate the degradation of a variety of substrates; increased intracellular calcium levels and activation of nucleases, leading to DNA degradation (Kaufmann and Hengartner 2001).
The expression of FasL is preceded and regulated by an increase of the transcription factor c-Myc (Genestier et al. 1999, Kasibhatla et al. 2000, Brunner et al. 2000). Our previous work (Olej et al. 1998) has shown that PHA-activated lymphocytes exposed to ouabain undergo apoptosis, and despite the fact that c-myc mRNA is increased in ouabain treated PHA-activated lymphocytes the expression of FasL in these cells was not investigated. On the other hand, the function of c-Myc in relation to Fas expression is controversial (de Alboran et al. 2001, Wang et al. 1998), and there are evidences that the transcription factor NFAT participate in the regulation of Fas expression in activated lymphocytes (Latinis et al. 1997).
The present work attempted to investigate the expression of Fas and Fas-L in mitogen activated human peripheral lymphocytes exposed to ouabain, a condition known to predispose these cells to apoptosis.
MATERIALS AND METHODS
PREPARATION OF PERIPHERAL BLOOD LYMPHOCYTES
PBL were obtained by fractionating heparinized blood from healthy volunteers on ficoll-hypaque (hystopaque, Sigma-Aldrich) density gradient centrifugation. Briefly, 4ml of blood was layered on 3ml of hystopaque and spun down for 30 min at 400× g. The PBL fraction was washed twice with 0.9% NaCl (7 min at 250× g). Cell pellets were then resuspended at 106 cells/ml in RPMI 1640 (Sigma-Aldrich), supplemented with 5 ×105 M b-mercaptoethanol, 25mM Hepes, pH adjusted to 7.4 with NaOH, 60mg/l penicillin, 100mg/l streptomycin (all purchased from Sigma-Aldrich) and 10% fetal calf serum (FCS) (Gibco), inactivated at 56ºC for 1h. The cell number was adjusted at 106 cells/ml and used in the specific assays.
TREATMENT OF CELLS WITH PHA AND OUABAIN
PBL at 106 cells/ml in supplemented RPMI medium with 10% FCS were incubated with 5µg/ml PHA (Sigma-Aldrich) in 96-well culture plates in a 5% CO2 atmosphere at 37ºC for 6, 24 and 72h, in the presence or absence of a freshly prepared solution of ouabain (100nM or 100µM) (Sigma-Aldrich) in RPMI medium. After this, cells were used for specific assays.
ANALYSIS OF FAS (CD95 OR APO-1) AND FAS LIGAND (FASL OR CD95L) EXPRESSION
Cells (106/ml) incubated for 6, 24, and 72h with or without PHA and/or 100nM ouabain, were spun down and the pellet was incubated with 1µg (2µl) of anti-human FAS-PE (clone number DX2) (BD PharmingenTM) or anti-human FAS-L (clone number NOK-1) (BD PharmingenTM), for 30 min at 4ºC, washed twice in chilled PBS and resuspended in PBS containing 1% formaldehyde. For FasL analysis, after primary incubation, cells were spun down and incubation with a secondary antibody goat anti-mouse IgG-FITC human adsorbed (BD PharmingenTM) Analysis for surface immunofluorescence was performed on a flow cytometer.
MEASUREMENT OF PLASMA MEMBRANE POTENTIAL WITH OXONOL DiBAC4(3)
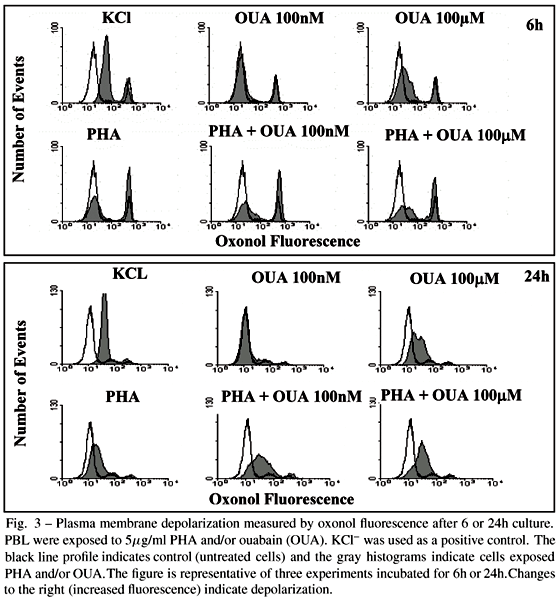
Acute changes in the plasma membrane potential were measured by flow cytometry using bis-(1,3-dibutilbarbiturate) trimethine oxonol DiBAC4 (3) (Molecular Probes). Cells were incubated for 6 and 24h with or without PHA and/or 100nM or 100µM ouabain. Oxonol was prepared according to manufacturers instructions and diluted in RPMI to be used at a final concentration of 150nM. The incubation time with oxonol was 30 min at 37ºC and 5% CO2. As a positive control, we used 50mM KCl in the last 10 min of incubation. Analysis for surface immunofluorescence was performed on a flow cytometer.
MEASUREMENT OF MITOCHONDRIAL MASS WITH MITOTRACKER GREEN
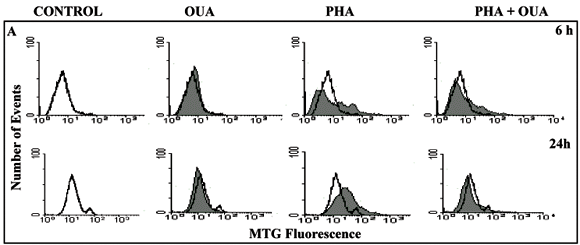
To investigate mitochondrial mass, we used thefluorescent probe mitotracker green FM (MTG) (Molecular Probes) which is a cell-permeant mitochondrion-selective dye that passively diffuses across the plasma membrane and accumulates in active mitochondria. PBL at 106 cells/ml were incubated for 6 and 24h with or without PHA and/or 100nM ouabain. The dye was diluted in DMSO, stored at 20ºC (1mM) and used at a final concentration of 50nM in supplemented RPMI. The incubation time with MTG was 30 min. at 37ºC and 5% CO2. Samples were analyzed by flow cytometry.
EVALUATION OF MITOCHONDRIAL MEMBRANE POTENTIAL WITH 3,3-DIHEXYLOXACARBO-CYANINE IODIDE-DiOC6(3)
To evaluate the mitochondrial membrane potential (DYm), we used 3,3-dihexyloxacarbocyanine iodide [DiOC6(3)] dye (Sigma-Aldrich). Cells were incubated for 6 and 24h with or without PHA and/or 100nM ouabain. The dye was diluted in RPMI and used at a final concentration of 5nM. Cells were incubated with DiOC6(3) for 40 min at 37ºC and 5% CO2. As a positive control, 50µM of the protonophore carbonylcyanide p-trifluoromethoxyphenylhydrazone (FCCP) (Sigma-Aldrich) was used in the last 20 min of incubation. DiOC6(3) and FCCP were diluted in ethanol and stored at 20ºC (1mM). Samples were analyzed using a flow cytometer.
MEASUREMENT OF APOPTOSIS BY ANNEXIN BINDING AND ANTI-FAS
Annexin V conjugated to fluorescein isothiocyanate (FITC) (TACS-Annexin kit FITC, RD systems) was used to determine changes in phosphatidylserine symmetry. 106 cells/ml were incubated for 6 or 24h with or without PHA and 100nM ouabain. At the last 2h of incubation, 1µg of anti-FAS-PE antibody (clone number DX2) (BD PharmingenTM) was added. Cells were spun down and incubated for 30 min at 4ºC with a secondary antibody rat anti-mouse Kappa-PE (clone number X36) (BD PharmingenTM). After this time, cells were spun down again and incubated for 15 min at room temperature in the presence or absence of 1µl Annexin V-FITC (diluted according to the manufacturer instructions). Samples were analyzed by flow cytometer FAS-PE versus Annexin V-FITC.
ANALYSIS BY FLOW CYTOMETRY
106 cells were incubated for the indicated time and cell size and complexity analyzed by flow cytometry using light scatter parameters: forward scattering (FSC) and side scattering (SSC). For analysis using fluorescent probes, samples were kept on ice up to the moment of the analysis. The final volume of incubation was 300µl. All samples were analyzed using a FACScalibur flow cytometer. The fluorescence was excited with an argon laser (excitation wavelength 488nm) and analyzed in FL-1 (wavelength 530±30nm Annexin V, FasL, MTG, DiOC6(3) and oxonol) or in FL-2 (wavelength 585nm CD95PE). At least 10,000 events were acquired. A life-gate based on light scatter parameters (forward and side scatter) and staining with 1µg/ml PI was done. Dead cells, aggregates and debris were excluded. Data were recorded on a Macintosh computer using Cell Quest Software and statistically analyzed using the WinMDI version 2.8 software.
STATISTICAL ANALYSIS
Results were reported as mean ± SE. Data were analyzed statistically by the Student's t-test and differences between means were consider to be significant when P < 0.05.
RESULTS
EFFECT OF OUBAIN ON FAS AND FASL EXPRESSION BY PHA-ACTIVATED LYMPHOCYTES
Mitogen activated cells show an increase in Fas expression. It has been reported that stimulation of these receptors induce the activation of Cl channels in lymphoid Jurkat cells (Szabò et al. 1998, Lepple-Wienhues et al. 1998). In our experiments an early rise of Fas expression could be observed within 6h of incubation on PHA-activated lymphocytes. The addition of 100nM ouabain to cells exposed to PHA did not significantly affect Fas or FasL expression. After 24h of incubation, the levels of Fas expression reached similar levels to those at 72h and no difference could be observed between PHA-activated lymphocytes and activated cultures that received ouabain (Fig. 1 and Table I). FasL was only studied at 6h and similar to what was seen with Fas, no difference could be observed between PHA-activated lymphocytes and activated cultures that received ouabain (Fig. 1 and Table II).
EFFECT OF OUBAIN ON ANTI-FAS-INDUCED APOPTOSIS
It has been demonstrated that ouabain is capable of potentiating some of the effects observed during anti-Fas-induced apoptosis in Jurkat lymphocytes (Bortner et al. 2001, Nobel et al. 2000). In the present work the addition of anti-Fas to PHA-activated lymphocytes cultured in the presence of ouabain, resulted in an increased amount of apoptotic cells. Furthermore, the majority of dead cells were CD95 positive cells (Fig. 2).
EFFECT OF OUBAIN ON PLASMA MEMBRANE DEPOLARIZATION OF PHA-ACTIVATED LYMPHOCYTES
It has been described (Kiefer et al. 1980) that mitogens are capable of inducing plasma membrane depolarization, although the role played by such a process during activation is not fully understood. In other systems, prevention of depolarization using a K+ channel blocker inhibits subsequent apoptosis (Maeno et al. 2000, Dallaporta et al. 1999). Being ouabain an inhibitor of Na+,K+ATPase it could be exerting its effect through plasma membrane depolarization. In our hands, the effect produced by ouabain was dose dependent; 100nM was incapable of inducing plasma membrane depolarization, while 100µM ouabain promoted depolarization. However, both 100nM and 100µM were capable of augmenting the degree of depolarization observed in the presence of PHA (Fig. 3 and Table III). PBL from 6h cultures always presented a smaller second peak that increased when PHA was used and was not formed by macrophages or dead cells measured by propidium iodide incorporation (data not shown).
EFFECT OF OUBAIN ON MITOCHONDRIAL MASS AND MITOCHONDRIA MEMBRANE POTENTIAL
Mitochondrial mass was assessed using mitotracker green dye. As early as 6h post-PHA stimulus there was a 30.3% increase of mitochondrial mass in activated PBL and at 24h this increase was 50%. The addition of 100nM ouabain slightly inhibited the increase seen at 6h and after 24h no increase in mitochondrial mass was observed in cultures stimulated by PHA in the presence of ouabain (Fig. 4A).
Disruption of mitochondrial membrane potential is a characteristic of many apoptotic cells. Using the cationic lipophilic dye DiOC6(3) it was found that by 6h, 100nM ouabain by itself did not induce mitochondrial membrane depolarization. The same occurred with 100µM ouabain (data not shown). Lymphocytes exposed to PHA showed an increase in the fluorescence level of DiOC6(3) compatible with the increase of mitochondrial mass, suggesting that this result might not represent hyperpolarization of mitochondrial membrane potential. After 24h of culture, PHA also induced an increase in DiOC6(3) fluorescence, but the combination PHA + ouabain led to a clear disruption of the mitochondrial membrane potential (Fig. 4B and Table IV).
DISCUSSION
The molecules Fas and FasL are expressed in PHA-activated lymphocytes independent of their exposure to ouabain. Nevertheless, the induction of the apoptotic process using anti-Fas as an agonist, led to an increased amount of apoptotic cells in PHA-activated cultures in the presence of ouabain. In the Jurkat cell line, ouabain potentiates Fas-induced apoptosis and the death program triggered by this molecule (Bortner et al. 2001, Nobel et al. 2000). In a different system, glucocorticoid induced apoptosis of thymocytes, both spontaneous and glucocorticoid-driven apoptosis were dependent on a decrease in Na+/K+ ATPase activity and ouabain potentiated this effect (Mann et al. 2001).
There are evidences that loss of plasma membrane potential without repolarization leads to cell shrinkage and apoptosis (Bortner et al. 2001). The activity of the Na+/K+ pump was not measured in the present study, but the concentration used, 107M ouabain, did not induce plasma membrane depolarization per se but potentiated the depolarization produced by PHA. An increased sensitivity of activated cells to ouabain has been described before (Brodie et al. 1995).
One of the earliest evidences of apoptosis is cell shrinkage, which seems to precede caspase activation and nuclear fragmentation (Bortner et al. 1997). Stimulation of the Fas receptor leads to inhibition of K+ uptake (Bortner et al. 2001) and activates outwardly rectifying Cl channels, via tyrosine kinase phosphorylation (Szàbo et al. 1998). Furthermore, by blocking volume regulatory Cl channels it was possible to rescue cells from apoptosis (Maeno et al. 2000, Szàbo et al. 1998). The release of K+ from the cells, activation of chloride channels and efflux of water produce the characteristic loss of volume (Bortner and Cidlowski 1996, Bortner et al. 1997).
A central role for K+ ions in apoptosis has been advanced. Hughes et al. (1997) described that reduced intracellular K+ concentration is a pre-requisite for apoptosis as normal concentrations of this ion inhibits caspase and nuclease activities. This reduction, however, is not sufficient to trigger the apoptotic process, an apoptotic stimuli being necessary. It is worth mentioning that ouabain was shown to increase the production of IL-1b. The enzyme capable of cleaving pro-interleukin 1b to the biologically active form is the caspase ICE (IL-1b converting enzyme) and its activation was reported to be enhanced by K+ depletion (Perregaux and Gabel 1994, Walev et al. 1995). It is quite possible therefore, that the processes of caspase activation for the induction of apoptosis and for IL-1b production are a reflection of the same mechanism.
A different view is put forward by Dallaporta et al. (1998). According to them loss of cytosolic K+ occurs downstream of mitochondrial depolarization, cytochrome c release and caspase activation, but they also consider this step crucial for the activation of endonucleases. In our experience, loss of mitochondrial membrane potential was clearly evident only when PHA-activated cells were exposed to ouabain and appeared to be a late event.
In conclusion, treatment of activated PBL with ouabain potentiated apoptosis induced by an anti-Fas agonist antibody. A synergy between ouabain and PHA was also observed with regard to plasma membrane depolarization in agreement with the observation that sustained plasma membrane depolarization favors apoptosis. The mitochondria pathway, resulting in depolarization and cytochrome c release, did not seem to be involved when only PHA was used. However, dissipation of mitochondrial membrane potential was observed when cells were also exposed to ouabain, and in this case it was a late event. It is possible that the inhibitory effect produced by ouabain on activated peripheral blood lymphocytes represents an exacerbation of the effects seen during AICD.
ACKNOWLEDGMENTS
We would like to thank Dr Fernanda Mello de Queiroz for the gift of oxonol dye and for introducing us to the technique of measurement of plasma membrane potential.
This work was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Programa de Apoio a Núcleos de Excelência (PRONEX) and Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ).
Manuscript received on January 14, 2005; accepted for publication on March 7, 2005; contributed by VIVIAN M. RUMJANEK*
- ANNER BM, LACOTTE D, ANNER RM AND MOOSMAYER M. 1994. Interaction of hypothalamic Na,K-ATPase inhibitor with isolated human peripheral blood mononuclear cells. Biosci Rep 14: 231-242.
- BORTNER CD AND CIDLOWSKI JA. 1996. Absence of volume regulatory mechanism contributes to the rapid activation of apoptosis in thymocytes. Am J Physiol 271: C950-C961.
- BORTNER CD, HUGHES FM AND CIDLOWSKI JA. 1997. A primary role for K+ and Na+ efflux in the activation of apoptosis. J Biol Chem 272: 32436-32442.
- BORTNER CD, GÓMEZ-ANGELATS M AND CIDLOWSKI JA. 2001. Plasma membrane depolarization without repolarization is an early molecular event in anti-Fas-induced apoptosis. J Biol Chem 276: 4304-4314.
- BRODIE C, TORDAI A, SALOGA J, DOMENICO J AND GELFAND EW. 1995. Ouabain induces inhibition of the progression phase in human T-cell proliferation. J Cell Physiol 165: 246-253.
- BRUNNER T, KASIBHATLA S, PINKOSKI MJ, FRUTSCHI C, YOO NJ, ECHEVERRI F, MAHBOUBI A AND GREEN DR. 2000. Expression of Fas ligand in activated T cells is regulated by c-Myc. J Biol Chem 275: 9767-9772.
- DALLAPORTA B, HIRSCH T, SUSIN SA, ZAMZAMI N, LAROCHETTE N, BRENNER C, MARZO I AND KROEMER G. 1998. Potassium leakage during the apoptotic degradation phase. J Immunol 160: 5605-5615.
- DALLAPORTA B, MARCHETTI P, PABLO MA, MAISSE C, DUC H-T, MÉTIVOER D, ZAMZAMI N, GEUSKENS M AND KROEMER G. 1999. Plasma membrane potential in thymocytes apoptosis. J Immunol 162: 6534-6542.
- DE ALBORAN IM, O'HAGAN RC, GARTNER F, MALYNN B, DAVIDSON L, RICKET R, RAJEWSKY K, DEPINHO RA AND ALT FW. 2001. Analysis of C-Myc function in normal cells via conditional gene-targeted mutation. Immunity 14: 45-55.
- GENESTIER L, KASIBHATLA S, BRUNNER T AND GREEN DR. 1999. Transforming growth factor b1 inhibits Fas ligand expression and subsequent activation-induced cell death in T cells via downregulation of c-Myc. J Exp Med 189: 231-239.
- HUGHES FM, BORTNER CD, PURDY GD AND CIDLOWSKI JA. 1997. Intracellular K+ suppresses the activation of apoptosis in lymphocytes. J Biol Chem 272: 30567-30576.
- JENSEN P, WINGER L AND NOWELL P. 1977. The mitogenic effect of A23187 in human peripheral lymphocytes. Biochim Biophys Acta 496: 374-383.
- KASIBHATLA S, BEERE HM, BRUNNER T, ECHEVERRI F AND GREEN DR. 2000. A 'non-canonical' DNA-binding element mediates the response of the Fas-ligand promoter to c-Myc. Curr Biol 10: 1205-1208.
- KAUFMANN SH AND HENGARTNER MO. 2001. Programmed cell death: alive and well in the new millennium. Trends Cell Biol 11: 526-534.
- KIEFER H, BLUME AJ AND KABACK HR. 1980. Membrane potential changes during mitogenic stimulation of mouse spleen lymphocytes. Proc Natl Acad Sci USA 77: 2200-2204.
- LATINIS KM, NORIAN LA, ELIASON SL AND KORETZKY GA. 1997. Two NFAT transcription factor binding sites participate in the regulation of CD95 (Fas) ligand expression in activated human T cells. J Biol Chem 272: 31427-31434.
- LEPPLE-WIENHUES A, SZABÒ I, LAUN T, KABA NK, GULBINS E AND LANG F. 1998. The tyrosine kinase p56 mediates activation of swelling-induced chloride channels in lymphocytes. J Cell Biol 141: 281-286.
- LYNCH DH, RAMSDELL F AND ALDERSON MR. 1995. Fas and FasL in the homeostatic regulation of immune responses. Immunol. Today 16: 569-574.
- MAENO E, ISHIZAKI Y, KANASEKI T, HAZAMA A AND OKADA Y. 2000. Normotonic cell shrinkage because of disordered volume regulation is an early prerequisite to apoptosis. Proc Natl Acad Sci USA 97: 9487-9492.
- MANN CL, BORTNER CD, JEWELL CM AND CIDLOWSKI JA. 2001. Glucocorticoid-induced plasma membrane depolarization during thymocyte apoptosis: association with cell shrinkage and degradation of Na/K-adenosine triphosphatase. Endocrinology 142: 5059-5068.
- MARTIN SJ AND GREEN DR. 1995. Protease activation during apoptosis: death by a thousand cuts? Cell 82: 349-352.
- MORAES VLG, OLEJ B, DE LA ROCQUE L AND RUMJANEK VM. 1989. Lack of sensitivity to ouabain in natural killer activity. FASEB J 3: 2425-2429.
- NAGATA S AND GOLDSTEIN P. 1995. The Fas death factor. Science 267: 1449-1456.
- NOBEL CSI, ARONSON JK, VAN DEN DOBBELSTEEN DJ AND SLATER AFG. 2000. Inhibition of Na+/K(+)-ATPase may be one mechanism contributing to potassium efflux and cell shrinkage in CD95-induced apoptosis. Apoptosis 5: 153-163.
- OLEJ B, DE LA ROCQUE L, CASTILHO FPD, MEDIANO IF, CAMPOS MM AND RUMJANEK VM. 1994. Effect of ouabain on lymphokine-activated killer cells. Int J Immunopharmacol 16: 769-774.
- OLEJ B, DOS SANTOS NF, LEAL L AND RUMJANEK VM. 1998. Ouabain induces apoptosis on PHA activated lymphocytes. Bioscience Reports 18: 1-7.
- ORLOV SN, THORIN-TRESCASES N, KOTELEVTSEV SV, TREMBLAY J AND HAMET P. 1999. Inversion of the intracellular Na+/K+ ratio blocks apoptosis in vascular smooth muscle at a site upstream of caspase-3. J Biol Chem 274: 16545-16552.
- PERREGAUX D AND GABEL CA. 1994. Interleukin-1 beta maturation and release in response to ATP and nigericin. Evidence that potassium depletion mediated by these agents is a necessary and common feature of their activity. J Biol Chem 269: 15195-15203.
- PIRES V, HARAB RC, OLEJ B AND RUMJANEK VM. 1997. Ouabain effects on activated lymphocytes: augmentation of CD25 expression on TPA-stimulated cells and of CD69 on PHA- and TPA-stimulated cells Int J Immunopharmac 19: 143-148.
- QUASTEL MR AND KAPLAN JG. 1968. Inhibition by Ouabain of human lymphocyte transformation induced by phytohaemagglutinin in vitro. Nature 219: 198-200.
- REDONDO JM, LOPEZ RIVAS A AND FRESNO M. 1986. Activation of the Na+/K+-ATPase by interleukin-2. FEBS Lett 206: 199-202.
- SKOU JC. 1990. The fourth Datta lecture: the energy coupled exchange of Na+ for K+ across the cell membrane. The Na+, K+-pump. FEBS Lett 268: 314-324.
- SZABÒ I, LEPPLE-WIENHUES A, KABA NK, ZORATTI M, GULBINS E AND LANG F. 1998. Tyrosine kinase-dependent activation of a chloride channel in CD95-induced apoptosis in T lymphocytes. Proc Natl Acad Sci USA 95: 6169-6174.
- SZAMEL M, SCHEIDER S AND RESCH K. 1981. Functional interrelationship between (Na+,K+)-ATPaseand lysolecithin acyltransferase in plasma membranes of mitogen-stimulated rabbit thymocytes. J Biol Chem 256: 9198-9204.
- SZAMEL M, LEUFGEN H, KURRLE R AND RESCH K. 1995. Differential signal transduction pathways regulating interleukin-2 synthesis and interleukin-2 receptor expression in stimulated human lymphocytes. Biochim Biophys Acta 1235: 33-42.
- WALEV I, RESKE K, PALMER M, VALEVA A AND BHAKDI S. 1995. Potassium-inhibited processing of IL-1 beta in human monocytes. EMBO J 14: 1607-1614.
- WANG R, BRUNNER T, ZHANG L AND SHI Y. 1998. Fungal metabolite FR901228 inhibits c-Myc and Fas ligand expression. Oncogene 17: 1503-1508.
Publication Dates
-
Publication in this collection
09 May 2005 -
Date of issue
June 2005
History
-
Received
14 Jan 2005 -
Accepted
07 Mar 2005