Abstracts
Background and the purpose of the study: The amount of elder people who suffer from Alzheimer disease is continuously increasing every year. Cholinesterase inhibitors have shown to be effective in alleviating the symptoms of the disease, thus opening a field of research for these treatments. Herbal products, owning a reputation as effective agents in many biological studies are now drawing attention for inhibiting acetylcholinesterase, in other words, Alzheimer disease. In the present study, the ability of three sesquiterpene lactones from Inula oculus-christi and I. aucheriana to inhibit AChE has been evaluated through Ellman assay.
Materials and Methods: Gaillardin and pulchellin C were obtained from I. oculus-christi and britannin from I. aucheriana by chromatographic methods. They were dissolved in methanol in concentration of 3 mg/mL and the AChEI activity of the compounds was determined by Ellman method using Acethylthiocholine iodide as the substrate and 5, 5′-dithiobis-2-nitrobenzoic acid as the reagent, in 96-well plates at 405 nm.
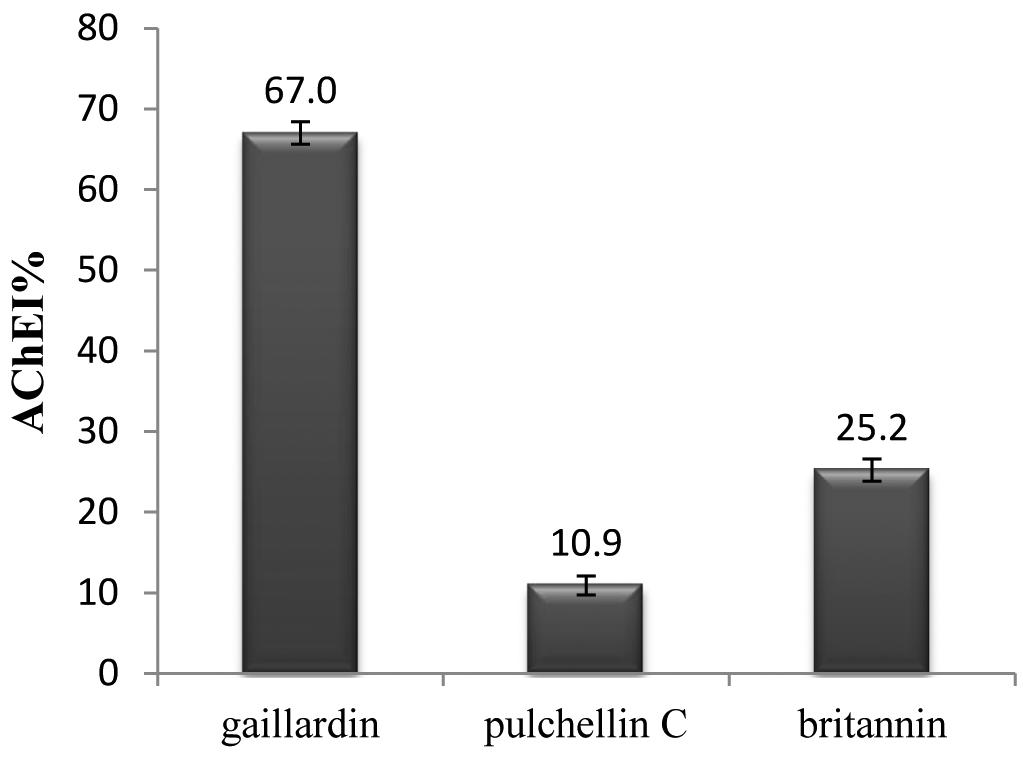
Results: AChEI activity of the examined compounds was obtained as 67.0, 25.2 and 10.9% in concentration of 300 µg/L for gaillardin, britannin and pulchellin C, respectively.
Conclusion: Among the three sesquiterpene lactones, gaillardin with 67% inhibition of AChE could be considered a good candidate for future Alzheimer studies.
Acetylcholinesterase inhibitor; Alzheimer; britannin; gaillardin; Inula ; pulchellin C
Embasamento teórico e proposta de estudo: A quantidade de pessoas idosas que sofrem da doença de Alzheimer está aumentando a cada ano. Inibidores de colinesterase têm mostrado serem eficazes em amenizar os sintomas da doença, abrindo, então, um campo de pesquisa para estes tratamentos. Produtos de origem vegetal, que possuem uma reputação como agentes eficazes em muitos estudos biológicos, ultimamente vem chamando a atenção por inibirem a acetilcolinesterase (AChE), em outras palavras, a doença de Alzheimer. No presente estudo, a capacidade de três lactonas sesquiterpênicas de Inula oculus - christi e I. aucherianapara inibir a AChE foi avaliada pelo ensaio de Ellman.
Materiais e Métodos: Gailardina e pulchelina C foram obtidas a partir de I. oculus - christi e a britanina de I. aucheriana por métodos cromatográficos. Elas foram dissolvidas em metanol na concentração de 3 mg/mL e a atividade AChEI dos compostos foi determinada pelo método de Ellman usando iodeto de acetiltiocolina como substrato e o ácido 5, 5′ - ditiobis - 2 - nitrobenzoico como o reagente, em placas de 96 poços a 405 nm.
Resultados: A atividade AChEI dos compostos avaliados foi de 67,0, 25,2 e 10,9% na concentração de 300 mg/mL para gailardina, britanina e pulchelina C, respectivamente.
Conclusão: Entre as três lactonas sesquiterpênicas, gailardina com 67% de inibição da atividade de AChE pode ser considerada uma boa candidata para futuros estudos na doença de Alzheimer.
inibidor de acetilcolinesterase; Alzheimer; britanina; gailardina; Inula ; pulchelina C
INTRODUCTION
The type of dementia that elder people often suffer, is called Alzheimer disease (AD) and influences the life of more than 25 million a year (D'Addario et al. 2012). The number is rapidly growing, which clearly determines the importance of the need for a careful diagnosis and the handling of the illness (Farlow and Lilly 2005Farlow MR and Lilly ML. 2005. Rivastigmine: an open-label, observational study of safety and effectiveness in treating patients with Alzheimer's disease for up to 5 years. BMC Geriatrics 5. doi:10.1186/1471-2318-5-3
https://doi.org/10.1186/1471-2318-5-3...
). Commonly, AD begins with a gradual worsening of the ability to remember new information. As damage expands, other difficulties occur, such as memory loss, confusion with time or place, misplacing things and losing the ability to retrace steps as well as a decreased or poor judgment. The cause of AD is still unknown but like other common chronic conditions, it progresses as a result of several factors and a variety of brain changes that begin as many as 20 years before symptoms show up (Alzheimer's Association 2012). Based on the cholinergic hypothesis, acetylcholinesterase inhibitors (AChEIs) are widely used to alleviate AD. Nowadays investigations to find AChEI materials from natural sources have increased and it has been proven that many compounds of different structures have AChEI property; among which alkaloids are the most ruling compounds. Agents such as the alkaloid galanthamine, are examples possessing the mentioned property (Feitosa et al. 2011Feitosa CM, Freitas RM, Luz NNN, Bezerra MZB and Trevisan MTS. 2011. Acetylcholinesterase inhibition by somes promising Brazilian medicinal plants. Braz J Biol 71: 783-789.).There are not as many reports about AChEI activity of sesquiterpenes as alkaloids, but the existing data are indicative of the mentioned activity in some herbs and their sesquiterpene constituents. The results of isolating sesquiterpene lactones from Amberboa ramosa, a plant of Asteraceae, have revealed their excellent AChEI activity (Ibrahim et al. 2013Ibrahim M, Farooq T, Hussain N, Hussain A, Gulzar T, Hussain I, Akash MS and Rehmani FS. 2013. Acetyl and butyryl cholinesterase inhibitory sesquiterpene lactones from Amberboaramosa. Chem Cent J 7: 116.); while the leaves essential oil of Hedychium gardnerianum has exhibited AChEI activity (Arruda et al. 2012Arruda M, Viana H, Rainha N, Neng NR, Rosa JS, Nogueira JM and Barreto M. 2012. Anti-acetylcholinesterase and antioxidant activity of essential oils from Hedychium gardnerianum Sheppard ex Ker-Gawl. Molecules 17: 3082-3092) recommending the oil to be used in aromatherapy. Since the constituents were mostly found to be sesquiterpenes, the authors had concluded that the AChEI activity was due to the presence of these compounds. The n-hexane fractions of the peels and leaves of bitter orange have also demonstrated to be inhibitors of AChE and gas chromatography-mass spectrometry analysis have again deduced the most dominant compounds to be mono- and sesquiterpenes (Loizzo et al. 2012Loizzo MR, Tundis R, Bonesi M, Menichini F, de Luca D, Colica C and Menichini F. 2012. Evaluation of Citrus aurantifolia peel and leaves extracts for their chemical composition, antioxidant and anti-cholinesterase activities. J Sci Food Agric 92: 2960-2967.). These results attracted our attention to the possibility of other sesquiterpenes to encompass the ability to inhibit AChE and in the present study, sesquiterpene lactones isolated from two species of Inula (Asteraceae) named I. oculus-christi L. and I. aucheriana DC. have been investigated through Ellman assay.
MATERIALS AND METHODS
Chemicals
Acetylcholinesterase (AChE) was purchased from Sigma, Germany. Acetylthiocholin Iodide (ATCI) was prepared from Fluka, Germany. 5, 5′-dithiobis-2-nitrobenzoic acid (DTNB) and other solvents and chemicals were provided from Merck, Germany.
Plant Material
In June 2009, Inula oculus-christi L. was collected from Golestan province and Inula aucheriana DC. was collected from West Azerbaijan province in Iran (July 2010). The species were authenticated by Mrs. Atefeh Pirani (Botanist), Traditional Medicine and Materia Medica Research Center (TMRC), Shahid Beheshti University of Medical Sciences, Tehran, Iran. Voucher specimens of both species have been deposited at TMRC Herbarium for future reference. The aerial parts of the plants were dried in shade and ground.
Extraction and Isolation
Gaillardin and pulchellin C were obtained from I. oculus-christi (Mosaddegh et al. 2010Mosaddegh M, Hamzeloo-Moghadam M, Ghafari S, Naghibi F, Ostad SN and Read RW. 2010. Sesquiterpene lactones from Inula oculus-christi. Nat Prod Commun 5: 511-514.); briefly, the chloroform extract of 250 g of the aerial parts was further subjected to column chromatography to afford gaillardin and pulchellin C. Britannin was obtained from chloroform fraction of 500 g I. aucheriana, through serial extraction with n-hexane, chloroform and methanol (Hamzeloo-Moghadam et al. 2012Hamzeloo-Moghadam M, Hajimehdipoor H, Saeidnia S, Atoofi A, Shahrestani Read RW and Mosaddegh M. 2012. Anti-proliferative activity and apoptotic potential of britannin, a sesquiterpene lactone from Inulaaucheriana, Nat Prod Commun 7: 979-980.). The chemical structures of the compounds are presented in figure 1.
Acetylcholinesterase Inhibitory Activity Assay
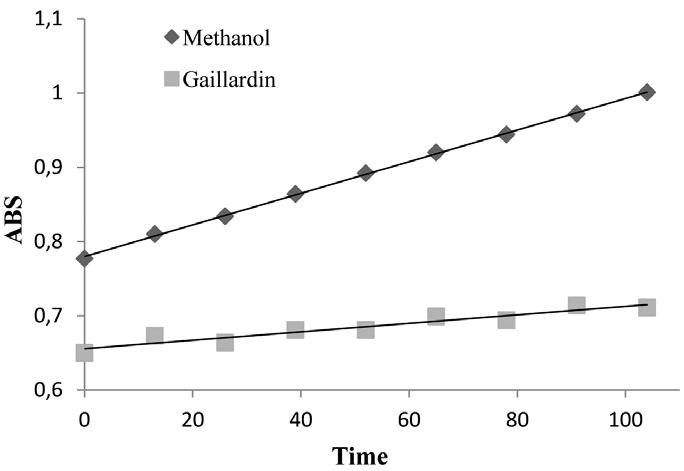
Ellman assay is usually applied in vitro to define the cholinesterase activity of materials. It was established by Ellman et al. (1961)Ellman GL, Courtney KD, Andres Jr V and Featherstone RM. 1961. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7: 88-95. and is based on the reaction of thiocholine (one of the products of enzymatic hydrolysis of acetylthiocholine) with DTNB (Ellman's reagent), forming a yellow product (5-mercapto- 2-nitrobenzoic acid and its dissociated forms) at pH 8 which can be detected at 405 nm (Komersova et al. 2007Komersova A, Komersa K and Cegan A. 2007. New Findings about Ellman's Method to Determine Cholinesterase Activity Z. Naturforsch 62c: 150-154.). Based on the above mentioned assay, the experiment was conducted according to modified microplate method (Mukherjee et al. 2007Mukherjee PK, Kumar V and Houghton PJ. 2007. Screening of Indian Medicinal Plants for Acetylcholinesterase Inhibitory Activity. Phytother Res 21: 1142-1145.). All samples were dissolved in methanol in concentration of 3 mg/mL. In the 96-well plates, 125 µL of 3 mM DTNB, 25 µL of 15 mM ATCI and 50 µL of phosphate buffer (pH 8), 25 µL of sample dissolved in methanol were added to the wells. The absorbance was read at 405 nm every 13 s for 65 s. 25 µL of 0.22 U/mL of AChE enzyme was then added and the absorbance was again measured every 13 s for 104 s using a TECAN microplate reader at 405 nm. Absorbance was plotted against time and enzyme activity was calculated from the slope of the line and expressed as a percentage compared to an assay using a methanol without any inhibitor. Any increase in absorbance due to the spontaneous hydrolysis of the substrate was corrected by subtracting the rate of the reaction before adding the enzyme, from the rate after adding the enzyme. Inhibition percentage was obtained by comparing the rates for the sample to the blank (methanol). Donepezil was used as the positive control.
RESULTS AND DISCUSSION
Acetylcholinesterase inhibitors (AChEIs) as treatments for AD, are the first class of agents approved by the US Food and Drug Administration (FDA). They are thought to bind to acetylcholinesterase in the synaptic cleft, giving more time to acetylcholine which has been released from the pre-synaptic cholinergic terminal; thus increasing the chance of interacting with the postsynaptic cholinergic receptors (Aisen et al. 2012Aisen PS, Cummings J and Schneider LS. 2012. Pharmacologic Treatment for Alzheimer Disease. Cold Spring Harb Perspect Med 2:a006395. doi: 10.1101cshperspect. a006935). The administration of AChEIs in some clinical trials have shown advantages in approximately 40– 50% of patients, though the effect was temporary. There is also a suggestion of neuroprotective effect of these treatments (Kemp et al. 2003Kemp PM, Holmes C, Hoffmann S, Wilkinson S, Zivanovic M, Thom J, Bolt L, Fleming J and Wilkinson DG. 2003. A randomised placebo controlled study to assess the effects of cholinergic treatment on muscarinic receptors in Alzheimer's disease. J Neurol Neurosurg Psychiatry 74: 1567-1570.).
Plants have a reputation of being rich sources of effective compounds for drug discovery and might have advantages regarding efficacy (Frydman-Marom et al. 2011Frydman-Marom A, Levin A, Farfara D, Benromano T and Scherzer-Attali R. 2011. Orally Administrated Cinnamon Extract Reduces b-Amyloid Oligomerization and Corrects Cognitive Impairment in Alzheimer's Disease Animal Models PLoS ONE 6: e16564. doi:10.1371/journal.pone.0016564
https://doi.org/10.1371/journal.pone.001...
). Physostigmine, galanthamine and huperzine A are the alkaloid-type of compounds isolated from the plants which serve as medicines for AD (Orhan et al. 2009Orhan G, Orhan I, Subutay-Öztekin N, Ak F and Sener B. 2009. Contemporary anticholinesterase pharmaceuticals of natural origin and their synthetic analogues for the treatment of alzheimer's disease. Recent Pat CNS Drug Discov 4: 43-51.). Most researches on acetylcholinesterase inhibitory effect of natural sources have been focused on alkaloids, regarding other groups of natural compounds, as next rates. Some examples of acetylcholinesterase inhibitors include steroidal alkaloids from Sarcoccasa ligna(Atta-ur-Rahman et al. 2004), semi-synthetic analogues piperidine alkaloids of Senna spectabilis (Viegas et al. 2005), triterpenoid alkaloids from Buxus hyrcana (Choudhary et al. 2006Choudhary MI, Shahnaz S, Parveen S, Khalid A, Mesaik MA, Ayatollahi SA and Atta-Ur-Rahman. 2006. New cholinesterase-inhibiting triterpenoid alkaloids from Buxushyrcana. Chem Biodivers 3: 1039-1052.), bisbenzylisoquinoline alkaloids from Cocculus pendulus(Atta-ur-Rahman et al. 2009) and alkaloid leptomerine from stems of Esenbeckia leiocarpa (Cardoso-Lopes et al. 2010Cardoso-Lopes EM, Maier JA, Da Silva MR, Regasini LO, Simote SY, Lopes NP, Pirani JR, Bolzani VS and Young MC. 2010. Alkaloids from stems of Esenbeckialeiocarpa Engl. (Rutaceae) as potential treatment for Alzheimer disease. Molecules 15: 9205-9213.).
The results of the present investigation proved that sesquiterpene lactone gaillardin inhibited the enzyme by 67% in concentration of 300 µg/mL which showed reasonable inhibition (figure 2). Pulchellin C and britannin showed 10.9% and 25.2% inhibition, respectively (figure 3).
The AChEI property of gaillardin draws attention to these versatile secondary metabolites and the plants containing sesquiterpene lactones, namely the Asteraceae species. The drawback to sesquiterpene lactones might be because of the reports of cytotoxic activity about most of them. Cytotoxicity of britannin, gaillardin and pulchellin C have also been reported before (Mosaddegh et al. 2010Mosaddegh M, Hamzeloo-Moghadam M, Ghafari S, Naghibi F, Ostad SN and Read RW. 2010. Sesquiterpene lactones from Inula oculus-christi. Nat Prod Commun 5: 511-514., Hamzeloo-Moghadam et al. 2012Hamzeloo-Moghadam M, Hajimehdipoor H, Saeidnia S, Atoofi A, Shahrestani Read RW and Mosaddegh M. 2012. Anti-proliferative activity and apoptotic potential of britannin, a sesquiterpene lactone from Inulaaucheriana, Nat Prod Commun 7: 979-980.). Their chemical structures make somewhat great differences in their cytotoxic effects which indicate britannin as the most and pulchellin C as the least effective one. Looking back at the results of AChEI activity of the three, demonstrates that gaillardin has presented much more activity than the other two sesquiterpene lactones and pulchellin C again is the least effective. Pulchellin C is an eudesmanolide sesquiterpene lactone, while the other two are both guaianolides. Regarding polarity, pulchellin C is the most polar with the least AChEI activity. It could be concluded that polarity might play a role in AChEI activity. Gaillardin which is the least polar compound, has the most AChEI activity (67%) and britannin and pulchellin C are in the next rates, respectively. In addition to polarity, chemical structure could make a lot of difference in binding to AChE. It is clear that all the three compounds belong to sesquiterpene lactones and they all bear the α-methylene-γ-lactone functional group but they do not simulate the same AChE inhibitory nor cytotoxic activity, suggesting a groundwork where medicinal chemistry rules. Hence derivatization might be a choice to be considered whenever cytotoxic activity is the hindrance, and it could even increase the biological activity, giving a chance to reach to more effective, less cytotoxic derivatives by manipulating the structures. The clinical efficacy and safety of gaillardin and other sesquiterpene lactones are still to be taken into account. There is limited or even no clinical data about these compounds, thus further assessments are crucial.
The authors express their appreciation to TMRC botanists Mrs. Pirani and Mr. Moazzeni. They also wish to thank the Traditional Medicine and Materia Medica Research Center, Shahid Beheshti University of Medical Sciences, for the financial support.
REFERENCES
- Aisen PS, Cummings J and Schneider LS. 2012. Pharmacologic Treatment for Alzheimer Disease. Cold Spring Harb Perspect Med 2:a006395. doi: 10.1101cshperspect. a006935
- Alzheimer's Association. 2012. Alzheimer's disease facts and figures. Alzheimer's & Dementia 8(2): 7-10.
- Arruda M, Viana H, Rainha N, Neng NR, Rosa JS, Nogueira JM and Barreto M. 2012. Anti-acetylcholinesterase and antioxidant activity of essential oils from Hedychium gardnerianum Sheppard ex Ker-Gawl. Molecules 17: 3082-3092
- Atta-Ur-Rahman, Atia-Tul-Wahab, Zia Sultani S, Nawaz SA and Choudhary MI. 2009. Bisbenzylisoquinoline alkaloids from Cocculuspendulus. Nat Prod Res 23: 1265-1273.
- Atta-Ur-Rahman, Feroz F, Naeem I, Zaheer-Ul-Haq, Nawaz SA, Khan N, Khan MR and Choudhary MI. 2004. New pregnane-type steroidal alkaloids from Sarcoccasaligna and their cholinesterase inhibitory activity. Steroids 69: 735-741.
- Cardoso-Lopes EM, Maier JA, Da Silva MR, Regasini LO, Simote SY, Lopes NP, Pirani JR, Bolzani VS and Young MC. 2010. Alkaloids from stems of Esenbeckialeiocarpa Engl. (Rutaceae) as potential treatment for Alzheimer disease. Molecules 15: 9205-9213.
- Choudhary MI, Shahnaz S, Parveen S, Khalid A, Mesaik MA, Ayatollahi SA and Atta-Ur-Rahman. 2006. New cholinesterase-inhibiting triterpenoid alkaloids from Buxushyrcana. Chem Biodivers 3: 1039-1052.
- D'Addario et al. 2012. Epigenetic Regulation of Fatty Acid Amide Hydrolase in Alzheimer Disease. PLoS ONE 7(6): 1-7.
- Ellman GL, Courtney KD, Andres Jr V and Featherstone RM. 1961. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7: 88-95.
- Farlow MR and Lilly ML. 2005. Rivastigmine: an open-label, observational study of safety and effectiveness in treating patients with Alzheimer's disease for up to 5 years. BMC Geriatrics 5. doi:10.1186/1471-2318-5-3
» https://doi.org/10.1186/1471-2318-5-3 - Feitosa CM, Freitas RM, Luz NNN, Bezerra MZB and Trevisan MTS. 2011. Acetylcholinesterase inhibition by somes promising Brazilian medicinal plants. Braz J Biol 71: 783-789.
- Frydman-Marom A, Levin A, Farfara D, Benromano T and Scherzer-Attali R. 2011. Orally Administrated Cinnamon Extract Reduces b-Amyloid Oligomerization and Corrects Cognitive Impairment in Alzheimer's Disease Animal Models PLoS ONE 6: e16564. doi:10.1371/journal.pone.0016564
» https://doi.org/10.1371/journal.pone.0016564 - Hamzeloo-Moghadam M, Hajimehdipoor H, Saeidnia S, Atoofi A, Shahrestani Read RW and Mosaddegh M. 2012. Anti-proliferative activity and apoptotic potential of britannin, a sesquiterpene lactone from Inulaaucheriana, Nat Prod Commun 7: 979-980.
- Ibrahim M, Farooq T, Hussain N, Hussain A, Gulzar T, Hussain I, Akash MS and Rehmani FS. 2013. Acetyl and butyryl cholinesterase inhibitory sesquiterpene lactones from Amberboaramosa. Chem Cent J 7: 116.
- Kemp PM, Holmes C, Hoffmann S, Wilkinson S, Zivanovic M, Thom J, Bolt L, Fleming J and Wilkinson DG. 2003. A randomised placebo controlled study to assess the effects of cholinergic treatment on muscarinic receptors in Alzheimer's disease. J Neurol Neurosurg Psychiatry 74: 1567-1570.
- Komersova A, Komersa K and Cegan A. 2007. New Findings about Ellman's Method to Determine Cholinesterase Activity Z. Naturforsch 62c: 150-154.
- Loizzo MR, Tundis R, Bonesi M, Menichini F, de Luca D, Colica C and Menichini F. 2012. Evaluation of Citrus aurantifolia peel and leaves extracts for their chemical composition, antioxidant and anti-cholinesterase activities. J Sci Food Agric 92: 2960-2967.
- Mosaddegh M, Hamzeloo-Moghadam M, Ghafari S, Naghibi F, Ostad SN and Read RW. 2010. Sesquiterpene lactones from Inula oculus-christi. Nat Prod Commun 5: 511-514.
- Mukherjee PK, Kumar V and Houghton PJ. 2007. Screening of Indian Medicinal Plants for Acetylcholinesterase Inhibitory Activity. Phytother Res 21: 1142-1145.
- Orhan G, Orhan I, Subutay-Öztekin N, Ak F and Sener B. 2009. Contemporary anticholinesterase pharmaceuticals of natural origin and their synthetic analogues for the treatment of alzheimer's disease. Recent Pat CNS Drug Discov 4: 43-51.
- Viegas Jr C et al. 2005. New selective acetylcholinesterase inhibitors designed from natural piperidine alkaloids. Bioorg Med Chem 13: 4184-4190.
Publication Dates
-
Publication in this collection
14 May 2014 -
Date of issue
June 2014
History
-
Received
7 Jan 2013 -
Accepted
14 Oct 2013