Abstracts
Analytical procedures for U-Pb isotope dilution analyses at the Pará-Iso isotope geology laboratory of the Federal University of Pará (UFPA) are described in detail. The procedures are applied to zircon, titanite, rutile, apatite, columbite-tantalite and whole rock. Reagent preparation and chemical processing are done in clean-room conditions. Samples are dissolved using TeflonTM microcapsules in steel jacket TeflonTM Parr InstrumentTM bomb or TeflonTM screw cap containers. U and Pb are separated using anion exchange AG 1x8 resin columns. Typical blanks for mineral sample amounts of 0.01 to 1.0 mg are less than 1 pg U and 20-30 pg Pb. Isotope analysis of the U and Pb from the same filament are carried out using a Finnigan MAT 262 mass-spectrometer in static and dynamic modes. The current analytical level is demonstrated on analyses of international standard zircon 91500 with three different 235U-205Pb and 235U-208Pb isotope tracers and whole rock standards. Results of analyses of two zircon samples are also presented.
U-Pb dating; zircon; accessory mineral; isotope dilution
Os procedimentos analíticos para análises U-Pb por diluição isotópica no Laboratório de Geologia Isotópica (Pará-Iso) da Universidade Federal do Pará (UFPA) são descritos detalhadamente. Esses procedimentos são aplicados para análises de zircão, titanita, rutilo, apatita, columbita-tantalita e rocha total. A purificação dos reagentes e os procedimentos químicos são feitos em salas limpas. As amostras são dissolvidas em microcápsulas de Teflon em bombas do tipo Parr InstrumentTM. U e Pb são separados em colunas com resina de troca iônica AG 1x8. Os brancos de procedimento para amostra típica(0,01-1 mg) são menores que 1 pg de U e 20-30 pg de Pb. As análises isotópicas de Pb e de U são feitas em um único filamento de Re em um espectrômetro de massa Finnigan MAT 262 nos modos estático e dinâmico. O nível analítico atual é comprovado pelas análises do padrão internacional de zircão 91500, usando três diferentes traçadores de U-Pb e nas análises de padrões de rocha total. Resultados geocronológicos obtidos em cristais de zircão de duas amostras de rochas arqueana e paleozóica são também apresentados e comentados.
geocronologia U-Pb; zircão; minerais acessórios; diluição isotópica
EARTH SCIENCES
Uranium-lead dating method at the Pará-Iso isotope geology laboratory, UFPA, Belém - Brazil
Robert S. Krymsky; Moacir J.B. Macambira; Jean-Michel Lafon; Gérson S. Estumano
Universidade Federal do Pará (UFPA), Pará-Iso, Centro de Geociências Rua Augusto Corrêa 01, Caixa Postal 8608, 66075-110 Belém, PA, Brasil
ABSTRACT
Analytical procedures for U-Pb isotope dilution analyses at the Pará-Iso isotope geology laboratory of the Federal University of Pará (UFPA) are described in detail. The procedures are applied to zircon, titanite, rutile, apatite, columbite-tantalite and whole rock. Reagent preparation and chemical processing are done in clean-room conditions. Samples are dissolved using TeflonTM microcapsules in steel jacket TeflonTM Parr InstrumentTM bomb or TeflonTM screw cap containers. U and Pb are separated using anion exchange AG 1x8 resin columns. Typical blanks for mineral sample amounts of 0.01 to 1.0 mg are less than 1 pg U and 20-30 pg Pb. Isotope analysis of the U and Pb from the same filament are carried out using a Finnigan MAT 262 mass-spectrometer in static and dynamic modes. The current analytical level is demonstrated on analyses of international standard zircon 91500 with three different 235U-205Pb and 235U-208Pb isotope tracers and whole rock standards. Results of analyses of two zircon samples are also presented.
Key words: U-Pb dating, zircon, accessory mineral, isotope dilution.
RESUMO
Os procedimentos analíticos para análises U-Pb por diluição isotópica no Laboratório de Geologia Isotópica (Pará-Iso) da Universidade Federal do Pará (UFPA) são descritos detalhadamente. Esses procedimentos são aplicados para análises de zircão, titanita, rutilo, apatita, columbita-tantalita e rocha total. A purificação dos reagentes e os procedimentos químicos são feitos em salas limpas. As amostras são dissolvidas em microcápsulas de Teflon em bombas do tipo Parr InstrumentTM. U e Pb são separados em colunas com resina de troca iônica AG 1x8. Os brancos de procedimento para amostra típica(0,01-1 mg) são menores que 1 pg de U e 20-30 pg de Pb. As análises isotópicas de Pb e de U são feitas em um único filamento de Re em um espectrômetro de massa Finnigan MAT 262 nos modos estático e dinâmico. O nível analítico atual é comprovado pelas análises do padrão internacional de zircão 91500, usando três diferentes traçadores de U-Pb e nas análises de padrões de rocha total. Resultados geocronológicos obtidos em cristais de zircão de duas amostras de rochas arqueana e paleozóica são também apresentados e comentados.
Palavras-chave: geocronologia U-Pb, zircão, minerais acessórios, diluição isotópica.
INTRODUCTION
The installation of U-Pb method began in October 1999 at the Pará-Iso laboratory of isotope geology (Federal University of Pará, Brazil). Before this time, the radiometric methods performed in routine were Rb-Sr, Sm-Nd, Pb-Pb on sulfide, feldspar and whole-rock, and Pb-Pb zircon evaporation. During the first two years, the U-Pb clean room infrastructure was constructed and the necessary equipments were acquired. For base procedure, it was used the methodology of Krogh (1973, 1982) for U-Pb analysis of zircon and the technique of Tera and Wasserburg (1975) and Chen and Wasserburg (1981) for analysis of whole rock. During 2000-2001, U-Pb techniques for analyses of whole rock and multigrain fractions of zircon were installed. Along 2002-2003, U-Pb methods were developed for the analysis of single grain of zircon and accessory minerals (apatite, titanite, rutile, tantalite and others). During the first two years, a 235U-208Pb tracer has been used for determination of U and Pb concentrations; the following year we used a 235U-205Pb tracer prepared at the University of Brasília, and after, a new 235U-205Pb tracer was prepared in the Pará-Iso laboratory. Some improvements of analytical procedures aiming blank reduction are in course.
MINERAL SEPARATION
The laboratory for mineral separation is equipped with DensitestTM and turbulent water flux tube for pre-concentration of heavy mineral fraction, heavy liquids for final concentration of heavy minerals and FrantzTM L-1 separator for separation of magnetic fractions. The minerals are extracted from specimens weighing from 3 to 80 kg by crushing, grinding, followed by a specific separation scheme for each type of mineral.
For zircon separation, rock fraction of 125-250µm is passed through turbulent water flux for pre-concentration of heavy minerals. Another way for heavy mineral pre-concentration for zircon separation is using Densitest® device. For the grain size of 90-125µm, the conditions are inclination 9º, pulsating voltage12V, air flux voltage 7.5V. For a grain size between 125 and 250µm, the conditions are: inclination 9º, pulsating voltage 13V, air flux voltage 9.0V. After that, heavy minerals are separated with bromoform and washed in ethanol. After separation of highly magnetic minerals by hand magnet, the non-magnetic fraction is separated by the Frantz magnetic separator with a frontal inclination of 20-25º and successive lateral inclinations of15º (0.5 A), 15º (1.0 A), 15º (1.2 A), 10º (1.2 A),5º (1.2 A) and 1-5º (1.2 A). Finally, individual zircon grains are handpicked with needle under the binocular microscope. Selected grains are loaded in small drop of immersion oil (ND = 1.515) on glass plate for digital photo imaging using a Leica DMLP microscope equipped with DC 300F camera.
Some zircon (and other mineral) fraction or individual grains can be polished in an air abrasion mill with pyrite powder following the technique of Krogh (1982). A pressure of 1.8 psi for zircon abrasion and of 1.2 psi for titanite and rutile abrasion is normally used. The time of abrasion varies between 20-60 min, depending on mineral size and resistance. Abraded grains are leached in warm 2-3 N HNO3 until complete dissolution of pyrite.
Another micro air abrasion mill with a chamber of 10 mm diameter and 5 mm high (design of A.N. Komarov, IPGG RAS, Russia) is used for a rapid abrasion of single crystals of zircon without using pyrite powder (pressure: 0.1-0.2 psi, duration: 30-80 sec).
CLEAN LAB FACILITIES
The clean chemistry laboratory consists of four separate rooms for (1) U-Pb chemistry, (2) acid distillation and water purification, (3) Rb-Sr, common Pb and Sm-Nd chemical procedures and (4) balances. All these rooms have temperature and humidity control, positive air pressure and high efficiency particulate air (HEPA) filters. These clean rooms are equipped with custom-made organic glass hoods, clean boxes with their own HEPA filters and air source and, laminar flow hoods (VECO® HLFS-09M) with class 100 HEPA filters. Laboratory air is not recirculated, and minimum metal is exposed.
The weighting room is equipped with Sartorius® CS-2 and R180D balances. The CS-2 balance has quoted precision of ±0.1 µg, linearity of ±0.9 µg with a maximum limit of 2.1 g. This balance is used to weight small quantity of mineral ( < 2 mg) and tracer (1-10 mg). To weight smaller fractions (1-500µg) and single grains, a small hand-made aluminum boat (1-1.5 mg) is used; for bigger mass of sample (0.5-2 mg), microcapsules of Teflon (model of Ludwig) are directly used. The R180D balance, with precision of ± 0.02 mg and mass limit of 33 g is used for 30-100 mg of mineral or whole rock samples.
For dissolution of zircon and some other minerals by method of Parrish (1987), we use Parr Instrument Company steel bomb (Model 4748) with 120 mL PTFE Teflon liner, equipped with two floor PTFE Teflon holder for 12 PFA Teflon microcapsules of Ludwig model (#7226, Savillex®). For other chemical operation (lower temperature dissolution, collection of U, Pb during ion exchange separation etc.), 3 mL and 6 mL Savillex® screw cup vessels are used. Centrifugation is performed in polypropylene 1.5 and 2 mL Eppendorf® centrifuge tubes and 0.2 mL PCR® tubes. Acid are stored in FEP Nalgene® bottles and drop dispensers.
REAGENT PURIFICATION
Water purification is carried out in three main steps: 1) pre-cleaning with cellulose filters; 2) Millipore® RiOs-5 system (pre-cleaning, reverse osmosis), and 3) Millipore® Milli-Q Academic final treatment (organic absorption, mixed-bed deionization, and 0.22 micron membrane filtration). The lead and uranium concentrations in water from our system are 1-3 pg/mL and < 0.1 pg/mL, respectively.
The different inorganic acids of Merk® (''for analysis'' quality) are multi-step distilled. We have three quartz sub-boilers and four Savillex® PFA Teflon bottle-to-bottle sub-boilers with heating blankets. Each acid is distilled in different ways: HCl is passed twice trough quartz sub-boiler and once trough the SavillexTM sub-boiler; HNO3 was cleaned only by triple distillation; HBr was distilled 4 times into a quartz sub-boiler; HF was treated 3 times into a SavillexTM sub-boiler, using principles of distillation by Mattinson (1972). The lead and uranium concentrations in purified concentrated acids are, respectively: HCl: 5 pg/mL and < 0.1 pg/mL; HNO3: 7 pg/mL and < 0.1 pg/mL; HF: 5 pg/mL and 0.2 pg/mL, and HBr: 13 pg/mL and < 0.1 pg/mL. Silica gel and H3PO4 mixture was obtained from Max-Planck-Institute Mainz for Chemistry and used for mass-spectrometric analysis of Pb and U (3-5 µL for each sample; blank of 2 pg of Pb and 0.01 pg of U). This mixture was added in SavillexTM collection beakers before evaporation of samples on hot plate.
U-Pb SPIKES AND STANDARDS
In modern U-Pb geochronological laboratories, different types of spike are widely used: 235U-208Pb, 235U-205Pb, 233U-235U-202Pb-205Pb and 233U-235U-230Th-205Pb. The spike with artificial 205Pb isotope was firstly used by Krogh and Davis (1975b). The use of 235U-205Pb spike, instead of 235U-208Pb, allows reducing by half the quantity of columns for chemical separation and in three times the quantity of filament locations in mass-spectrometer, which gives a great advantage in economy of time and materials. Other advantages of 205Pb are a reduction of pollution introduced by aliquoting of sample and an increasing precision of lead isotope analysis.
For determination of U and Pb concentrations by isotope dilution analysis, the Pará-Iso laboratory has two 235U-208Pb mixed spike solutions named ''IPGG'' and ''A'', respectively from the Institute of Precambrian Geology and Geochronology (St. Petersburg, Russia) and prepared in our laboratory and two 235Pb-205Pb spike solutions (''B'' from the University of Brasília and ''PARA'', prepared in our laboratory).
The 235U-208Pb spike is used for U -Pb analysis of whole rock and sulfide samples and any quantity of mineral fractions higher than 2 mg. The characteristics of spike ''A'' are: [235U] = 9.19 nmol/g, [208Pb] = 3.96 nmol/g, 235U/238U = 7766 and 206Pb/208Pb = 0.000096. The 235U-205Pb spike is useful for small fractions (0.001-2 mg) of zircon and other U-rich minerals. The characteristics of spike ''PARA'' are: [205Pb] = 0.0863 nmol/g, [235U] = 7.87 nmol/g, 206Pb/205Pb = 0.000704, 238U/235U = 0.0000123 and 235U/205Pb = 91.19. Spikes ''A'' and ''PARA'' were calibrated using 5 ppm solution of NBS983 and 10 ppm solution of natural U, which were previously calibrated with ''IPGG'' and ''B'' spikes. Finally, calibration of spikes were verified and adjusted by analyses of the 91500 international standard zircon.
For determination of mass fractionation of Pb and U in the mass spectrometer, 90 ng of NBS982 and178 ng of NBS500 standards are used. For calibration of the ion counter efficiency in relation to that of Faraday cup, 220 ng of NBS983 is used in every magazine.
The isotope ratios for standards during 2000-2003 period were:
NBS-982 (25 analyses):
204Pb/206Pb = 0.027216 (±0.015%),
207Pb/206Pb = 0.467013 (±0.006%) and
208Pb/206Pb = 0.999829 (±0.005%);
NBS-983 (46 analyses):
204Pb/206Pb = 0.000368 (±0.170%),
207Pb/206Pb = 0.071213 (±0.007%) and
208Pb/206Pb = 0.013623 (±0.042%);
NBS-U500 (14 analyses):
238U/235U = 1.00037 (±0.015%)
(errors at 2s level; data corrected from mass-fractionation). All measured values for NBS-982, NBS-983 and NBS-U500 standards agree with standard values. Thallium interference was controlled on 203Tl peak by periodic scanning of mass spectrum in 200-210 mass range. At work temperature of 1430ºC, all Tl from sample was usually gone.
DISSOLUTION AND CHEMICAL SEPARATION OF Pb AND U
ZIRCON ANALYSIS
Selected grains, previously photographed and abraded (Krogh 1982), are transferred to small PTFE Teflon beakers. In some special cases, zircon grains can be step-wise leached (Krogh and Davis 1975a, Mattinson 1994, 1997, Corfu 2000, Davis and Krogh 2000). The Mattison (1994) technique is preferred for complex and metamictic grains. In usual analysis, samples are consecutively washed in bi-distilled methanol with ultrasonic bath (20 min), hot (90ºC) 7N HNO3 (30 min) and rinsed 3 times with pure water. After drying, they are weighed in aluminum foil boat using CS-2 balance and loaded in PFA Teflon microcapsules. When using 235U-205Pb tracer, 1-5 µl of ''PARA'' spike are added into each microcapsule and weight on the same balance. When the 235U-208Pb tracer is used, we add it after zircon dissolution and aliquoting.
In each microcapsule, 10 µl or 30-50 µl of hydrofluoric acid are introduced, respectively for single grain and for multi grain fraction. A Set of 12 microcapsules is arranged into a 120 ml PTFE Teflon bomb enclosed in a stainless steel jacket Model 4748 Parr Instrument, following the method outlined by Parrish (1987). Five ml of HF are added to the large PTFE bomb and the whole assembly is closed and heated 24 h in an oven at 240-245ºC. The complete dissolution of the zircon grains is checked under a stereomicroscope and the sample solutions evaporated to dryness. Then, the fluoride salts are digested overnight at 200ºC with 30-60 µL of 3N HCl in the capsules and 5 ml in the large bomb.
There are 5 main ways to separate U and Pb from zircon solution:
1) direct analysis without separation of Pb and U (Lancelot et al. 1976, Van Schmus and Chemale Jr. 1996);
2) classical method of Pb-U separation on columns with anionic resin AG 1x8 in HCl and H2O (Krogh 1973, Roddick et al. 1987, Parrish et al. 1987);
3) the Pb-U separation on columns with mixture of specific resins TRU Spec and Sr Spec of EichromTM in HCl (Paquette and Pin 2001);
4) single bead separation of Pb by resin AG 1x8 in 0.6 M HBr (Manton 1988), and
5) separation on columns with AG 1x8 in HBr, HCl for Pb and HNO3 and H2O for U (Tera and Wasserburg 1975).
The Pará-Iso laboratory uses either the Parrish et al. (1987) or the Paquette and Pin (2001) methods, both with modifications.
Description of modified method of Parrish et al. (1987)
For this procedure hand-made shrinkable Teflon micro-columns with internal diameter of 1.8 mm and height of 11 mm with reservoir for 1.5 ml (diameter is 6 mm) are used. The columns are washed with hot HNO3 and hot water for 3 days and they are filled with 40 µl of Dowex® 1x8 200-400 mesh resin, previously washed. When new, the resin is washed 20-40 times with 6N HCl and H2O, alternately. Between analyses, the columns are washed 8-10 times with 6N HCl and H2O. The resin usually is not disposable because of long period of pre-washing and it can be used up to 20 times, if the lead blank does not increase. For counting the acid volume, 30 ml NalgeneTM drop-dispensers are used, which drop is about 30 µl. The sequence of elements separation with consists of:
Resin media equilibration: 2x3 drops of 3N HCl;
Loading in column: 3N HCl sample solution;
Sample washing: 1+2+2 drops of 3N HCl;
Pb elution: 10 drops of 6N HCl (collection in 3 ml PFA Teflon beaker);
U elution: 10 drops of H2O (collection in the same beaker as Pb);
Addition of 5 µl of silica gel-H3PO4 mixture and evaporation;
Sample loading on Re filament.
Total blanks for this zircon procedure are 20-30 pg of Pb and 0.1-0.5 pg of U.
Description of Paquette and Pin (2001) modified method
For this technique, PFA Teflon micro-columns (Savillex model #7272) are used, with 8 mm capillary (internal diameter = 1.5 mm) and 2 ml reservoir. The columns are washed in the same way as for the procedure described above. The resin mixture consists of 0.5 g of Eichrom Sr Specific resin (50-100 µ) and 1.5 g of Eichrom TRU Specific resin (50-100 µ). After being pre-washed in 10 ml plastic column with 300 ml three-distilled 6N HCl, about 22-25 µL of this mixture are introduced into each micro-column in 6N HCl media. The resin mixture serves only for one separation procedure. The sequence of elements separation is the following:
Resin washing: 2 ml of 6N HCl;
Resin media equilibration: 2x2 drops of 2.5N HCl;
Loading in column: 2.5N HCl sample solution;
Sample washing: 1+1+2+2+2 drops of 2.5N HCl;
Pb elution: 4 drops of 6N HCl (collection in microcapsule);
U elution: 4 drops of 0.25N HCl + 0.3N HF mixture (collection in the same microcapsule) and evaporation;
Sample loading on Re filament with 3 µl of silica gel-H3PO4 mixture.
Total blanks for this zircon procedure are 4 pg of Pb and 0.05 pg of U. This procedure also can be applied for analysis of other accessory minerals (titanite, rutile, apatite), but separation of Ca and Fe from the sample is not ideal.
After analysis, the PFA Teflon beakers are washed on hot plate at 90ºC in HF, H2O, HNO3, H2O, HCl and H2O during 1 day for each step. Microcapsules are washed in similar manner, but with three-distilled HF and HCl in Parr bomb at 230ºC additional washing, during one night with each acid.
OTHER ACCESSORY MINERALS ANALYSIS
Depending on the characteristics of accessory mineral, we different procedures of washing are applied. Apatite is washed with methanol, 5% Na-EDTA solution and water. Rutile, titanite, garnet, other oxides and silicates are washed with methanol, 0.5-2N HCl and water. The mineral fractions may be previously polished by air abrasion mill without pyrite powder.
The U-Pb analysis of mineral small quantities ( < 1 mg) is carried out with 235U-205Pb tracer. For bigger samples the 235U-208Pb tracer is utilized. For digestion of 10-50 mg of apatite we use 14N HNO3 in 3 ml PFA Teflon closed beakers. Parrish-type PFA Teflon microcapsules (#7235 SavillexTM) on hot plate at 100ºC during 6-8 h are used for digestion of 0.2-1 mg samples. For dissolution of oxides and silicates it is used Ludwig-type or Parrish-type PFA Teflon microcapsules or 3 ml PFA Teflon screw-cup vials (depending on sample weight) inside Parr bomb. HF or a HF+HNO3 mixture (from 60 µl up to 500 µl) is used in the microcapsules or vials. Then, the Parr bomb liner is filled with 5 ml HF and introduced in an oven for 15-20 h at 245ºC. Samples are evaporated, and re-equilibrated with 3N HCl during 10h at 200ºC in Parr bomb. In case of using 235U-208Pb spike, aliquoting is made and spike is added after dissolution. Some minerals, such as carbonates, phosphates and Fe oxides are dissolved in HCl or HCl-HNO3 mixture in the 3 ml PFA Teflon screw-cup vials at 100ºC on hot plate.
Depending on U and Pb concentrations and weight of samples, micro-columns (the same as for zircon separation) or small columns (120 µl of AG 1x8 resin) are used. The micro-column is used for 0.001-1 mg samples, whereas the small column is used for 1-50 mg samples. Small amounts of titanite, apatite and monazite are processed in micro-columns using the same technique used for zircon (with HCl and H2O). The oxides and silicate mineral solutions are separated with HBr using a modified technique of Tera and Wasserburg (1975). In the case of HBr medium, the chloride solution is evaporated, 10 µl of concentrated HBr are added and evaporated to dry; then 3-5 drops of 0.6N HBr are added. The HBr solution, after being heated on hot plate at 100ºC, stay for about 20 min in ultrasonic bath and is centrifuged in 0.2 ml PCR® tube. The Pb separation sequence is the following:
Resin media equilibration: 2x3 drops of 0.6 N HBr;
Loading: 0.6 N HBr sample solution / collecting solution for U separation;
Sample washing: 2+2+2+10 drops of 0.6N HBr / collecting solution for U separation;
Pb elution: 10 drops of 6N HCl (collection in a PFA Teflon 3 ml beaker);
Addition of 5 µl of silica gel-H3PO4 mixture and evaporation.
Uranium can be separated by the same method as mentioned for zircon separation with HCl and H2O or by the following HNO3 technique:
Evaporation of solution and conversion of the salts to nitrate by 2 drops 14N HNO3, evaporation;
Addition of 5 drops of 7N HNO3, transfer to microcapsule, heating for 30 min at 100ºC and centrifugation in PCR® tube;
Column washing 3 times with H2O and 6N HCl;
Resin media equilibration: 3x5 drops of 7N HNO3;
Loading: 7N HNO3 sample solution;
Sample washing: 2+2+2+4 drops of 7N HNO3;
U elution: 15 drops of 0.25N HNO3 (collection in a 3 ml PFA Teflon beaker);
Addition of 3 µl of silica gel-H3PO4 mixture in a beaker with U and evaporation.
The current blanks for total procedure (digestion, separation with HBr-HNO3 technique and deposition) for accessory mineral are 60 pg of Pb and 0.1 pg of U. When using the same analytical procedures for accessory mineral as for zircon analysis, the total blanks are < 30 pg for Pb and < 0.5 pg for U. The next step for Pará-Iso laboratory will be the implantation of Paquette and Pin (2001) U-Pb separation technique for accessory minerals dating, which will permit to obtain Pb blank level smaller than 10 pg, using current acid quality.
WHOLE ROCK ANALYSIS
Fifty mg powdered whole rock samples can be dissolved by different ways, depending on their mineralogical composition: in Teflon steel jacket bombs at 150ºC in an oven; in 6 ml PFA Teflon (Savillex®) screw-cup vials at 150ºC in an oven or on hot plate, or in special PTFE Teflon bombs in microwave oven. Ordinary digestion in oven normally takes 3-7 days and, in some cases, even more. For dissolution in Savillex® vial it is used 3 ml of HF+HNO3 (5:1) mixture.
In case of microwave digestion, a BOLA-digestion vessel with PTFE Teflon liner (A250-04 model; Bohlender GmbH, Germany) is used with 4 ml HF+HNO3 (3:1) mixture. The microwave digestion conditions are: 36 min at 400 W; 3 min cooling, 15 min at 400 W; 3 min cooling. After digestion first steps, the sample is evaporated, and the procedure is repeated. Then the solution is evaporated, 3 ml 6N HCl are added and the sample is processed in the microwave oven at the same conditions as for the HF+HNO3 mixture.
After dissolution and cooling, the solution is weighed and splitted in two aliquots: 2/3 volume for isotope composition and 1/3 volume for U-Pb concentration. The concentration aliquot is spiked with 235U-208Pb tracer ''A''. Both aliquots are evaporated, 0.6 ml of concentrated HBr are added and evaporated again.Then, 1.2 ml of 0.6N HBr is added to sample beakers, passed in ultrasonic bath, heated in closed beakers at 100ºC for 2 hours, and centrifuged.
The separation of Pb and U are carried out on small columns (internal diameter = 3 mm, height = 20 mm and reservoir = 1.5 ml) filled with 120 µl of AG 1x8 resin (200-400 mesh). The Pb separation sequence is the following:
Resin media equilibration: 2x3 drops of 0.6 N HBr;
Loading: 0.6 N HBr sample solution / collecting solution for U separation;
Sample washing: 1+0.5 mL drops of 0.6N HBr / collecting solution for U separation;
Pb elution: 1.5 mL of 6N HCl (collection in a PFA Teflon 3 mL beaker);
Addition of 5 µL of silica gel-H3PO4 mixture in a beaker with Pb and evaporation.
The U separation procedure using the same columns is following:
Evaporation of solution for U separation, conversion of the salts to nitrate by 10 drops 14N HNO3 and evaporation;
Addition of 40 drops 7N HNO3, transfer to beaker, heating for 30 min at 100ºC and centrifugation in 2 ml tube;
Washing column: 3 times with H2O and 6N HCl;
Resin media equilibration: 3x10 drops of 7N HNO3;
Loading: 7N HNO3 sample solution;
Sample washing: 1+5+5+5 drops of 7N HNO3;
U elution: 90 drops of 0.25N HNO3 (collection in PFA Teflon 6 ml beaker);
Addition 3 µl of silica gel-H3PO4 mixture and evaporation.
The blanks for total whole rock procedure are 0.5 ng of Pb and 10 pg of U.
MASS SPECTROMETRY
Isotopic compositions of Pb and U are measured on a Finnigan MAT 262 variable multicollector mass spectrometer equipped with 7 Faraday cups and 1 ion counter. The numbers of Faraday cups are 2, 3, 4, 5, 6, 7 and 8, the ion counter position corresponding to the cup 5.
The lead-uranium fraction mixed with the silica gel-phosphoric acid solution is loaded under HEPA laminar flow on a single outgassed rhenium filament. The transfer of the sample to the filament is done using 1 µl pure water. For procedure with Eichrom resins, the sample is loaded to filament by 3 µl of silica gel - phosphoric acid mixture. The filament is heated slowly from 0.8 to 2.2 A, until glow rose color.
Different configurations of Faraday cups and ion counter are utilized for the lead analysis with different spikes and different signal levels. In case of 235U-208Pb tracer application, the analysis is carried out in static mode. The signals of masses 206, 207 and 208 are registered on the Faraday cups 4, 3 and 2, while the signal of 204 mass is received on the ion counter. In case of using 235U-205Pb tracer, the configuration of this 7-collector mass-spectrometer does not permit the analysis of all necessary lead mass in a static mode, because only three cups adjacent to ion counter (IC) are available in one side. Therefore, the following dynamic mode is applied: first run - 204 (IC), 205 (cup 4), 206 (cup 3) and 207 (cup 2) (measured ratios are 206/204, 206/205 and 206/207); second run - 206 (cup 4), 207 (cup 3) and 208 (cup 2) (measured ratio is 206/208). In case of low signal of lead (I206 < 10 mV) all masses are analyzed on ion counter in peak jumping mode. The ion counter gain is calibrated in each magazine with NBS-983 standard by ''IC-deflection'' operation and adjusted for 206/204 ratio by comparison with the same ratio, obtained on Faraday cups. The filament with sample is heated during 10 min and analyzed at temperature interval of 1400-1500ºC with maximal signal at 1470ºC.The total time required for acquisition of 5 blocks of data is about 30 min. The mass fractionation correction for lead is 0.12% a.m.u.-1, calculated from 25 analyses of NBS 982 standard.
The U is analyzed as UO2 just after lead measurement from the same filament, by measurement of masses: 267 (235U16O2) and 270 (238U16O2). Depending on signal level, UO2 is analyzed in static mode on Faraday cups (#6 for 267 and #5 for 270) or peak jumping mode on ion counter. The initial temperature for UO2 measurement is 1500-1530ºC and maximal temperature is 1570ºC. The time for collection of 3 blocks is 15 min. The mass fractionation correction for UO2 was not applied because of its insignificant small value ( ~ 0.002-0.03% a.m.u.-1) obtained from NBS-500 analyses.
DATA REDUCTION
The first step of statistical data reduction is carried out using the custom made laboratory software named ''Finres2'' and developed by Thomas Scheller. This program calculates the measured isotope ratios and their 2 sigma errors. After that, the data set is processed by PbDatprogram (version 1.24; Ludwig 1993), which give Pb/U and Pb/Pb isotope ratios corrected for blanks, spike contribution and common lead. Finally, the data are processed with Isoplot for Excel program (version 2.49e; Ludwig 2001) and plotted in the concordia diagram. Errors on final results are quoted at a 2s level.
The measured lead isotope composition of total blank in the Pará-Iso laboratory is generally close to common lead isotope composition by Stacey and Kramers (1975) model for t = 0 Ma. Therefore, for most zircon samples, the correction of measured lead on contamination by common lead by Stacey and Kramers (1975) model gives satisfactory results. For zircon and accessory minerals with measured 206Pb/204Pb ratio < 1000 it is necessary to do distinct correction on blank and sample common lead, which can be estimated from Stacey and Kramers (1975) model for adequate age or from the K-feldspar lead isotope composition.
The current precision of determination of U and Pb concentration is 1% (2gs), precision of Pb/U isotope ratios is 0.5-0.1 % (2s). The best precision of U-Pb age determination achieved in the laboratory is 1 Ma for 91500 standard zircons and for some other zircons, which have not been included in the paper as, for example, for zircons from Mid Atlantic ridge.
STANDARD ZIRCON 91500 DATA
For testing the U-Pb procedures installed in our lab, we used the international standard 91500 Canadian zircon. The international standard 91500 is a concordant zircon with an age of 1065.4 ± 0.3 Ma and concentrations of U and Pb of 71-86 ppm (mean value: 81.2 ppm) and 13-16 ppm (mean value: 14.8 ppm), respectively (Wiedenbeck et al. 1995). Three sets of analyses of this standard were carried out using three different U-Pb spikes. The results are displayed in Table I.
The first test was performed using the 235U-208Pb ''IPGG'' spike. The chemical procedures include spiking after digestion and aliquoting. U-Pb results from five fragments yielded an upper intercept age of 1065.1 ± 0.8 Ma (Fig. 1), which is good agreement with previously published values for this standard. Some variations on the Pb/U ratios are observed, which are probably due to the high content of common lead or to the fact that the zircon fragments were not polished in the air-abrasion device.
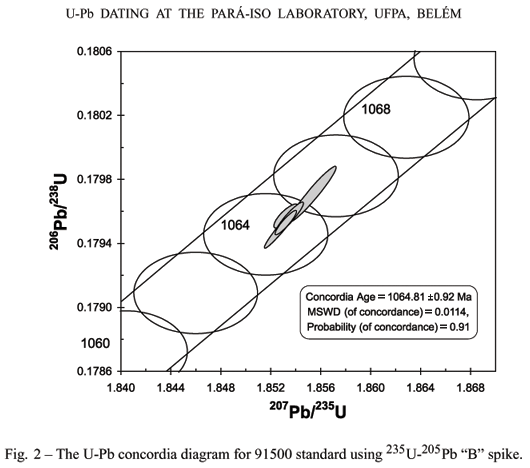
The second test was made with 235U-205Pb ''B'' spike. The chemical procedures include spiking before digestion. The zircon grains were abraded. The weights of analyzed samples were slightly lower than those of the first test. All the analytical points are concordant and an U-Pb Concordia age of 1064.8 ± 0.9 Ma (Fig. 2) was obtained from four zircon fragments, which is undistinguishable from the accepted value for this standard.
The third test was made with 235U-205Pb ''PARA'' spike. Zircon fragments were also abraded and spiking was done before digestion. The U-Pb Discordia age calculated from four zircon fragments is 1066.7 ± 1.3 Ma (Fig. 3), which is similar within errors to the age obtained by Wiedenbeck et al. (1995) and Paquette and Pin (2001). The larger scattering of data ellipses relatively to those on figure 2 is possibly caused by not ideal calibration of ion counter gain, or by too strong washing of zircon fragments with HNO3.
The ages calculated from all three sets of 91500 standard are excellent agreement within error limit with the standard value previously published (Wiedenbeck et al. 1995). However, as it was shown on these three figures, the best results in respect to reproducibility of Pb/U isotopic ratios and concordance are provide by 235U-205Pb spike in comparison with 235U-208Pb spike. Also, it is necessary to underline the variations in concentrations of U (70-92 ppm) and Pb (13-17 ppm), which slightly exceed the interval yielded by Wiedenbecket al. (1995).
ANALYSES OF STANDARDS OF GRANITE GH AND BASALT BHVO-1
In order to test the 238U-208Pb ''A'' tracer for determination of the concentration of U and Pb contents in whole rock samples we analyzed the international USGS standards of granite (GH) and basalt (BHVO-1). The first standard has high U and Pb concentrations, while the second has low concentrations of these elements. The values of concentration for these standards were determined by analytical procedures described above for whole rock samples, and compared with values published by Govindaraju (1989) (see Table II). The mass of standards used for analyses was about 17-18 mg. The obtained values are similar to those of Govindaraju (1989), which permit to conclude that the installed procedures are sufficient for a precise determination of U and Pb contents in whole rock samples.
EXAMPLES OF ANALYSES OF ZIRCONS
Zircon crystals from two samples of different ages have been selected to test the capability of the Pará-Iso laboratory to furnish confident U-Pb data. One sample came from previously dated metavolcanic rocks of the Carajás Formation (Grão Pará Group) from the Neoarchean meta-volcano-sedimentary sequences of the northern Carajás Province, eastern Amazonian craton, Brazil. The regional geological setting of the Carajás Province and the main geological features of the Carajás Formation have been exhaustively described (Machado et al. 1991, Macambira and Lafon 1995, Nogueira et al. 1995, Pinheiro and Holdsworth 1997, Trendall et al. 1998, Tassinari et al. 2000). The second sample came from a granitic body of the Proterozoic Khanka Lake superterrain (Far East, Russia). This specimen was collected during regional geological survey of 1999-2000 field seasons. Regional geological background can be found elsewhere (Khanchuk et al. 1995, Rub and Rub 1988, Ryazantseva 1976). The age of this intrusion was suggested to be approximately 480 Ma. based on K-Ar measurements (M.D. Ryazantseva, pers. communication).
The considered examples show the capability of the Pará-Iso Laboratory to analyze multi and single grain zircon samples of different ages with appropriate precision.
THE ARCHEAN METAVOLCANIC ROCK OF GRÃO PARÁ GROUP, CARAJÁS PROVINCE, BRAZIL
One sill of volcanic rock, concordant with layers of the Band Iron Formation in the base of Carajás Formation (Grão Pará Group) was collected from the N4E mine. This rock is strongly weathered, but has numerous euhedral crystals of quartz and feldspar in the hematite - kaolin matrix. According to the classification scheme of Pupin (1980, J.P. Pupin, unpublished data), this rock was probably a trachyte. Zircons of sample #10979 are prismatic (elongation 2.4) with rectangular or square section, bi-pyramidal, transparent, rose color, without zoning and containing dark spherical inclusions.
The analyses U-Pb of multigrain fractions (10-20 crystals in each fraction) were carried out with 235U-208Pb ''IPGG'' tracer. A first test showed very low 206Pb/ 204Pb ratio (60-300), therefore the following analyses were made with previous leaching using a mixture of acids 10N HF + 14N HNO3 (30 min., 100ºC) and by 6N HCl (30 min, 100ºC). After leaching, the zircon crystals were colorless, and the dark inclusions disappeared. The 206Pb/204Pb ratio increased to 500-5000, showing that an effective leaching of common lead occurred. The results of U-Pb analyses of zircon fractions are shown in Table III. The seven analytical points defined a discordia line with an upper intercept at 2751 ±4 Ma (Fig. 4), 207Pb/206Pb ages varied between 2730-2752 Ma. The age 2751 ±4 Ma is interpreted as crystallization age for the volcanic rock; this age is similar to the age of 2757 ±18 Ma obtained by the Pb-evaporation method(Macambira et al. 1996) and to the U-Pb age 2743 ±11 Ma achieved by ion microprobe SHRIMP-II (Trendall et al. 1998) for the same sample.
THE PALEOZOIC GRANITE OF NEVSKY MASSIF, KHANKA REGION, RUSSIA
Sample #3054 was collected from a biotite granite of Nevsky massif, Khanka Lake region, Far East, Russia. Zircons are clear, rose-brown with well developed prismatic and pyramid faces. Some crystals show zoning and cracks; gas-liquid inclusions are rare. The crystal 3054-10 has short prismatic shape; others have long prismatic form. The single zircon grains, weighting from 2.5 to 9.4 mg, were analyzed using 235U-205Pb tracer. These zircons are characterized by high uranium (3200-5600 ppm) and lead (260-410 ppm) concentrations(Table IV). The 206Pb/204Pb ratio varies from 685 to 1654. The four analytical points lie on a discordia line with an upper intercept at 482.5 ±1.8 Ma (Fig. 5), and a lower intercept at about zero (-22 ± 88 Ma). Point 3054-10 lies on the concordia curve. 207Pb/206Pb ages varied between 481-484 Ma. The age of 482 Ma was interpreted as crystallization age of Nevsky granite massif. Such ages were obtained for different granite bodies in this region mainly by the Rb-Sr method on whole-rock samples and interpreted as a time of main Paleozoic magmatic activity in the Khanka terrain (Gerasimov et al. 1990, Govorov et al. 1994, Ryazantseva et al. 1995, Khetchikov et al. 1994).
CONCLUSIONS
During the time period between 2000 and 2003, the U-Pb experimental procedures, including mineral separation, chemical digestion, chromatography extraction and mass spectrometry measurements, were installed at the Pará-Iso laboratory and the U-Pb method presently runs in routine. The procedures are useful for dating of whole-rock, multi- and single-zircon and accessory minerals (titanite, rutile, apatite and columbite-tantalite). The analyses of NBS-982, NBS-983, NBS-U500, 91500 zircon and whole-rock international standards showed results which are in good agreement with the accepted values of U and Pb concentrations and Pb isotope composition for these standards. The experimental procedures for U-Pb geochronology have been purposely described in details for the benefit of other laboratories in Brazil and of the customers using or evaluating the data produced in Pará-Iso laboratory. The U-Pb method increases considerably the capability of the Federal University of Pará to utilize geochronology and isotope geology as research tools in a wide variety of Earth science projects. An improvement of this analytical technique in the laboratory is currently under progress with testing procedure for U and Pb separation with TRU-Sr specific Eichrom resins for accessory mineral dating. The reduction of blank level is also a challenge for the laboratory.
ACKNOWLEDGMENTS
This work was supported by grant of Conselho Nacional de Desenvolvimento Científico e Tecnológico - CNPq (nº 300795/01-7). Thanks are due to anonymous reviewers for their constructive comments which significantly improved the paper. This is a contribution to Programa de Apoio a Núcleos de Excelência (PRONEX) 103-98 project ''Magmatismo, metalogênese e evolução crustal da Província Mineral de Carajás e províncias adjacentes''.
Manuscript received on March 3, 2005; accepted for publication on March 22, 2006; presented by ALCIDES N. SIAL
Correspondence to: Jean Michel Lafon
E-mail: lafonjm@ufpa.br
- CHEN JH AND WASSERBURG GH. 1981. Isotopic determinations of uranium in picomole and sub-picomole quantities. Anal Chem 53: 2060-2067.
- CORFU F. 2000. Extraction of Pb with artificially too-old ages during stepwise dissolution experiments on Archean zircon. Lithos 53: 279291.
- DAVIS DW AND KROGH TE. 2000. Preferential dissolution of 234 U and radiogenic Pb from alpha-recoil damaged sites in zircon: implications for thermal histories and Pb isotopic fractionation. Chem Geol 172: 41-58.
- GERASIMOV NS, RODIONOV SM AND KOMPANICHENKO VN. 1990. Rb-Sr age dating of tin-bearing granites from the Central Sikhote-Alin. Reports of USSR Academy of Sciences 312: 1183-1185.
- GOVINDARAJU K. 1989. Compilation of working values and sample description for 272 geostandards. Geost Newsl 16: 111-300.
- GOVOROV IN, GERASIMOV NS AND RYAZANTSEVA MD. 1994. Rb-Sr isochrons and petrogenetic patterns of magmatic rocks from the Voznesenka ore district, Primorye. Geol Pacific Ocean 4: 60-73.
- KHANCHUK AI, RATKIN VV, RYAZANTSEVA MD, GOLOZUBOV VV AND GONOKHOVA NG. 1995. Geology and mineral deposits of Primorsky Krai, Dalnauka, Khanchuk ed., Vladivostok, Russia, 65 p.
- KHETCHIKOV LN, GERASIMOV NS AND PAKHOMOVA VA. 1994. Rb-Sr age data and results of quartz inclusions study of Grigor'evsky granite complex, Khanka Laketerrain. Reports of the Russian Academy of Sciences 343: 91-93.
- KROGH TE. 1973. A low contamination method for hydrothermal decomposition of zircon and extraction of U and Pb for isotopic age determination. Geochim Cosmochim Acta 37: 485-494.
- KROGH TE. 1982. Improved accuracy of U-Pb zircon ages by the creation of more concordant systems using air abrasion technique. Geochim Cosmochim Acta 46: 636-649.
- KROGH TE AND DAVIS GL. 1975a. Alteration in zircons and differential dissolution of altered and metamict zircon. Carnegie Inst Wash Yearb 73: 560-567.
- KROGH TE AND DAVIS GL. 1975b. The production and preparation of 205Pb for use as tracer for isotope dilution analyses. Carnegie Inst Wash Yearb 75: 416-417.
- LANCELOT JR, VITRAC A AND ALLEGRE CJ. 1976. Uranium and lead isotopic dating with grain by grain zircon analysis: a study of complex geological history with a single rock. Earth Plan Sci Lett 29: 357-366.
- LUDWIG KR. 1993. PBDAT. A Computer program for processing Pb-U-Th isotope data. Version 1.24. United State Geological Survey Open File Report 88-542, USA, 34 p.
- LUDWIG KR. 2001. User's manual for Isoplot/ Ex Version 2.49. A geochronological toolkit for Microsoft Excel. Berkeley Geochronology Center, Berkeley, CA, USA,Spec Pub 1a, 59 p.
- MACAMBIRA MJB AND LAFON JM. 1995. Geocronologia da Província Mineral de Carajás. Síntese dos dados e novos desafios. Bol Mus Para Emílio Goeldi, Série Cięncias da Terra 7: 263-288.
- MACAMBIRA JB, MACAMBIRA MJB, SCHELLER T AND GOMES ACB. 1996. Geocronologia Pb/Pb e tipologia de zircőes de rochas vulcânicas da formaçăo Carajás-Pará;indicador da idade dos BIF. In: CONGRESSO BRASILEIRO DE GEOLOGIA 39, Salvador, BA, Brasil 6: 516-519.
- MACHADO N, LINDENMAYER Z, KROGH TE AND LINDENMAYER D. 1991. U-Pb geochronology of Archean magmatism and basement reactivation in the Carajás area, Amazon shield, Brazil. Precambrian Research 49: 329-354.
- MANTON WI. 1988. Separation of Pb from young zircons by single-bead ion exchange. Chem Geol 73: 147-152.
- MATTINSON JM. 1972. Preparation of hydrofluoric, hydrochloric and nitric acids at ultralow lead levels. Anal Chem 44: 1715-1716.
- MATTINSON JM. 1994. A study of complex discordance in zircons using step-wise dissolution techniques. Contrib Mineral Petrol 116: 117-129.
- MATTINSON JM. 1997. Analysis of zircon by multi-step partial dissolutions: the good, the bad, and the ugly. Geol Assoc Can Meet, Ottawa, Abstract, A98.
- NOGUEIRA ACR, TRUCKENBRODT W, COSTA JBS AND PINHEIRO RVL. 1995. Formaçăo Águas Claras, Pré-Cambriano da Serra dos Carajás. Redescriçăo e redefiniçăo. Bol Mus Para Emilio Goeldi, Série Cięncias da Terra 7: 177-197.
- PAQUETTE JL AND PIN C. 2001. A new miniaturized extraction chromatography method for precise U-Pb zircon geochronology. Chem Geol 176: 311-319.
- PARRISH RR. 1987. An improved micro-capsule for zircon dissolution in U-Pb geochronology. Chem Geol 66: 99-102.
- PARRISH RR, RODDIK JC, LOVERIDGE WD AND SULLIAN RW. 1987. Uranium-lead analytical techniques at the geochronology laboratory, Geological Survey of Canada. In: RADIOGENIC AGE AND ISOTOPIC STUDIES: Report 1, Geological Survey of Canada, Paper 87-2: 3-7.
- PINHEIRO RVL AND HOLDSWORTH RE. 1997. The structure of the Carajás N-4 ironstone deposit and associated rocks: relationship to Archaean strike-slip tectonics and basement reactivation in the Amazon region, Brazil. J South Amer Earth Sci 10: 105-119.
- PUPIN JP. 1980. Zircon and granite petrology. Contrib Mineral Petrol 73: 207-220.
- RODDICK JC, LOVERIDGE WD AND PARRISH RR. 1987. Precise U/Pb dating of zircon at the sub-nanogram Pb level. Chem Geol (Isot Geosci Sect) 66: 111-121.
- RUB M AND RUB AK. 1988. New data referring geological and geochemical peculiarities of magmatic intrusions within Khanka region. Geol Pacific Ocean 5: 57-67.
- RYAZANTSEVA MD. 1976. Comparative characteristics of the Voznesenka and Grodekovo granites of the south part of the Khanka terrain. Igneous rocks of the Eastern Asia, Vladivostok, Far East Scientific Center, USSR Academy of Sciences, p. 69-73.
- RYAZANTSEVA MD, GERASIMOV NS AND GOVOROV IN. 1995. Rb-Sr isotopes and petrogenesis of igneous rocks in the Voznesenka ore district, Primorye. Geol Pacific Ocean 11: 579-596.
- STACEY JS AND KRAMERS JD. 1975. Approximation of terrestrial lead isotope evolution by a two-stage model. Earth Planet Sci Lett 26: 207-221.
- TASSINARI CCG, BETTENCOURT JS, GERALDES MC, MACAMBIRA MJB AND LAFON JM. 2000. The Amazonian Craton. In: CORDANI UG ET AL. (Eds), Tectonic Evolution of South America, 31 Int Geological Congress, p. 41-96.
- TERA F AND WASSERBURG GJ. 1975. Precise isotopic analysis of lead in picomole and subpicomole quantities. Anal Chem 47: 2214-2220.
- TRENDALL AF, BASEI MAS, DE LAETER JR AND NELSON DR. 1998. SHRIMP zircon U-Pb constraints on the age of Carajás formation, Grăo Pará Group, Amazon Craton. J South Amer Earth Sci 11: 265-277.
- VAN SCHMUS WR AND CHEMALE JR F. 1996. U-Pb isotope dilution analyses of single zircon grains without chemical separation. In: CONGRESSO BRASILEIRO DE GEOLOGIA 39, Salvador, BA, Brazil 6: 617-619.
- WIEDENBECK M, ALLE P, CORFU F, GRIFFIN WL, MEIER M, OBERLI F, VON QUADT A, RODDICK JC AND SPIEGEL W. 1995. Three natural zircon standards for U-Th-Pb, Lu-Hf, trace element and REE analyses. Geostandards Newsletter 191: 1-23.
Publication Dates
-
Publication in this collection
23 Mar 2007 -
Date of issue
Mar 2007
History
-
Accepted
22 Mar 2006 -
Received
03 Mar 2005