ABSTRACT
Agapanthus (Agapanthaceae) has 10 species described. However, most taxonomists differ respect to this number because the great phenotypic plasticity of the species. The cytogenetic has been an important tool to aid the plant taxon identification, and to date, all taxa of Agapanthus L'Héritier studied cytologically, presented 2n = 30. Although the species possess large chromosomes, the group is karyologically little explored. This work aimed to increase the cytogenetic knowledge of Agapanthus africanus (L.) Hoffmanns by utilization of chromosome banding techniques with DAPI / CMA3 and Fluorescent in situ Hybridization (FISH). In addition, flow cytometry was used for determination of DNA content and the percentage of AT / GC nitrogenous bases. Plants studied showed 2n = 30 chromosomes, ranging from 4.34 - 8.55 µm, with the karyotype formulae (KF) = 10m + 5sm. Through FISH, one 45S rDNA signal was observed proximally to centromere of the chromosome 7, while for 5S rDNA sites we observed one signal proximally to centromere of chromosome 9. The 2C DNA content estimated for the species was 2C = 24.4 with 59% of AT and 41% of GC. Our data allowed important upgrade for biology and cytotaxonomy of Agapanthus africanus (L.) Hoffmanns.
Key words:
African lily; chromosome banding; cytotaxonomy; flow cytometry; genome size; molecular cytogenetics
RESUMO
Agapanthus (Agapanthaceae) possui 10 espécies descritas. No entanto a maioria dos taxonomistas difere relativamente a este número, devido à grande plasticidade fenotípica das espécies. A citogenética tem sido uma ferramenta importante para auxiliar a identificação de taxa de plantas, e até o momento, todas as espécies de Agapanthus L'Héritier estudadas citologicamente, apresentaram 2n = 30. Embora as espécies possuam grandes cromossomos, o grupo é cariologicamente pouco explorado. Este trabalho teve como objetivo aumentar o conhecimento citogenético de Agapanthus africanus (L.) Hoffmanns pela utilização de técnicas de bandeamento cromossômico com DAPI / CMA3 e Hibridização in situ Fluorescente (FISH). Além disso, a citometria de fluxo foi utilizada para a determinação do conteúdo de DNA e do percentual de bases nitrogenadas AT e GC. As plantas estudadas apresentaram 2n = 30 cromossomos, variando de 4,34 - 8,55 µm, com a fórmula cariotípica (KF) = 10m + 5sm. Através da FISH, um sinal para rDNA 45S foi observada proximalmente ao centrômero do cromossomo 7, enquanto que para os sítios de rDNA 5S, observamos um sinal proximal ao centrômero do cromossomo 9. O conteúdo 2C de DNA estimado para a espécie foi de 2C = 24,4 com 59% de AT e 41% de GC. Nossos dados possibilitaram uma atualização importante para a biologia e citotaxonomia de Agapanthus africanus (L.) Hoffmanns.
Palavras-chave:
lírio Africano; bandeamento cromossômico; citotaxonomia; citometria de fluxo; tamanho do genoma; citogenética molecular
INTRODUCTION
The genus Agapanthus L'Héritier (Agapantahceae) is a monocotyledonous, herbaceus and perennial plant group endemic to southern Africa, with ten species in which three are considered evergreen and seven deciduous plants (Leighton 1965Leighton FM. 1965. The genus Agapanthus L'Héritier. J South African Bot IV(Suppl.): 1-50.). Nevertheless, Agapanthus L'Héritier taxonomy is considered very difficult once there are few unique characters and a high plasticity depending on circumstances of growing. This feature takes some authors to believe in the existence of only one species with innumerous varieties (McNeil 1972McNeil G. 1972. The Katberg Agapanthus. J Roy Hort Soc 97(12): 534-536.). So, the obtaining and exploring of new characters, less plastic to identification of these species are of great importance for Agapanthus L'Héritier taxonomy.
The use of karyological data in taxonomy, contribute widely to evaluate the genetic relationship among species or populations and to a better understanding of the way they diverged from each other (Guerra 2008Guerra M. 2008. Chromosome numbers in plant cytotaxonomy: concepts and implications. Cytogenet Genome Res 120: 339-350., Sousa et al. 2013Sousa SM, Reis AC and Viccini LF . 2013. Polyploidy, B chromosomes and heterochromatin characterization of Mimosa caesalpiniifolia Benth. (Fabaceae-Mimosoideae). Tree Genet Genomes 9: 613-619., Reis et al. 2014Reis AC, Sousa SM, Vale AA, Pierre PMO, Franco AL, Campos JMS, Vieira RF and Viccini LF. 2014. Lippia alba (Verbenaceae): A new tropical autopolyploid complex? Am J Bot 101: 1002-1012.). In Agapanthus L'Héritier, all plants measured until the moment have the same chromosome number, 2n = 30 (Riley and Mukerjee 1960Riley HP and Mukerjee D. 1960. Chromosomes in Agapanthus. Genetics 45: 1008., Sharma and Mukhopadhyay 1963Sharma AK and Mukhopadhyay S. 1963. Chromosome study in Agapanthus and the phylogeny of its species. Caryologia 16: 127-137.), with little differences only in chromosome length. Since chromosome length can vary with condensation, this parameter in plastic plants, studied without another chromosome marker is not a good tract for detection of speciation. For this, more detailed karyological studies are very important to increment the utilization of chromosome length like a parameter of species identification in Agapanthus L'Héritier.
Agapanthus africanus (L.) Hoffmanns, an endemic South African species, highly desired as ornamental, has been grown around the world, being adapted to conditions in Europe, Australia, New Zealand and North as well as South America, since the late seventeenth century (Win Snoeijer 2004Win Snoeijer FLS. 2004. Agapanthus: a revision of the genus. Timber Press, 2 Station Road. Swavesey: Cambridge, 320 p.). Besides its ornamental attributes the species are traditionally used by local communities in South Africa as medicinal for various disorders: during pregnancy to induce labour (Kaido et al. 1997Kaido TL, Veale DJH, Havlik I and Rama DBK. 1997. Preliminary screening of plants used in South Africa as traditional herbal remedies during pregnancy and labour. J Ethnopharmacol 55: 185-191.); to treat constipation in pregnancy (Van Wyk et al. 1997Van Wyk BE, Van Oudtshoorn B and Gericke N. 1997. Medicinal plants of South Africa, Briza Publications, South Africa, p. 32.); as antenatal or post-natal treatment of the mother and for high blood pressure (Duncan et al. 1999Duncan A, Jager AC and van Staden JK. 1999. Screening of Zulu medicinal plants for angiotensin-converting enzyme (ACE) inhibitors. J Ethnopharmacol 68: 63-70.). Additionally some studies have shown that Agapanthus africanus (L.) Hoffmanns has an important antifungal potential against plant pathogens (Singh et al. 2008Singh DN, Verma N, Raghuwanshi S, Shukla PK and Kulshreshtha DK. 2008. Antifungal activity of Agapanthus africanus extractives. Fitoterapia 79: 298-300., Tagegne et al. 2008Tagegne G, Pretorius JC and Swart WJ. 2008. Antifungal properties of Agapanthus africanus L. extracts against plant pathogens. Crop Protection 27: 1052-1060. ).
Due its varied application and importance around the world, the aim of the present work was to obtain new chromosomal markers for Agapanthus africanus (L.) Hoffmanns (an important informations for taxonomy and breeding of the species) utilizing molecular cytogenetics, chromosomes banding and flow cytometry which are scarce approaches for the genus.
MATERIALS AND METHODS
Plant Material and Preparation of Mitotic Spreads
Seeds of Agapanthus africanus (L.) Hoffmanns were collected at three different sites: Bom Pastor Neighborhood (21°46'31.6"S and 43°20'42.3"W), São Pedro Neighborhood (21°46›19.9"S and 43°22›39.1"W), and in the Cam pus of the Federal University of Juiz de Fora (21°46'23.3"S and 43°22'09.3"W), in Juiz de Fora Minas Gerais, Brazil. For each locality around 50 seeds were collected and herbarium voucher specimens were deposited at the CESJ Herbarium of the Federal University of Juiz de Fora (available at http://www.splink.org.br).
After that, seeds were germinated and roots tips were pre-treated with 8-hydroxyquinoline solution (0.003 M) at room temperature for 9h and then fixed in ethanol and acetic acid (3:1 v/v) for 24h at -20ºC. Root meristems were submitted to enzymatic maceration (2% Celullase: 20% Pectinase) for 5h at 37ºC. The slides were prepared according to Carvalho and Saraiva 1997Carvalho CR and Saraiva LS. 1997. High-resolution HKG-banding in maize mitotic chromosomes. J Plant Res 110: 417-420. (1993Carvalho CR and Saraiva LS. 1993. A new heterochromatin banding pattern revealed by modified HKG banding technique for maize chromosomes. Heredity 70: 515-519. , ).
Chromosome Morphometry
Chromosome length, short and long arms length and ratio between chromosome arms were measured and chromosome classification was done according to Levan et al. (1964Levan A, Fredga A and Sanderber A. 1964. Nomenclature for centromeric position in chromosomes. Hereditas 52: 201-220. ). The ideogram was mounted according to decreasing size order of chromosomes short arms.
Chromosome Banding
Chromosome banding was performed according Schweizer (1976Schweizer D. 1976. Reverse fluorescent chromosome banding with chromomycin and DAPI. Chromosoma 58: 307-324.). Five aged slides from each localities were stained with chromomycin A3 (0.5 mg/mL) for 1h, dystamicyn (0.1 mg/mL) for 30 min and 2-4 diamidino-2-phenylindole (2 µg/mL) for 30 min. The slides were mounted in Mcllvaine's pH 7.0 buffer-glycerol (1:1 v/v). For this analysis ten metaphases from plants of each locality were observed and captured in an Olympus DP72 digital camera. The chromosomes were observed using an epifluorescence microscope (Olympus BX 51) with appropriate filter set.
After images documentation the slides were discolored in 100% ethanol for 24h at room temperature and used for molecular cytogenetic processes.
Molecular Cytogenetic
Fluorescence in situ hybridization (FISH) was performed using 45S rDNA probes from Triticum aestivum L. (Gerlach and Bedbrook 1979Gerlach WL and Bedbrook JR. 1979. Cloning and characterization of ribosomal RNA genes from wheat and barley. Nucleic Acids Res 7: 1869-1885.) and 5S probes from Zea mays L. (courtesy Koo and J. Jiang). Each probe was labeled with digoxigenin by nick translation and then hybridized according to Jiang et al. (1995)Jiang J, Gill BS, Wang GL, Ronald PC and Ward DC. 1995. Metaphase and interphase fluorescence in situ hybridization mapping of the rice with bacterial artificial chromosomes. Proc Natl Acad Sci USA 92: 4487- 4491. with minor modifications. The hybridization mixture was denatured at 85°C for 10 min and immediately transferred to an icebox. The slides were denatured at 85°C for 1 min and treated with a series of ethanol washes (70%, 90%, and 100% for 5 min each). The hybridization mixture was then added to the slides and the chromosomes allowed to hybridize at 37°C for 48h. Posthybridization washes were carried out using 2 × SSC buffer for 15 min and 1 × PBS buffer for 5 min. Probes were detected with anti-DIG conjugate with rhodamine and postdetection washes were performed using 1 × TNT buffer and for 15 min and 1 × PBS buffer for 5 min at room temperature. Chromosomes were counter-stained with 2 µg/mL of DAPI. The slides were mounted in Vectashield (Vector) and samples were rehybridized after slides discoloration in 100% ethanol for 24h. Images with 45S and 5S signals were merged using CellSens software (Olympus). Chromosomes were observed using an epifluorescence microscope (Olympus BX 51) with appropriate filter set.
Flow Cytometry (FCM)
Nuclear DNA content was determined according to the method of Galbraith et al. (1983Galbraith DW, Harkins KR, Maddox JM, Ayres NM, Sharma DP and Firoozabady E. 1983. Rapid flow cytometric analysis of the cell cycle in intact plant tissues. Science 220: 1049-1051.). Approximately 20-30 mg of young and fresh leaves of 3 plants of Agapanthus africanus (L.) Hoffmanns from each locality and the same amount of young foliar tissue of standard references (Pisum sativum L.) were chopped on ice with 1 mL of OTTO I lysis buffer solution (0.1 M C6H8O7; 0.5% Tween 20) (Otto 1990Otto FJ. 1990. DAPI staining of fixed cells for high-resolution flow cytometry of nuclear DNA. Methods Cell Biol 33: 105-110.) supplemented with 50 µg/mL-1 RNAse. The suspension was filtered through 40nm mesh into 2 mL microcentrifuge tube and centrifuged at 1,100 rpm for 5 minutes. The pellet was incubated in 100 µL OTTO I lysis buffer for 10 minutes and then was added 300 µL of OTTO I: OTTO II (0.4 M Na2HPO4.12H2O) (1:2, v/v) buffers. The sample were homogenized and stained with 50 µg/mL-1 of propidium iodide (PI) to determine the total DNA content.
AT/GC composition was determined by adding DAPI (4,6-diamidino-2-phenylindole) 4 µg/mL-1 to the samples. At least 10,000 nuclei were analyzed per sample in a FACSCantoII (BectonDickinson) flow cytometer. The histograms were analyzed using Flowing 2.5.1 software (www.flowingsoftware.com).
DNA nuclear content (pg) of each sample was estimated by the relative fluorescence intensity of the sample and the internal reference standard (Pisum sativum L. 9.09 pg). Three samples, of each accession, were measured according to the following equation (Dolezel 2003Dolezel J, Bartos J, Voglmayr H and Greilhuber J. 2003. Nuclear DNA content and genome size of trout and human. Cytometry 51: 127-128.):

(PIFI is the fluorescence intensity of propidium iodide in G1 cells).
The AT percentage of Agapanthus africanus (L.) Hoffmanns was measured in relation to P. sativum L. reference standard, following the formula described by Godelle et al. (1993Godelle B, Cartier D, Marie D, Brown CS and Siljak-Yakovlev S. 1993. Heterochromatin study demonstrating the non-linearity of fluorometry useful for calculatinggenomicbase composition. Cytometry 14: 618-626.):

(R is the ratio of fluorescence intensity between the peak of Agapanthus africanus (L.) Hoffmanns and Pisum sativum L., and r (binding length) = 3 for DAPI dye).
The percentage of the complementary bases was calculated as GC% = 100 - AT%.
Taking into account that 1 pg = 978 Mpb (Dolezel et al. 2003Dolezel J, Bartos J, Voglmayr H and Greilhuber J. 2003. Nuclear DNA content and genome size of trout and human. Cytometry 51: 127-128.), the combination of cytometric and cytogenetic data were provided.
RESULTS AND DISCUSSION
Samples of Agapanthus africanus (L.) Hoffmanns from the three different localities studied here showed symmetrical karyotype with 2n = 30, of which ten chromosomes with centromeres at the median region (m, Arm Ratio = 1 - 1.7), and five of them at submedian (sm, Arm Ratio = 1.71 - 3.0) (Table I). No secondary constrictions were observed and absolute chromosome length ranged from 4.34 - 8.55 µm (Table I) while the relative chromosome length revealed that the largest chromosome represented around 9.46% of the genome size and the shortest one 4.80% (Table I). Our results are in conformity with the cytogenetics morphometric data in the literature for Agapanthus L'Héritier (Riley and Mukerjee 1962Riley HP and Mukerjee D. 1962. Chromosomes of some species of Agapanthus. Cytologia 27: 325-332., Sharma and Mukhopadhyay 1963Sharma AK and Mukhopadhyay S. 1963. Chromosome study in Agapanthus and the phylogeny of its species. Caryologia 16: 127-137., Yaacob and Taha 2014Yaacob JS and Taha RM. 2014. Genetic stability of in vitro propagated African blue lily (Agapanthus praecox ssp. minimus). Caryologia 67: 227-233.), however, no cytogenetics data for the taxa Agapanthus africanus (L.) Hoffmanns has been described. In the most recent revision of the genus (Win Snoeijer 2004Win Snoeijer FLS. 2004. Agapanthus: a revision of the genus. Timber Press, 2 Station Road. Swavesey: Cambridge, 320 p.) Agapanthus africanus (L.) Hoffmanns is considered like synonymy of Agapanthus minor Lodd., Agapanthus umbellatus L'Héritier, Agapanthus umbelliferus Poir., Agapanthus tuberosus L. ex DC. and Agapanthus variegatus Steud.Sharma and Mukhopadhyay (1963)Sharma AK and Mukhopadhyay S. 1963. Chromosome study in Agapanthus and the phylogeny of its species. Caryologia 16: 127-137. studing the cytogenetics of varieties of Agapanthus umbellatus L'Hér. observed a very similar chromosomal morphology with the data observed for us, principally when we compare it with the taxa Agapanthus umbellatus var. intermedius Tubergen where the size of chromosomes varied from 4.1-8.9 µm, with all chromosomes with centromeres at median and sub-median regions. Nevertheless, theses authors observed one pair of chromosomes with secondary constrictions, no observed in our work with DAPI coloration.
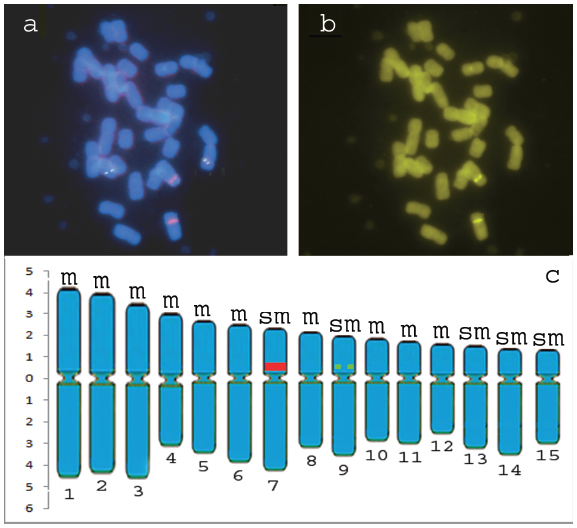
Although chromosomes in Agapanthus L'Héritier are very large and of easy obtainment, the cytogenetics of the group is very little explored. Our observations are the first data related to molecular cytogenetics and with the mapping of heterochromatin for the genus. The 45S rDNA signal was observed in one chromosome pair on the interstitial portion, proximally to the centromere (in the short arm of chromosome 7) (Fig. 1a, c), while 5S rDNA signals was observed in interstitial portion, proximally to the centromere, in the short arm of chromosome 9 (Fig. 1a, c). The 45S rDNA sites showed greater bands when compared with those ones observed for 5S rDNA sites, which showed pairs of little dots (Fig. 1a, c). This experiment was very important to determine the number of rDNA sites in this species once no secondary constrictions were observed in the slides prepared here. Once again our data are in conformity with observations of Sharma and Mukhopadhyay (1963Sharma AK and Mukhopadhyay S. 1963. Chromosome study in Agapanthus and the phylogeny of its species. Caryologia 16: 127-137.) for the Agapanthus umbellatus var. intermedius Tubergen since these authors observed one pair of secondary constrictions (these data indicate that at least one pair of 45S rDNA is present in this species once that in major plants this sites are coincident). Nevertheless, like us, Panzera et al. (1996Panzera F, Giménez-Abián MI, López-Sáez JF, Giménez-Martin G, Cuadrado A, Shaw PJ, Beven AF, Cánovas JL and De La Torre C. 1996. Nucleolar organizer expression in Allium cepa L. chromosomes. Chromosoma 105: 12-19. ) also observed in Allium cepa L. sites of active 45S rDNA no coincident with secondary constrictions, confirming that these sites can or can't form secondary constrictions.
Representative metaphases with 5S rDNA and 45S rDNA mapping (a); CMA3 banding pattern (b) and Ideogram of Agapanthus africanus (L.) Hoffmanns (c) (green squares= 5S rDNA, red rectangle = 45S rDNA), m = metacentric and sm = submetacentric.
For the chromosome banding, no centromeric, interstitial or terminal DAPI bands were observed. However, fluorochrome staining with CMA3 revealed bands on chromosome pair 7, which were also DAPI negative (Fig. 1b). The observed bands in homologues chromosomes showed similar size and bright being coincident with 45S rDNA (Fig. 1a, b), a common feature in a wide variety of plants (Sumner 1990Sumner AT. 1990. Chromosome banding. Unwin Hyman: Boston, 434 p., Guerra and Felix 2000Guerra M and Felix LP. 2000. O cariótipo de Nothoscordum pulchellum (Alliaceae) com ênfase na heterocromatina e sítios de DNAr. Bol Soc Argent Bot 35: 283-289., Sousa et al. 2013Sousa SM, Reis AC and Viccini LF . 2013. Polyploidy, B chromosomes and heterochromatin characterization of Mimosa caesalpiniifolia Benth. (Fabaceae-Mimosoideae). Tree Genet Genomes 9: 613-619.) . No additional CMA bands were observed in any of the chromosome pairs (Fig. 1b).
Regarding to the nuclear genome size estimation, the FCM provided high quality histograms with G0/G1 peaks showing CV = 2.47. The 2C DNA content estimated for the species was 2C = 24.4 pg. Taking into account that 1pg = 978 Mpb (Dolezel et al. 2003Dolezel J, Bartos J, Voglmayr H and Greilhuber J. 2003. Nuclear DNA content and genome size of trout and human. Cytometry 51: 127-128.), and combining cytometric and cytogenetic data, the largest chromosome (chromosome 1) corresponds to 1128.72 Mpb (~9.5% of the genome), while the smallest one (chromosome 15) corresponds to 572.71 Mpb (~4.8% of the genome) (Table I). Genome size, obtained by flow cytometry has been successfully used to investigate Agapanthus L'Héritier (Zonneveld 2004Zonneveld BJM. 2004. Genome size in species and cultivars of Agapanthus L'Hér. (Agapanthaceae). In: Win Snoeijer FLS (Ed), Agapanthus: a revision of the genus. Timber Press, 2 Station Road. Swavesey: Cambridge, p. 20-36.). When all species in a genus have the same chromosome number, as in Agapanthus L'Héritier, differences in nuclear DNA content have proven to be very effective in delimiting of infrageneric divisions in a number of taxa (Ohri 1998Ohri D. 1998. Genome size variation and plant systematics. Ann Bot 82(Suppl. A): 75-83., Zonneveld 2004Zonneveld BJM. 2004. Genome size in species and cultivars of Agapanthus L'Hér. (Agapanthaceae). In: Win Snoeijer FLS (Ed), Agapanthus: a revision of the genus. Timber Press, 2 Station Road. Swavesey: Cambridge, p. 20-36.). With this approaches, Zonneveld and Duncan (2003)Zonneveld BJM and Duncan GD. 2003. Taxonomic implications of genome size and pollen colour and vitality for species of Agapanthus L'Héritier (Agapanthaceae). Plant Syst Evol 241: 115-123. suggest that there are six species in Agapanthus L'Héritier and not ten as described by Leigthon (1965). The species in this context would be: Agapanthus campanulatus F. M. Leight. (2C = 22.3 pg), Agapanthus caulescens Spreng. (2C = 23.2 pg), Agapanthus coddii F.M.Leight. (2C = 24.0 pg), Agapanthus praecox Willd. (2C = 25.5 pg), Agapanthus inapertusBeauverd (2C = 25.1 pg), and Agapanthus africanus (L.) Hoffmanns (2C = 31.6 pg), while the other four, according to the new proposal, would receive a status below species rank. When compared with the value observed for Zonneveld and Duncan (2003)Zonneveld BJM and Duncan GD. 2003. Taxonomic implications of genome size and pollen colour and vitality for species of Agapanthus L'Héritier (Agapanthaceae). Plant Syst Evol 241: 115-123., the DNA content obtained by us (2C = 24.4 pg) is very next of Agapanthus coddii F. M. Leight. (2C = 24.0 pg) while the content obtained for Agapanthus africanus (L.) Hoffmanns for these authors was different of our (2C = 31.6 pg). Once more, this observation shows the great intraspecific variation observed in the genus.
In addition to the DNA content, PI and DAPI fluorochromes index allowed, for the first time, the estimation of base composition of the genome of Agapanthus africanus (L.) Hoffmanns. The percentage of base was 59% for AT and 41% for GC. The representative histograms of DNA content and base composition can be seen in figure 2.
Flow cytometry histograms representing Agapanthus africanus (L.) Hoffmanns using Pisum sativum L. (9.09 pg) as internal standard. (a) The fluorochrome propidium iodide (PI) was used to measurement of DNA amount (24.4 pg; CV=2.47) (2C values). (b) The fluorochrome 4′,6-diamidino-2-phenylindole (DAPI) was used to determine the AT base percentual (59%; CV = 1.64).
Finally, our data constitute an important upgrade in the biology of the Agapanthus africanus (L.) Hoffmanns, and can be used to future taxonomic, phylogenetic studies and for the chromosome evolution of the genus.
ACKNOWLEDGMENTS
The authors wish to thank Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for financial support.
REFERENCES
- Carvalho CR and Saraiva LS. 1993. A new heterochromatin banding pattern revealed by modified HKG banding technique for maize chromosomes. Heredity 70: 515-519.
- Carvalho CR and Saraiva LS. 1997. High-resolution HKG-banding in maize mitotic chromosomes. J Plant Res 110: 417-420.
- Dolezel J, Bartos J, Voglmayr H and Greilhuber J. 2003. Nuclear DNA content and genome size of trout and human. Cytometry 51: 127-128.
- Duncan A, Jager AC and van Staden JK. 1999. Screening of Zulu medicinal plants for angiotensin-converting enzyme (ACE) inhibitors. J Ethnopharmacol 68: 63-70.
- Galbraith DW, Harkins KR, Maddox JM, Ayres NM, Sharma DP and Firoozabady E. 1983. Rapid flow cytometric analysis of the cell cycle in intact plant tissues. Science 220: 1049-1051.
- Gerlach WL and Bedbrook JR. 1979. Cloning and characterization of ribosomal RNA genes from wheat and barley. Nucleic Acids Res 7: 1869-1885.
- Godelle B, Cartier D, Marie D, Brown CS and Siljak-Yakovlev S. 1993. Heterochromatin study demonstrating the non-linearity of fluorometry useful for calculatinggenomicbase composition. Cytometry 14: 618-626.
- Guerra M. 2008. Chromosome numbers in plant cytotaxonomy: concepts and implications. Cytogenet Genome Res 120: 339-350.
- Guerra M and Felix LP. 2000. O cariótipo de Nothoscordum pulchellum (Alliaceae) com ênfase na heterocromatina e sítios de DNAr. Bol Soc Argent Bot 35: 283-289.
- Jiang J, Gill BS, Wang GL, Ronald PC and Ward DC. 1995. Metaphase and interphase fluorescence in situ hybridization mapping of the rice with bacterial artificial chromosomes. Proc Natl Acad Sci USA 92: 4487- 4491.
- Kaido TL, Veale DJH, Havlik I and Rama DBK. 1997. Preliminary screening of plants used in South Africa as traditional herbal remedies during pregnancy and labour. J Ethnopharmacol 55: 185-191.
- Leighton FM. 1965. The genus Agapanthus L'Héritier. J South African Bot IV(Suppl.): 1-50.
- Levan A, Fredga A and Sanderber A. 1964. Nomenclature for centromeric position in chromosomes. Hereditas 52: 201-220.
- McNeil G. 1972. The Katberg Agapanthus J Roy Hort Soc 97(12): 534-536.
- Ohri D. 1998. Genome size variation and plant systematics. Ann Bot 82(Suppl. A): 75-83.
- Otto FJ. 1990. DAPI staining of fixed cells for high-resolution flow cytometry of nuclear DNA. Methods Cell Biol 33: 105-110.
- Panzera F, Giménez-Abián MI, López-Sáez JF, Giménez-Martin G, Cuadrado A, Shaw PJ, Beven AF, Cánovas JL and De La Torre C. 1996. Nucleolar organizer expression in Allium cepa L. chromosomes. Chromosoma 105: 12-19.
- Reis AC, Sousa SM, Vale AA, Pierre PMO, Franco AL, Campos JMS, Vieira RF and Viccini LF. 2014. Lippia alba (Verbenaceae): A new tropical autopolyploid complex? Am J Bot 101: 1002-1012.
- Riley HP and Mukerjee D. 1960. Chromosomes in Agapanthus Genetics 45: 1008.
- Riley HP and Mukerjee D. 1962. Chromosomes of some species of Agapanthus Cytologia 27: 325-332.
- Schweizer D. 1976. Reverse fluorescent chromosome banding with chromomycin and DAPI. Chromosoma 58: 307-324.
- Sharma AK and Mukhopadhyay S. 1963. Chromosome study in Agapanthus and the phylogeny of its species. Caryologia 16: 127-137.
- Singh DN, Verma N, Raghuwanshi S, Shukla PK and Kulshreshtha DK. 2008. Antifungal activity of Agapanthus africanus extractives. Fitoterapia 79: 298-300.
- Sousa SM, Reis AC and Viccini LF . 2013. Polyploidy, B chromosomes and heterochromatin characterization of Mimosa caesalpiniifolia Benth. (Fabaceae-Mimosoideae). Tree Genet Genomes 9: 613-619.
- Sumner AT. 1990. Chromosome banding. Unwin Hyman: Boston, 434 p.
- Tagegne G, Pretorius JC and Swart WJ. 2008. Antifungal properties of Agapanthus africanus L. extracts against plant pathogens. Crop Protection 27: 1052-1060.
- Van Wyk BE, Van Oudtshoorn B and Gericke N. 1997. Medicinal plants of South Africa, Briza Publications, South Africa, p. 32.
- Win Snoeijer FLS. 2004. Agapanthus: a revision of the genus. Timber Press, 2 Station Road. Swavesey: Cambridge, 320 p.
- Yaacob JS and Taha RM. 2014. Genetic stability of in vitro propagated African blue lily (Agapanthus praecox ssp. minimus). Caryologia 67: 227-233.
- Zonneveld BJM. 2004. Genome size in species and cultivars of Agapanthus L'Hér. (Agapanthaceae). In: Win Snoeijer FLS (Ed), Agapanthus: a revision of the genus. Timber Press, 2 Station Road. Swavesey: Cambridge, p. 20-36.
- Zonneveld BJM and Duncan GD. 2003. Taxonomic implications of genome size and pollen colour and vitality for species of Agapanthus L'Héritier (Agapanthaceae). Plant Syst Evol 241: 115-123.
Publication Dates
-
Publication in this collection
18 Aug 2016 -
Date of issue
2016
History
-
Received
03 Aug 2015 -
Accepted
27 Nov 2015