ABSTRACT
Brachiaria brizantha is a forage grass well adapted to tropical areas and cultivated in millions of hectares in Brazil. The apomictic mode of reproduction in this species, in addition to differences in ploidy between sexual and apomictic plants, impairs crossbreeding. The development of a methodology to transform apomictic cultivars will provide an option to introduce agronomic important traits to B. brizantha cv. Marandu. In addition, it will open the possibility to study in vivo the function of candidate genes involved in the apomictic reproduction. The objective of this work was to evaluate peeled seeds, isolated embryo from mature seeds, embryogenic calluses and embryogenic cell suspensions, as target explant for genetic transformation via biolistics. Plasmids bearing the marker genes gus and hptII under the control of the rice actin 1 promoter (pAct1-Os) or the maize ubiquitin 1 promoter (pUbi1Zm) were used. All the target-explants used were suitable for transient gene expression after bombardment, showing gus expression and resistance to hygromycin. Using embryogenic calluses and cell suspensions as target tissues, transgenic plants were regenerated and transgenes detected.
Key words:
forage; gus; hptII; hygromycin
INTRODUCTION
Brachiaria brizantha (syn. Urochloa brizantha;Shirasuna 2015SHIRASUNA RT. 2015. Urochloa. In: Jardim Botânico do Rio de Janeiro. Lista de espécies da flora do Brasil, Rio de Janeiro. Dísponível em: <http://floradobrasil.jbrj.gov.br/jabot/floradobrasil/FB26028>. Acessado em 4 de Janeiro de 2016.
http://floradobrasil.jbrj.gov.br/jabot/f...
) cv. Marandu is a forage grass well adapted to tropical areas. Only in Brazil Brachiaria is cultivated in millions of hectares. This cultivar is tetraploid and reproduces asexually by apomixis. Analysis of the reproductive mode, based on the embryo sac structure of 275 B. brizantha accessions from the Brazilian germplasm collection, displayed one single sexual diploid, the remaining being apomicts and polyploids (Valle and Savidan 1996VALLE CB AND SAVIDAN Y. 1996. Genetics, cytogenetics, and reproductive biology of Brachiaria. In: Miles JW, Maass BL and Valle CB (Eds), Brachiaria: biology, agronomy, and improvement, Cali-Colômbia: CIAT, p. 147-163.). The difference in ploidy level between sexual and apomictic plants, in addition to the apomictic mode of reproduction, impairs breeding, reducing the chances of incorporating new traits to the cultivar. Interspecific crosses have been used to allow breeding, and more recently intraspecific crosses using an artificial tetraploid female progenitor (Monteiro et al. 2016MONTEIRO LC, VERZIGNASSI JR, BARRIOS SCL, VALLE CB, FERNANDES CD, BENTEO GL AND LIBÓRIO CB. 2016. Brachiaria decumbens intraspecific hybrids: characterization and selection for seed production. J Seed Sci 38: 62-67.). Considering all the bias resulting from these crosses, genetic transformation is an option to introduce agronomic important traits to the apomictic cultivated B. brizantha cv. Marandu. Another important aspect of Brachiaria transformation is the possibility to study the function of candidate genes involved in the apomictic reproduction (Silveira et al. 2012SILVEIRA ED, GUIMARÃES LA, DUSI DMA, SILVA FR, MARTINS NF, CARMO COSTA MM, ALVES-FERREIRA M AND CARNEIRO VTC. 2012. Expressed sequence-tag analysis of ovaries of Brachiaria brizantha reveals genes associated with the early steps of embryo sac differentiation of apomictic plants. Plant Cell Report 31: 403-416., Guimarães et al. 2013GUIMARÃES LA, DUSI DMA, MASIERO S, RESENTINI F, GOMES ACMM, SILVEIRA ED, FLORENTINO LH, RODRIGUES JCM, COLOMBO L AND CARNEIRO VTC. 2013. BbrizAGL6 is differentially expressed during embryo sac formation of apomictic and sexual Brachiaria brizantha plants. Plant Mol Biol Report 31: 1397-1406.). Moreover, establishing methodology to express gene transiently may assist the investigation of promoters and genes using silencing and gain-of-function approaches in this species.
Transformation via biolistic resulted in the production of transgenic plants in many recalcitrant species of monocotyledonous that are not susceptible to Agrobacterium (Sharma et al. 2005SHARMA KK, BHATNAGAR-MATHUR P AND THORPE TA. 2005. Genetic transformation technology: status and problems. In Vitro Cell Dev Biol - Plant 41: 102-112., Sood et al. 2011SOOD P, BHATTACHARYA A AND SOOD A. 2011. Problems and possibilities of monocot transformation. Biol Plant 55: 1-15.). Embryogenic cultures or immature embryos are the preferred target explants to transform cereals and forage grasses (Harwood 2012HARWOOD WA. 2012. Advances and remaining challenges in the transformation of barley and wheat. J Exp Bot 63: 1791-1798.). Actually, embryogenic calluses of the sexual diploid B. ruziziensis were transformed by biolistic with a vector containing a phosphinothricin resistance marker gene (bar) and the reporter gene gus, both under the control of the maize ubiquitin 1 promoter, pUbi1Zm (Ishigaki et al. 2012ISHIGAKI G, GONDO T, SUENAGA K AND AKASHI R. 2012. Fertile transgenic Brachiaria ruziziensis (ruzigrass) plants by particle bombardment of tetraploidized callus. J Plant Physiol 169: 546-549.). Two transgenic plants were recovered, they had normal phenotype, however, they did not produce seeds. One fertile transgenic plant was obtained only from an artificially tetraploidized callus (Ishigaki et al. 2012).
In vitro plant regeneration protocols using as explants peeled seeds, isolated embryos from mature seeds, embryogenic calluses and embryogenic cell suspensions of B. brizantha cv. Marandu were developed (Cabral et al. 2011CABRAL GB, CARNEIRO VTC, LACERDA AL, DO VALLE CB, MARTINELLI AP AND DUSI DMA. 2011. Somatic embryogenesis and organogenesis in apomictic and sexual Brachiaria brizantha. Plant Cell Tissue Organ Cult 107: 271-282., 2015). Despite the progress in the development of tissue culture protocols, B. brizantha genetic transformation is still a challenge. The aim of this work was to evaluate the viability of these somatic embryogenesis systems for transient and stable expression of transgenes via biolistics using plasmids bearing the marker genes gus and hptII.
MATERIALS AND METHODS
Seeds of a natural tetraploid apomictic genotype of Brachiaria brizantha cv. Marandu (Embrapa’s collection number BRA 000591) were used for all experiments. Four types of explants were evaluated for genetic transformation via biolistics: (i) embryo isolated from mature seed; (ii) mature dehusked seed; (iii) embryogenic callus; (iv) cell suspension.
The following plasmids were used for bombardment: a) pAct1-D, derived from pUC (McElroy et al. 1990) containing the gus gene under the rice actin 1 promoter pAct1-Os; b) pAHC27 (Christensen and Quail 1996CHRISTENSEN AH AND QUAIL PH. 1996. Ubiquitin promoter-based vectors for high-level expression of selectable and/or screenable marker genes in monocotyledonous plants. Transgenic Res 5: 213-218.) containing the gus gene under the maize ubiquitin 1 promoter (pUbi1Zm); pAHUG, derived from pUC19, containing the hygromycin phosphotransferase II (hptII) gene under pAct1Os promoter, and the gus gene under the pUbiZm promoter; two binary vectors pGPro1, and pGPro2 (Thilmony et al. 2006THILMONY R, GUTTMAN M, CHINIQUY D AND BLECHL A. 2006. pGPro1, a novel binary vector for monocot promoter characterization. Plant Mol Biol Report 24: 57-69., 2009), containing the marker hptII gene under the control of pAct1Os promoter (Thilmony et al. 2006). These vectors have a multiple restriction site to clone promoters, therefore, the promoter pUbi1Zm, isolated from plasmid pAHC27 (Christensen and Quail 1996), was inserted into PstI restriction site to drive the expression of the gus gene.
For bombardment, explants were positioned in a 1-cm-circle defined in the center of the 6 cm diameter Petri dish containing M1.3 medium with 0.7 % Phytagel (bombardment medium). Plasmidial DNA (8 µg) was precipitated with tungsten microparticles (M10). The physical parameters were: helium pressure of 900 psi (unless otherwise specified), target distance of 6 cm and 27 lb Hg vacuum, one or two shots per plate.
Tissue culture media M1.2, M1.3, MS3 and PMM were described in Cabral et al. (2011CABRAL GB, CARNEIRO VTC, LACERDA AL, DO VALLE CB, MARTINELLI AP AND DUSI DMA. 2011. Somatic embryogenesis and organogenesis in apomictic and sexual Brachiaria brizantha. Plant Cell Tissue Organ Cult 107: 271-282.), NBBAP, NBreg, DD1 were described in Cabral et al. (2015) (Table I). Culture room temperature was kept at 25 ± 2°C, transformation controls (bombardment without plasmid) and regeneration controls (culture without selection pressure) were performed for all experiments.
For PCR and DNA gel blot analysis of PCR, DNA was extracted from calluses, cell suspension and young leaves from hygromycin-resistant plants. Explants were macerated in a 1.5 mL buffer Tris HCl pH 8 0.1 M, NaCl 0.25 M, EDTA 25 mM and SDS 0.5 %, at room temperature. The samples were centrifuged for 5 min at 12,000 rpm. Supernatant was transferred to a new tube, and one volume of isopropanol was added, and incubated for 5 min at room temperature. The samples were then centrifuged for 5 min at 12,000 rpm, the supernatant discarded and the sediment resuspended in 20 μL of sterilized ultrapure water. PCR was performed using primers for the gus gene, to amplify a fragment of 420 bp (forward TTGGGCAGGCCAGCGTATCGT and reverse ATCACGCAGTTCAACGCTGAC); for hptII gene, the primers used amplify a fragment of 473 bp (forward TCCGCAAGTGCTTGACATTGG and reverse ATGTTGGCGACCTCGTATTGG). Products were visualized by electrophoresis in 1.0 % agarose gel, and then transferred by capillarity to a nylon membrane (Hybond-N+ Amersham Pharmacia Biotech) (Sambrook et al. 1989SAMBROOK JF, FRITSCH EF AND MANIATIS T. 1989. Molecular Cloning: a laboratory manual, 2nd ed., New York: Cold Spring Harbor Laboratory Press, 1626 p.). The membrane was hybridized with [α32P] dCTP labeled probes from gus or htpII genes.
GENETIC TRANSFORMATION USING FOUR TYPES OF EXPLANTS
Isolated embryo and seed
After peeling and disinfesting seeds according to Cabral et al. (2011CABRAL GB, CARNEIRO VTC, LACERDA AL, DO VALLE CB, MARTINELLI AP AND DUSI DMA. 2011. Somatic embryogenesis and organogenesis in apomictic and sexual Brachiaria brizantha. Plant Cell Tissue Organ Cult 107: 271-282.), embryos were excised. These were cultured on somatic embryo induction medium (M1.2), in the dark, for one or three days, 110 embryos per treatment, two replications each treatment. To evaluate the gus expression stability, the embryos were bombarded and histochemically evaluated after 1 d (control of bombardment), 10 d, and 20 d. To establish the best time of induction prior to bombardment and type of explant, both, peeled seeds and isolated embryos were evaluated. The explants were plated on somatic embryogenesis induction medium M1.2 for 1, 3, 5 or 7 d of culture before bombardment, in a total of 75 explants per treatment. After the induction period, the explants were placed in Petri dishes with M1.2 medium and bombarded with plasmid pAct1-D. After 48 h a GUS histochemical assay was performed (Jefferson 1987JEFFERSON RA. 1987. Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol Biol Report 5: 387-405.).
Embryogenic callus
Calluses resulting from seeds cultivated in embryogenesis induction M1.3 medium (Table I; Cabral et al. 2011CABRAL GB, CARNEIRO VTC, LACERDA AL, DO VALLE CB, MARTINELLI AP AND DUSI DMA. 2011. Somatic embryogenesis and organogenesis in apomictic and sexual Brachiaria brizantha. Plant Cell Tissue Organ Cult 107: 271-282., 2015) for 7, 15 or 30 d, were transferred to bombardment medium in Petri dishes, and maintained in the dark, in culture room for 24 h before bombardment with pAHUG plasmid. A minimum of 390 calluses were bombarded for each treatment. After bombardment, the calluses were kept for 24 h in the dark. Half of the calluses was submitted to GUS histochemical assay while the other half was transferred either to the induction medium M1.3 with hygromycin (5 mg/L) and kept in the dark for 20 d, or to the regeneration medium, directly to the light. After this period, they were transferred to regeneration medium MS3 with hygromycin (10 mg/L) and kept at 12 h photoperiod, for 30 d. The resultant selected embryogenic calluses which regenerated 2-cm-long shoots, resistant to hygromycin, were transferred to plant maintenance medium (PMM), for 30 d supplemented with hygromycin (20 mg/L). Regenerated plantlets were transferred to 1:1 (v/v) substrate:vermiculite in the greenhouse. DNA was extracted from leaves for PCR analysis. Putative transgenic plants were transferred to soil and another PCR reaction was performed to confirm the presence of the transgene.
Cell suspensions
Cell suspensions (CS) induced in M1.3 or NBBAP pH 4 (Table I) were placed in filter paper to drain the excess of medium. Approximately 200 to 300 mg of cell aggregates were spread on the bombardment medium, in a diameter of 1 cm avoiding the center of the Petri dish, and left for 4 to 24 h at room temperature. Plasmids pAHC27, pAct1D, pGPro1pUbi1, and pGPro2pUbi1 were bombarded at 900 psi and the last two were also bombarded at 1,200 psi. After bombardment, the cells were incubated in the dark for 24 or 48 h. Part of the cells was collected for GUS histochemical analysis, the other part was transferred to liquid NBBAP pH 4, or liquid or solid M1.3, with or without hygromycin (10 mg/L), for two weeks.
CS cultivated in liquid media were then transferred to pre-regeneration DD1 or PRM liquid medium containing 10, 20 or 30 mg/L hygromycin, under orbital agitation of 100 rpm in the dark. After one week, cells were transferred to regeneration medium MS3, liquid or solid, containing hygromycin (20 mg/L), at 16 h photoperiod and 50 µmol m-2 s-1 intensity light for 3 d, when they were transferred to 75 µmol m-2 s-1 light intensity.
CS from solid M1.3 were cultivated for 20 d in the dark and then transferred to regeneration media MS3 or NBreg, supplemented with hygromycin (20 mg/L) for 15 d. Regenerated shoots were transferred to PMM medium, without hygromycin, in magenta boxes, for 20 d. Shoots were then individually transferred to test tubes with PMM medium. After one month, the plantlets were acclimatized in 1:1 (v/v) substrate:vermiculite and thereafter transferred to soil in the greenhouse.
RESULTS AND DISCUSSION
ISOLATED EMBRYOS AND SEEDS
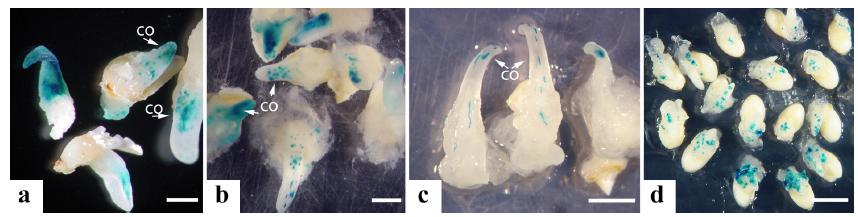
The highest number of explants expressing gus was observed in embryos bombarded after 3 d in induction medium M1.2 (Table II, Fig. 1a). In general, the expression of gus decreased at 10 d (Fig. 1b) and 20 d after bombardment (Fig. 1c), comparing to the 1 d control, embryos induced for 1 d being more affected. The reduction of expression after bombardment possibly indicates low plasmid integration into the plant genome, but also a low proliferation of cells stably expressing gus. Coleoptile cells elongate during development, giving a linear pattern of expression (Fig. 1c). The expression of gus was more evident in coleoptile cells (Fig. 1a-c) rather than in friable callus cells, in which somatic embryo development preferentially occurs (Cabral et al. 2011CABRAL GB, CARNEIRO VTC, LACERDA AL, DO VALLE CB, MARTINELLI AP AND DUSI DMA. 2011. Somatic embryogenesis and organogenesis in apomictic and sexual Brachiaria brizantha. Plant Cell Tissue Organ Cult 107: 271-282.).
Expression of gene gus in Brachiaria brizantha cv. Marandu isolated embryos and seeds after bombardment with pAct1-D: (a) embryos cultured for 3 d in induction medium and 48 h after bombardment; (b) embryos induced for 3 d before bombardment and cultivated for 10 d after bombardment; (c) embryos 20 d after bombardment; (d) dehulled seeds 48 h after bombardment. co = coleoptile. Bars: (a) 0.5 cm; (b, c) 1 cm; (d) 2 cm.
Expression of gus in isolated embryos of Brachiaria brizantha cv. Marandu after bombardment, according to different periods of time in induction medium prior to bombardment.
Table III shows the percentage of gus expression in isolated embryos and peeled seeds subjected to different periods of culture in somatic embryogenesis induction medium and bombardment with plasmid pAct1-D. The percentage of isolated embryos showing gus expression increased with the days in culture before bombardment reaching up to 39 % with 5 d decreasing at 7 d in culture. The percentage of seeds with gus expression also increased with the period in culture in M1.2 medium before bombardment, being 37 % with 1 d, up to 78 % with 7 d. Besides being easier to manipulate, the higher percentage of seeds expressing gus (Fig. 1d) when compared to isolated embryos, makes seeds a preferred explant for testing vectors and constructions via transient expression after bombardment.
Expression of gus in isolated embryos and seeds of Brachiaria brizantha cv. Marandu, 75 explants per treatment cultivated before bombardment on induction medium M1.2 for different periods of time, evaluated 48 h after bombardment with pAct1-D.
EMBRYOGENIC CALLUSES
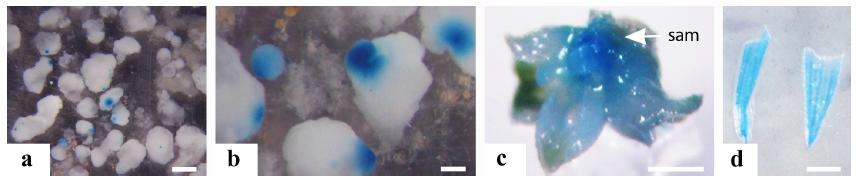
Calluses cultivated for 7, 15 or 30 d before bombardment showed gus expression 24 h after bombardment with the plasmid pAHUG (Fig. 2a). Calluses transferred directly to regeneration medium, promptly differentiated somatic embryos that turned out to be chimeric for gus expression (Fig. 2b). Calluses that were initially transferred to induction medium supplemented with hygromycin (5 mg/L) proliferated into embryogenic calluses. After multiplying and transferring to regeneration medium with a higher hygromycin concentration (10 mg/L), these calluses produced a few shoots after 30 d (Fig. 2c). The percentage of shoots resistant to hygromycin obtained from calluses cultivated for 7 d in induction medium prior to bombardment with pAHUG was 1.5 %. This percentage increased when calluses were cultivated for 15 d (3 %) and 30 d (8.5 %). Regeneration of putative transgenic plantlets was observed only from calluses induced for 30 d before bombardment (Fig. 2d). Only one of the regenerated plants survived acclimatization (Fig. 2e). For this plant, GUS histochemical assay was negative. However, PCR analysis showed a fragment of 473 bp of the hptII gene, with the specificity confirmed by PCR DNA gel blot analysis, and no amplification corresponding to gus (Fig. 2f, g). The transgenic plant was transferred to soil and after two months (Fig. 2h) a PCR performed with DNA extracted from fragments of seven individual leaves did not amplify the hptII. It is possible that the plant was a chimera and during the development, the transgenic tillers were lost.
Transformation of Brachiaria brizantha cv. Marandu embryogenic calluses via biolistics, and molecular analyses of putative transformed plants: (a) gus expression in embryogenic calluses induced for 30 d on M1.3 medium and bombarded with pAHUG; (b) longitudinal section of a somatic embryo developed from embryogenic callus bombarded with pAHUG, and cultivated in regeneration medium, showing gus expression in the coleoptile and leaf primordium, and absence of expression in the shoot apical meristem; (c) germinating somatic embryos in MS3 medium with hygromycin showing anthocyanin pigments; (d) hygromycin resistant plantlets (left) and untransformed control (right) in medium containing 20 mg/L hygromycin; (e) acclimatized hygromycin resistant plantlet; (f) detection of gus (1-5) and hptII (6-10), by PCR; pAHUG (5 and 10) as positive control; putative transformed plants (1-4 and 6-9); column 6 is a plant showing an amplified band corresponding to hptII (arrow), M = marker; (g) DNA gel blot with positive signal for hptII in column 6 (arrow); (h) two-month-old plant, arrow indicate sampling areas for PCR. co = coleoptile; lp = leaf primordium; sam = shoot apical meristem; pe = proembryos. Bars: (e) 1 cm; (f) 0.2 mm; (g, h) 2 cm.
CELL SUSPENSIONS (CS)
CS plated 24 h before bombardment got shrunk and brownish after bombardment, and did not show gus expression. Cells bombarded 4 h after introduction in bombardment medium showed best results, in terms of viability and gene expression. Cell aggregates bombarded with plasmids pGPro1pUbi1 and pGPro2pUbi1 showed transient gus expression in a higher percentage when pressure condition was 1,200 psi compared to 900 psi (Table IV). Nevertheless, at 1,200 psi they died after one month of culturing.
Only the cell aggregates kept for two weeks in NBBAP containing hygromycin (10 mg/mL), and transferred to DD1 supplemented with hygromycin (10 mg/L) or PRM with hygromycin (30 mg/L) proliferated new embryogenic calluses resistant to the antibiotic. These embryogenic cell agregates kept their proliferative capacity after successive subcultures, during 10 months, and showed stable gus expression (Fig. 3a, b). The gus and hptII genes were detected by PCR (Fig. 3c, d), and confirmed by DNA gel blot analysis of the amplified bands (Fig. 3e, f), ratifying stable transformation. These stably transformed embryogenic cell aggregates did not regenerate shoots and at this point, the calluses had a vitrified aspect, possibly due to several subcultures.
Cell suspension of Brachiaria brizantha transformed by biolistic and molecular analysis of putative transformants. (a-b) cell suspension obtained in NBBAP (pH 4) after bombardment with pGPro1pUbi1; (a) stable gus expression in embryogenic calluses after cultivation for 10 m in regeneration medium (RM) in the presence of hygromycin (20 mg/L); (b) detail of (a) with proembryos expressing gus; (c-d) detection of gus (c) and hptII (d) by PCR: column 1, DNA from hygromycin resistant calluses (arrows), column 2 negative control (water), column 3 plasmid pGPro1pUbi1 used as positive control; (e-f) PCR DNA gel blot hybridized with a gus probe (e, arrows) and with hptII probe (f, arrows). M, molecular weight marker (1 kb). pe = proembryos. Bars: (a) 1 cm; (b) 0.2 cm.
Cell suspensions bombarded with pAct1D and pAHC27 also showed gus expression (Table IV; Fig. 4a, b). CS bombarded with pAHC27 resulted in the regeneration of three multiple shoots, in a medium without selective agent, and when isolated two shoots showed gus expression (Fig. 4c, d).
Cell suspension of Brachiaria brizantha transformed by biolistic. (a, b) gus expression after cell suspension bombardment with pAHC27, 24 h after bombardment; (c) gus expression in shoot apical meristem; (d) leaf fragments expressing gus. sam = shoot apical meristem. Bars: (a, c, d) 1 cm; (b) 0.2 cm.
The competence to regenerate transgenic plants is generally genotype-dependent, a limiting step in plant biotechnology (Harwood 2012HARWOOD WA. 2012. Advances and remaining challenges in the transformation of barley and wheat. J Exp Bot 63: 1791-1798., Tie et al. 2012TIE W, ZHOU F, WANG L, XIE W, CHEN H, LI X AND LIN Y. 2012. Reasons for lower transformation efficiency in indica rice using Agrobacterium tumefaciens-mediated transformation: lessons from transformation assays and genome-wide expression profiling. Plant Mol Biol 78: 1-18.). In Brachiaria,Ishigaki et al. (2012ISHIGAKI G, GONDO T, SUENAGA K AND AKASHI R. 2012. Fertile transgenic Brachiaria ruziziensis (ruzigrass) plants by particle bombardment of tetraploidized callus. J Plant Physiol 169: 546-549.) reported transgenic plants from the diploid and from the colchicine-tetraploidized calluses of B. ruziziensis. They recovered two transgenic plants that presented a normal phenotype, however, no seeds were produced by the diploid one, and a fertile transgenic plant was obtained only from an artificially tetraploidized callus (Ishigaki et al. 2012). In this work, we showed that Brachiaria brizantha cv Marandu, an apomictic tetraploid, is competent to regenerate transgenic plants using either embryogenic calluses or cell suspension systems when bombarded and selected in hygromicin, besides the production of some escapes. Nowadays, we have been using these systems with different selections to obtain a high throughput method to produce B. brizantha transgenic plants.
The present work provides valuable information towards the development of a methodology to obtain transgenic plants of B. brizantha, which is still considered as a recalcitrant monocot species.
CONCLUSIONS
In conclusion, all the explants tested and earlier successfully reported as efficient for somatic embryogenesis were suitable for transient gene expression after bombardment, showing gus expression and resistance to hygromycin. B. brizantha mature seeds cultured for seven days in somatic embryo induction medium, embryogenic calluses cultured for 15 or 30 days, as well as cell suspensions, proved to be the best explants to test vectors and constructions via transient gene expression after particle bombardment. The use of embryogenic calluses and cell suspensions as targets, allowed for the recovery of transgenic plants. However, the combined action of the marker gene hptII expression and selection in hygromycin seems to favor regeneration from untransformed cells, suggesting that other selective agents should be tested for more effectiveness in selecting the transformed cells.
ACKNOWLEDGMENTS
We would like to thank Dr. Thilmony, USDA-ARS, Dr. Phil Mullineaux and Dr. Roger Hellens, John Innes Centre, and the Biotechnology and Biological Sciences Research Council (BBSRC) for providing the vectors pGPro1 and pGPro2, and Dr. Cacilda B. do Valle, Embrapa Beef Cattle for providing the seeds. This work was financially supported by Embrapa and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). APM acknowledges CNPq for research fellowships.
REFERENCES
- CABRAL GB, CARNEIRO VTC, LACERDA AL, DO VALLE CB, MARTINELLI AP AND DUSI DMA. 2011. Somatic embryogenesis and organogenesis in apomictic and sexual Brachiaria brizantha. Plant Cell Tissue Organ Cult 107: 271-282.
- CABRAL GB, CARNEIRO VTC, ROSSI ML, DA SILVA JP, MARTINELLI AP AND DUSI DMA. 2015. Plant regeneration from embryogenic callus and cell suspensions of Brachiaria brizantha. In Vitro Cell Dev Biol - Plant 51: 369-377.
- CHRISTENSEN AH AND QUAIL PH. 1996. Ubiquitin promoter-based vectors for high-level expression of selectable and/or screenable marker genes in monocotyledonous plants. Transgenic Res 5: 213-218.
- GUIMARÃES LA, DUSI DMA, MASIERO S, RESENTINI F, GOMES ACMM, SILVEIRA ED, FLORENTINO LH, RODRIGUES JCM, COLOMBO L AND CARNEIRO VTC. 2013. BbrizAGL6 is differentially expressed during embryo sac formation of apomictic and sexual Brachiaria brizantha plants. Plant Mol Biol Report 31: 1397-1406.
- HARWOOD WA. 2012. Advances and remaining challenges in the transformation of barley and wheat. J Exp Bot 63: 1791-1798.
- ISHIGAKI G, GONDO T, SUENAGA K AND AKASHI R. 2012. Fertile transgenic Brachiaria ruziziensis (ruzigrass) plants by particle bombardment of tetraploidized callus. J Plant Physiol 169: 546-549.
- JEFFERSON RA. 1987. Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol Biol Report 5: 387-405.
- MCELROY D, ZHANG W, CAO J AND WU R. 1990. Isolation of an efficient actin promoter for use in rice transformation. Plant Cell 2: 163-171.
- MONTEIRO LC, VERZIGNASSI JR, BARRIOS SCL, VALLE CB, FERNANDES CD, BENTEO GL AND LIBÓRIO CB. 2016. Brachiaria decumbens intraspecific hybrids: characterization and selection for seed production. J Seed Sci 38: 62-67.
- SALLAUD C ET AL. 2003. Highly efficient production and characterization of T-DNA plants for rice (Oryza sativa L.) functional genomics. Theor and Appl Genet 106: 1396-1408.
- SAMBROOK JF, FRITSCH EF AND MANIATIS T. 1989. Molecular Cloning: a laboratory manual, 2nd ed., New York: Cold Spring Harbor Laboratory Press, 1626 p.
- SHARMA KK, BHATNAGAR-MATHUR P AND THORPE TA. 2005. Genetic transformation technology: status and problems. In Vitro Cell Dev Biol - Plant 41: 102-112.
- SHIRASUNA RT. 2015. Urochloa. In: Jardim Botânico do Rio de Janeiro. Lista de espécies da flora do Brasil, Rio de Janeiro. Dísponível em: <http://floradobrasil.jbrj.gov.br/jabot/floradobrasil/FB26028>. Acessado em 4 de Janeiro de 2016.
» http://floradobrasil.jbrj.gov.br/jabot/floradobrasil/FB26028 - SILVEIRA ED, GUIMARÃES LA, DUSI DMA, SILVA FR, MARTINS NF, CARMO COSTA MM, ALVES-FERREIRA M AND CARNEIRO VTC. 2012. Expressed sequence-tag analysis of ovaries of Brachiaria brizantha reveals genes associated with the early steps of embryo sac differentiation of apomictic plants. Plant Cell Report 31: 403-416.
- SILVEIRA ED, RODRIGUES JCM, CABRAL GB, LEITE JÁ, COSTA SS AND CARNEIRO VTC. 2003. Evaluation of exogenous promoters for use in Brachiaria brizantha transformation. J Plant Biotechnol 5: 87-93.
- SOOD P, BHATTACHARYA A AND SOOD A. 2011. Problems and possibilities of monocot transformation. Biol Plant 55: 1-15.
- THILMONY R, GUTTMAN M, CHINIQUY D AND BLECHL A. 2006. pGPro1, a novel binary vector for monocot promoter characterization. Plant Mol Biol Report 24: 57-69.
- THILMONY R, GUTTMAN M, THOMSON JG AND BLECHL AE. 2009. The LP2 leucine-rich repeat receptor kinase gene promoter directs organ-specific, light-responsive expression in transgenic rice. Plant Biotechnol J 7: 867-882.
- TIE W, ZHOU F, WANG L, XIE W, CHEN H, LI X AND LIN Y. 2012. Reasons for lower transformation efficiency in indica rice using Agrobacterium tumefaciens-mediated transformation: lessons from transformation assays and genome-wide expression profiling. Plant Mol Biol 78: 1-18.
- VALLE CB AND SAVIDAN Y. 1996. Genetics, cytogenetics, and reproductive biology of Brachiaria. In: Miles JW, Maass BL and Valle CB (Eds), Brachiaria: biology, agronomy, and improvement, Cali-Colômbia: CIAT, p. 147-163.
Publication Dates
-
Publication in this collection
Apr-Jun 2018
History
-
Received
20 Oct 2017 -
Accepted
18 Dec 2017