Abstract
Abstract: Medicinal plants have long been used as an alternative to traditional drugs for the treatment of inflammatory conditions due to the classical side effects and restricted access of various commercially available drugs, such as steroids (GCs) and nonsteroidal anti-inflammatory drugs (NSAIDs). Sambucus australis is a Brazilian herb that is commonly used to treat inflammatory diseases; however, few studies have examined the use of this species in the treatment of inflammatory conditions. The present study aims to evaluate the potential anti-inflammatory activity of S. australis in vitro. We established spleen cell cultures stimulated with pokeweed mitogen (PWM) to evaluate the production of proinflammatory cytokines, such as IL-4, IL-5, IFN-y, and IL-10 (by ELISA), and the expression of the transcription factor NF-kB (by RT-PCR). In addition, we evaluated the levels of nitric oxide in macrophage cultures and the membrane-stabilizing activity of S. australis methanolic extract (EMSA). Treatment with EMSA at concentrations of 100, 50, 25 and 12.5 µg/ml significantly decreased IL-4 (p<0.001) and IL-5 (p<0.001) levels. Treatment with 100 µg/ml EMSA reduced IFN-у (p<0.001) levels. Moreover, at 100 mg/ml, EMSA also increased IL-10 production and reduced NF-kB expression (p<0.01). In macrophage cultures stimulated with LPS, EMSA decreased nitric oxide levels (p<0.001) at all concentrations tested (100, 50, 25 and 12.5 µg/ml). Additionally, EMSA had a protective effect in the erythrocyte membrane stabilization assay. Taken together, these results suggest that S. australis has anti-inflammatory potential in vitro, characterized by the reduction of both inflammatory cytokines and the expression of NF-kB along with the up-regulation of IL-10.
Key words
Sambucus australis; elderberry; anti-inflammatory; NF-kB
INTRODUCTION
Although inflammation is a key process in the resolution of many physiopathological alterations, the persistence of inflammation can be harmful (Serhan et al. 2010SERHAN CN, WARD PA and GILROY DW. 2010. Fundamentals of Inflammation. Cambridge University Press, EUA, 473 p.). The inflammatory process initially involves many proinflammatory mediators, such as arachidonic acid metabolites, and it also includes cytokines (IL-4, IL-5, TNF, IFN-y, IL-13), which together increase the permeability of the endothelial wall and promote the formation of edema and the recruitment of polymorphonuclear leukocytes (PMN) and macrophages. These changes produce the characteristic signs of inflammation, which include warmth, pain, redness and swelling, and can lead to loss of tissue function (Serhan et al. 2010, Norling and Serhan 2010NORLING LV and SERHAN CN. 2010. Profiling in resolving inflammatory exudates identifies novel anti-inflammatory and pro-resolving mediators and signals for termination. J Intern Med 268: 15-24.).
Among the many existing inflammatory mediators, nuclear factor kappa B (NF-kB) plays an important role, as it is responsible for the production of cytokines, chemokines, and growth factors regulating the expression of genes involved in the immune and inflammatory responses (Frode-Saleh and Calixto 2000). Therefore, the detection of NF-kB is essential to determine whether a specific molecule can act as an anti-inflammatory agent (Hanada and Yoshimura 2002HANADA T and YOSHIMURA A. 2002. Regulation of cytokine signaling and inflammation. Cytokine Growth Factor Rev 13: 413-421.). Anti-inflammatory mediators (IL-10, TGF-b) can attenuate the exacerbation of the immune response, promoting the resolution of inflammation and the restoration of injured tissue (Serhan et al. 2010, Ariel and Serhan 2012ARIEL A and SERHAN CN. 2012. New Lives Given by Cell Death: Macrophage Differentiation Following Their Encounter with Apoptotic Leukocytes during the Resolution of Inflammation. Front Immunol 31: 3-4.).
Considering that inflammation has an impact on a large number of immune-mediated diseases, such as systemic lupus erythematosus and rheumatoid arthritis, systemic inflammatory response syndrome (sepsis), asthma and rhinitis, and considering the side effects of available drugs [(glucocorticoids (GCs) and nonsteroidal anti-inflammatories (NSAIDs)] to treat inflammatory conditions, it is necessary to identify new and more effective drugs with fewer side effects to develop significant immunomodulatory therapeutic protocols with beneficial effects on inflammatory diseases (Kim 2004KIM HP, SON KH, CHANG HW and KANG SS. 2004. Anti-inflammatory plant flavonoids and cellular action mechanisms. J Pharmacol Sci 96: 229-245., Stahn et al. 2007STAHN C, LÖWENBERG M, HOMMES DW and BUTTGEREIT F. 2007. Molecular mechanisms of glucocorticoid action and selective glucocorticoid receptor agonists. Mol Cell Endocrinol 275: 71-78., Serhan et al. 2007SERHAN CN, BRAIN SD, BUCKLEY CD, GILROY DW, HASLETT C, O’NEILL LA, PERRETTI M, ROSSI AG and WALLACE JL. 2007. Resolution of inflammation: state of the art, definitions and terms. FASEB J 21: 325-332.).
The study of natural products has led to the discovery of many clinically useful drugs, and approximately 30% of the medicines produced by developed countries are derived from natural products. Sambucus australis Cham. & Schltdl of the genus Sambucus (Judd et al. 1999JUDD WS, CAMPBELL CS, KELLOG EA and STEVENS PF. 1999. Plant Systematics: a Phylogenetic Approach. Massachusetts: Sinauer Associates.), popularly known as the elderberry, is a medicinal herb commonly used for the treatment of inflammatory disease in “Todos os Santos” Bay, Bahia, Brazil. However, few studies have explored the anti-inflammatory potential of this herb to date.
The chemical components of S. australis Cham. & Schltdl includes flavonoids, quercetin glycosides, triterpenes, volatile oils and phenolic acids, which resembles the composition of the species S. nigra. This species is native to Europe and is well described in the literature (Lamaison 1991LAMAISON JL, PETIT JEAN-FREYTET C and CARNAT A. 1991. Présence de 3-glucoside et de 3-rutinoside dʼisorhamnétine dans les fleurs de Sambucus nigra L. Ann Pharm Fr 49: 258-262., Bacigalupo 1974BACIGALUPO NM. 1974. Sambucaceae. In: Burkart A (Ed), Flora Ilustrada de Entre Rios (Argentina), Caprifoliaceae. INTA 1974: 50-52., Alice 1990ALICE CB, SIQUEIRA NCS, HALBIG JC, MENTZ LA, SILVA GAA and GOETTEMSEI. 1990. Diagnose comparativa de compostos polifenólicos de Sambucus nigra L. e Sambucus australis Cham. & Schldht. Rev Bras Farm 71: 88-90.).
One of the main component of S. australis is the ursolic acid (UA). UA is a pentacyclic triterpene with antitumor and anti-inflammatory activity due to the inhibition of NF-kB activation; it has antioxidant and antibacterial effects, promotes hypoglycemia and prevents the deposition of fat (Rao et al. 2011RAO VS, DE MELO CL, QUEIROZ MGR, LEMOS TLG, MENEZES DB, MELO TS and SANTOS FA. 2011. Ursolic Acid, a pentacyclic triterpene from Sambucus australis, prevents abdominal adiposity in mice fed a high-fat diet. J Med Food 14: 1375-1382., Alqahtani 2013ALQAHTANI A, HAMID K, KAM A, WONG KH, ABDELHAK Z, RAZMOVSKI-NAUMOVSKI V, CHAN K, LI KM, GROUNDWATER PW and LI GQ. 2013. The pentacyclic triterpenoids in herbal medicines and their pharmacological activities in diabetes and diabetic complications. Curr Med Chem 20: 908-931., Kim and Moon 2015KIM ES and MOON A. 2015. Ursolic acid inhibits the invasive phenotype of SNU-484 human gastric cancer cells. Oncol Lett 9: 897-902., Chun et al. 2014CHUN J, LEE C, HWANG SW, IM JP AND KIM JS. 2014. Ursolic acid inhibits nuclear factor-κB signaling in intestinal epithelial cells and macrophages, and attenuates experimental colitis in mice. Life Sci 110: 23-34., Zhang et al. 2014ZHANG JF, GAO L, DUAN HQ, WANG W, LI Y and MA YG. 2014. Mechanism of antitumor effect of ursolic acid on T (8;21) leukemia cell kasumi-1. Zhongguo Shi Yan Xue Ye Xue Za Zhi 22: 687-691., Yoon et al. 2014YOON JH, YOUN K, HOCT, KARWE MV, JEONG WS AND JUN MP. 2014. Coumaric acid and ursolic acid from Corni fructus attenuated β-amyloid(25-35)-induced toxicity through regulation of the NF-κB signaling pathway in PC12 cells. J Agric Food Chem 62: 4911-4916., Ma et al, 2014MA JQ, DING J, ZHANG L AND LIU CM. 2014. Ursolic acid protects mouse liver against CCl4-induced oxidative stress and inflammation by the MAPK/NF-κB pathway. Environ Toxicol Pharmacol 37: 975-983., Zhao et al. 2012ZHAO J, CHEN J, LIU T, FANG J, WAN J, ZHAO J, LI W, LIU J, ZHAO X and CHEN S. 2012. Anti-viral effects of urosolic acid on guinea pig cytomegalovirus in vitro. J Huazhong Univ Sci Technolog Med Sci 32: 883-887., Nascimento et al. 2014NASCIMENTO PG, LEMOS TL, BIZERRA AM, ARRIAGA ÂM, FERREIRA DA, SANTIAGO GM, BRAZ-FILHO R AND COSTA JG. 2014. Antibacterial and antioxidant activities of ursolic acid and derivatives. Molecules 19: 1317-1327., Liobikas 2011LIOBIKAS J, MAJIENE D, TRUMBECKAITE S, KURSVIETIENE L, MASTEIKOVA R, KOPUSTINSKIENE DM, SAVICKAS A and BERNATONIENE J. 2011. Uncoupling and antioxidante effects of ursolic acid in isolated rat heart mitochondria. J Nat Prod 74: 1640-1644.). These studies suggest that S. australis Cham. & Schltdl may have immunomodulatory effect due to the presence of UA.
Although different species of the genus Sambucus have been studied and evaluated for their pharmacobiological effects, S. australis Cham. & Schltdl is rarely investigated (Badescu et al. 2012BADESCU L, BADULESCU O, BADESCU M and CIOCOIU M. 2012. Mechanism by Sambucus nigra Extract Improves Bone Mineral Density in Experimental Diabetes.Evid. Based Complement Alternat Med, 6 p.). Although its flowers and leaves are used as a diuretic, anti-inflammatory agent, and laxative and to treat feverish conditions resulting from respiratory diseases, no scientific studies have evaluated the mechanisms of the anti-inflammatory activity of S. australis Cham. & Schltdl to date (Scopel 2007SCOPEL M, NUNES E, VIGNOLI-SILVA M, VENDRUSCOLO GS, HENRIQUES AT and MENTZ LA. 2007. Caracterização farmacobotânica das espécies de Sambucus (Caprifoliaceae) utilizadas como medicinais no Brasil. Parte I. Sambucus nigra L. Rev Bras Farmacogn 17: 249-261.).
Therefore, the aim of this study is to investigate the potential anti-inflammatory activity of S. australis Cham. & Schltdl in an in vitro murine model of inflammation.
MATERIALS AND METHODS
COLLECTION AND IDENTIFICATION OF S. australis Cham. & Schltdl.
To avoid loosing the volatile oils within S. australis Cham. & Schltdl, the aerial parts of the herb were obtained before sunrise and stored in ventilated bags. These parts were then allowed to dry in a ventilated room with no sun and no humidity prior to the preparation of extracts. Representative specimens of the plant parts were analyzed to confirm the identity of the species. The species was confirmed and registered in the Herbarium UNEB, N°. 28311 at the Universidade do Estado da Bahia, Campus VIII, Paulo Afonso, Bahia, Brazil.
PREPARATION OF METHANOLIC EXTRACT FROM S. australis Cham. & Schltdl.
The dried leaves were crushed in a Willye mill (ET-650-Tecinal), and the extract was prepared by successive macerations with methanol using the Soxhlet technique (Fisatom) (The leaves were soaked 3 times for 4 with each reagent). After filtration, the extracts were concentrated in vacuo at 40°C with a rotary evaporator. They were placed in an oven to evaporate all the solvent and kept frozen until use.
STANDARDIZATION OF THE METHANOLIC EXTRACT OF S. australis Cham. & Schltdl.
The methanolic extract of S. australis Cham. & Schltdl (EMSA) was characterized and the standardization was developed and validated for the quantification of ursolic acid (AU) by high performance liquid chromatography (HPLC) using a Shimadzu Prominence LC-20AT with a SPD-M20A diode array detector, SIL-20AC autosampler, CTO-20A oven and DGU-20A5 degasser. UA is described as a major component in S. australis Cham. & Schltdl, and according to the literature, it has various biological activities; therefore, it was chosen as the standard. A Luna C-18 column (150 mm x 4.6 mm x 5 μm Phenomenex) was used for chromatographic separation. The mobile phase contained H2O (solvent A) and acetonitrile 0.1% TFA (solvent B) at a ratio of 20:80, respectively. The flow rate was set at 1.0 ml/minute, the detection wavelength was 203 nm and the temperature was set to 35°C. Nylon filters (0.45 μm) were used (Whatman) to filter the samples, and all solvents were of HPLC grade.
Calibration curves were plotted over the concentration range of 0.031 to 1.00 mg/ml of an external standard, and 20 μL of each concentration was administered in triplicate. The limit of detection (LOD) and limit of quantitation (LOQ) were 2.289 μg and 0.755 μg, respectively. For the quantification of UA in EMSA, 20 μL of the methanol extract was injected at a concentration of 12.0 mg/ml. This procedure was performed in the Bioprospecting Phytochemistry Laboratory, Department of Molecular Sciences, Universidade Federal Rural de Pernambuco, Brazil.
ANIMALS
Male BALB/c mice (25-30 g) were used in this study. These animals were obtained from the Fundação Oswaldo Cruz, Bahia, Brazil, and allowed free access to food and water under controlled environmental conditions. All experimental procedures were approved by the Ethics Committee for the Use of the Institute of Animal Health Sciences, Instituto de Ciências da Saúde da Universidade Federal da Bahia, Brazil (protocol number: 028/2012).
SPLEEN CELL CULTURE
BALB/c mice were anesthetized, and their spleens were aseptically removed. After maceration of the organ, a cell count was performed, and the spleen cells (5x106 cells/ml) of normal mice were resuspended in RPMI. These cells were cultured at 37°C in 5% CO2. Two positive controls using PWM (1 μg/ml or 2.5 μg/ml) and one negative control using RPMI were used. For each of these conditions, we had wells with and without different concentrations of EMSA (100, 50, 25 and 12.5 µg/ml). After 24, 48 and 72 hours, supernatants were collected to assess inflammatory markers.
EVALUATION OF THE CYTOTOXICITY OF THE EMSA BY MTT-TETRAZOLIUM
In 96-well plates, 100 µl of a suspension (5 x 106 cells/ml) of spleen cells from normal mice (BALB/c) supplemented with 5% FCS both with or without 50 µl solution PWM (final concentration 1.0 µg/ml) in the presence or absence of different dilutions of the EMSA (100, 50, 25 and 12.5 µg/ml) was added to each well. The cultures were incubated for 72 hours at 37°C in a 5% CO2 atmosphere. After this period, MTT-tetrazolium [3-(4,5-dimethylthiazol-2-yl-2,5-diphenyltetrazolium bromide)] (5 mg/ml) Sigma-Aldrich (St. Louis, MO, USA) was added to the culture and incubated for an additional 4 hours. This compound was then removed from the culture medium and added to 100 µl of DMSO (dimethylsulfoxide, Sigma). Cell viability was determined based on the colorimetric method using an absorbance at 560 nm for all concentrations; viability experiments were performed in triplicate (Mosmann 1983MOSMANN T. 1983. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65(1-2): 55-63.).
IL-4, IL-5, IL-10 AND IFN- у PRODUCTION
Cytokine levels were quantified from the spleen cell culture supernatants at 24, 48 and 72 hours by standard ELISA as recommended by the manufacturer (BD Pharmingen, USA).
MEMBRANE STABILITY TEST
The stability of the membrane was determined as previously described (Govindappa et al. 2011GOVINDAPPA M, NAGA SRAVYA S, POOJASHRI MN, SADANANDA TS, CHANDRAPPA CP, SANTOYO G, SHARANAPPA P and ANIL KUMAR NV. 2011. Antimicrobial, antioxidant and in vitro anti-inflammatory activity and phytochemical screening of water extract of Wedelia trilobata (L.) Hitchc. J Med Plants Res 5: 5718-5729., Shinde et al. 1999SHINDE UA, PHADKE AS, NARI AM, MUNGANTIWAR AA, DIKSHIT VJ and SARAF MN. 1999. Membrane stabilization activity - a possible mechanism of action for the anti-inflammatory activity of Cedrus deodara wood oil. Fitoterapia 70: 251-257.) with several modifications. The assay was based on the effect of temperature on the membrane; with increasing temperature, the rate of diffusion of molecules (which require activation energy) to the cell increases due to molecular motion, increasing hemolysis.
Initially, whole blood was obtained from mice and transferred into heparinized tubes, followed by centrifugation at 1000 g for 10 minutes. The supernatant was discarded, leaving only the portion with erythrocytes. This portion was washed 3x with saline solution (0.9% m/v), alternating with centrifugation. After the last wash, the portion of erythrocytes was also diluted with normal saline, leaving the 10% v/v. The final volume of the mixture was 5 ml, which contained 4.5 ml of the standard (acetylsalicylic acid, 200 μg/ml) or test drug (EMSA 100, 50, 25, 12.5 μg/ml) and 500 μl of erythrocyte suspension. Saline (9%) was used for the control. All tubes were kept in a water bath at 56°C for 30 minutes. After the incubation period, the tubes were cooled in warm water. After the tubes were cooled, they were centrifuged at 800 g for 5 minutes, and the supernatants of the samples were placed in a 96-well plate and read at an absorbance of 560 nm. The procedure was performed in duplicate. The percentage of membrane stabilization and the inhibition of hemolysis was calculated according to the equation:
NITRIC OXIDE (NO) PRODUCTION
For evaluation of NO production, BALB/c mice were intraperitoneally injected with 20 mg LPS. At 72 hours after the injection, peritoneal lavage was performed to obtain macrophages. The macrophage culture was performed using 5 µg/ml of LPS in vitro. EMSA was added at 100, 50, 25 and 12.5 μg/ml. After 24 hours, NO production was determined by the Griess reaction using the nitrite concentration in culture (Green et al. 1982GREEN LC, WAGNER DA, GLOGOWSKI J, SKIPPER PL, WISHNOK JS and TANNENBAUM SR. 1982. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem 126: 131-138.). The absorbance was determined in an ELISA reader ((Titertek Multiscan, INC BIOMEDICALS INC - EUA) with a 550-nm filter against blanks with culture medium and Griess reagent (v/v). The results are expressed as micromoles of NO2, based on a standard curve with known concentrations of sodium nitrate in H2O (5 - 60 mM of NO2-).
GENE EXPRESSION OF THE TRANSCRIPTION FACTOR NF-kB BY QUANTITATIVE REVERSE TRANSCRIPTASE-POLYMERASE CHAIN REACTION (RT-PCR)
Spleen cell culture was performed as described in section 2.7 with the EMSA at 100 μg/ml. After 72 hours of culture, RNA was extracted and cDNA was synthesized to evaluate the expression of the transcription factor NF-kB by real-time PCR using the SYBR Green QuantStudio platform (Applied BiosystemsAPPLIED BIOSYSTEMS. 1997. User Bulletin. Applied Biosystems Boston, MA.).
RNA extraction and cDNA synthesis
RNA was isolated from spleen cell culture with the RNeasy Mini Kit (Qiagen, Hamburg, Germany) according to the manufacturer’s protocol. Subsequently, 0.3 μg of total RNA of each sample was reverse transcribed into cDNA using 200 U of Superscript III Reverse Transcriptase (Life Technologies) and 500 ng of Oligo(dT) (Life Technologies) according to the manufacturer’s instructions. Sterilized and filtered DEPC-treated water was used in all cDNA synthesis reactions.
Reverse transcription quantitative PCR (RT-qPCR)
The PCR primers that detected NF-kB and β-actin were designed based on the sequences reported in GenBank with Primer Express software (Applied Biosystems). The parameters chosen were no or low secondary structures or primer-primer interactions, and high specificity was validated by BLAST in NCBI, as described in Table I. Both primers targeted different exons to eliminate potentially contaminating genome DNA. The cDNA samples derived from the investigated genes were detected using a QuantStudio 12K Sequence Detection System (Applied Biosystems) according to the recommendations of the manufacturer. Each RT-PCR used a 10-ng cDNA sample in 10 μl of SYBR-PCR MasterMix 2X (Applied Biosystems), 1 μl of the respective primer mix (NF-kB: Forward 100 nM and Reverse 100 nM; β-actin: Forward 500 nM; Reverse 250 nM), and 20 μl purified and deionized H2O. Relative quantification was performed using the comparative threshold cycle (ΔΔCT) method as previously described (Applied Biosystems 1997).
Primers design. The table shows the primer sequences, amplicon length and the annealing temperature.
Before using the ΔΔCT method for quantification, a validation experiment was performed to verify that the efficiencies of the target and control were approximately equal. The amplification of all samples exhibited the same efficiency for the precise quantification of real-time PCR (RT-PCR) data. Serial fivefold dilutions starting with 100 ng of cDNA from the control group were used. The mean CT values, measured in duplicate, versus the log10 of the dilution were plotted. The values from the linear regressions applied to these plots were also presented (not shown). The amplification efficiencies (E = 10(−1/slope)) were close to 1.0 (100%).
STATISTICAL ANALYSIS
Analysis of variance (ANOVA) and Tukey’s test (for normally distributed data) were used to determine statistical significance among experimental groups. Values of p≤ 0.05 were considered statistically significant. Each experiment was repeated at least three times.
RESULTS
UA QUANTIFICATION AND CHARACTERIZATION OF THE EMSA
Figure 1 shows the chromatograms of the EMSA (Figure 1a) and a UA solution (Figure 1b). The estimated percentage of UA in EMSA was 4.47%, indicating that 1 g of methanol extract contains 44.75 mg of UA.
Chromatogram obtained by HPLC-DAD for the UA standard (a); chromatogram of the methanol extract of the leaves of S. australis Cham. & Schltdl obtained by HPLC-DAD (b). Wavelength was 203 nm. Retention time was 9.94 min for UA.
CELL VIABILITY
None of the EMSA concentrations tested were toxic in spleen cell culture, as shown in Figure 2.
Evaluation of the cytotoxic effect of the methanol extracts of the leaves of Sambucus australis Cham. & Schltdl (EMSA). Evaluation of EMSA cytotoxicity in a splenocyte culture from BALB/c mice for 72 hours at 37ºC and 5% CO2 by the MTT-tetrazolium method. *p<0.05 vs. control. ANOVA and Tukey’s tests.
EMSA REDUCES IL-4, IL-5 AND IFN- у LEVELS in vitro
To analyze the possible mechanisms related to the effects of EMSA, we determined the cytokine profiles of Th2 cells, including IL-4 and IL-5, and Th1 cells, including IFN-у, from the supernatants of spleen cell cultures stimulated with PWM. Stimulation with PWM led to an increase in cytokine production of IL-4 (p<0.001), IL-5 (p<0.001) and IFN-y (p<0.001) compared to the negative control group. When EMSA was added to the cultures upon PWM stimulation at various concentrations, significant reductions in the levels of IL-4 (12.5, 25, 50 and 100 μg/ml, p<0.001) and IL-5 (12.5, 25, 50 and 100 μg/ml, p<0.001) were observed, as shown in Figure 3a and b, respectively. Alternatively, only 100 μg/ml of EMSA significantly reduced IFN-у levels (p<0.001) compared to the positive control (Figure 3c).
(a) Effect of EMSA on IL-4 production in culture stimulated with PWM. n = 6 (p ### <0.001 vs. Control; *** p<0.001 vs. PWM). ANOVA and Tukey’s tests. (b) Effect of EMSA on IL-5 production in culture stimulated with PWM. n = 6 (p ### <0.001 vs. Control; *** p<0.001 vs. PWM). ANOVA and Tukey’s tests. (c) Effect of methanol extract of EMSA on the production of IFN-у in culture stimulated with PWM. n = 6 (p ### <0.001 vs. Control; *** p<0.001 vs. PWM). ANOVA and Tukey’s tests. (d) Effect of EMSA on the production of IL-10 in culture stimulated with PWM. n = 6 (p ### <0.001 vs. Control. ** p<0.01 vs. PWM). ANOVA and Tukey’s tests.
METHANOLIC EXTRACT OF Sambucus australis Cham. & Schltdl INCREASES IL-10 LEVELS in vitro
We evaluated the effect of EMSA on regulatory cytokine IL-10. The data showed that stimulation with PWM led to increased production of IL-10 (p<0.001) compared to the negative control. Treatment with 100 μg/ml of EMSA significantly increased IL-10 production (p<0.01) compared to the positive control, but it was not significant for the other tested concentrations (12.5, 25 and 50 μg/ml) (Figure 3d).
EFFECT OF EMSA ON THE STABILITY OF ERYTHROCYTE MEMBRANES
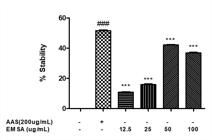
The membrane-stabilizing effect of S. australis Cham. & Schltdl was evaluated. The results showed a significant stabilizing activity of the standard drug, acetylsalicylic acid, compared to the control, by approximately 50%. Comparing the effect of different concentrations of EMSA, the stabilizing membrane potential was distinguished from the standard at all concentrations (12.5, 25, 50 and 100 μg/ml) tested, with estimated percentages of 38% (p<0.001), 42% (p<0.001), 16% (p<0.001) and 11% (p<0.001), respectively (Figure 4). Concentrations of 100 μg/ml and 50 μg/ml had the strongest effect on stabilizing the membrane.
Effect of EMSA on the percentage of membrane stabilization of BALB/c mouse erythrocytes compared to the control (100% hemolysate); hemolysis assay of cells treated or not treated with EMSA at 100, 50, 25 and 12.5 g/ml or standard acetylsalicylic acid (ASA) at 200 μg/ml (### p<0.001 vs. Control; *** p<0.001 vs. ASA). ANOVA and Tukey’s tests.
EFFECT OF EMSA ON THE PRODUCTION OF NO
Stimulation with LPS (5 μg/ml) significantly increased NO levels (p<0.001) compared to the negative control. Treatment with different concentrations of EMSA significantly decreased NO production in vitro (12.5, 25, 50 and 100 μg/ml, p<0.001) (Figure 5a).
(a) Effect of EMSA in NO production in LPSstimulated macrophages. n = 6 (p ### <0.001 vs Control; *** p<0.001 vs. LPS). ANOVA and Tukey’s tests. (b) Effect of EMSA on the expression of NF-kB in culture stimulated with PWM. n = 6, (### p<0.001 vs. Control; ** p<0.01 vs. PWM) ANOVA, Tukey. ### p<0.001 vs. Control; ***p<0.001 vs. PWM). ANOVA and Tukey’s tests.
EFFECT OF EMSA ON THE EXPRESSION OF THE TRANSCRIPTION FACTOR NF-kB
NF-kB activation is involved in the development of inflammation; thus, the effect of EMSA on the expression of this factor was determined by RT-PCR. PWM stimulation in spleen cell culture increased the expression of the transcription factor NF-kB (p<0.001) compared to the negative control. Treatment with the highest concentration of EMSA significantly decreased the expression of NF-kB (100 μg/ml, p<0.01) compared to the positive control stimulated with PWM (Figure 5b). At 100 μg/mL, EMSA was able to modulate the previously evaluated parameters.
DISCUSSION
This study aimed to explore the anti-inflammatory potential of the standardized methanolic extract of S. australis, in vitro. EMSA was estimated to have around 4.47% of ursolic acid by using high-performance liquid chromatography. Previous reports have described that S. australis is rich in triterpenes such as ursolic acid (UA) (Lamaison 1991, Alice 1990). Several biological activities were attributed to UA such as antitumor, antioxidant, antibacterial, hypoglycemic and anti-inflammatory activities by inhibiting the activation of NF-kB (Rao et al. 2011, Alqahtani et al. 2013, Kim and Moon 2015, Chun et al. 2014, Zhang et al. 2014, Yoon et al. 2014, Ma et al. 2014, Zhao et al. 2012, Nascimento et al. 2014).
Here in, we have demonstrated the effect of S. australis Cham. & Schltdl on inflammatory cytokines and a regulatory cytokine in vitro, showing that EMSA can significantly reduce levels of Th2 cytokines, including IL-4, and IL-5, in a concentration-dependent manner (Figure 3a and b) and increase levels of the regulatory cytokine IL-10 (Figure 3d) at different concentrations. Thus, we hypothesize that S. australis Cham. & Schltdl plays a protective role in the inflammatory process because it can reduce inflammatory cytokines and modulate IL-10 production. In contrast, there was no reduction of IFN-y (Figure 3c) at any of the concentrations tested, unlike IL-4 and IL-5. In contrast to our results, other Sambucus species have been shown to participate in the suppression of inflammatory processes by increasing IL-10 levels and decreasing the secretion of inflammatory cytokines, such as IL-12 (SoccaSOCCA EAR and BRITO ARMS. 2010. Effects of Sambucus nigra L. monomeric anthocyanins on TNBS induced ulcerative colitis. Dissertação de Mestrado. (Unpublished data) 2010).
Other inflammatory mediators are released during the early inflammatory process, such as NO, which is produced by activated macrophages that stimulate the production of the inflammatory cytokines previously described. This accounts for the high levels of radicals observed in inflammation. NO may also be released in acute inflammation sites and modular edema (Ialenti et al. 1992IALENTI A, IANARO A, MONCADA S and DI ROSA M. 1992. Modulation of acute inflammation by endogenous nitric oxid. Eur J Pharmacol 211: 177-182.). Therefore, we evaluated the modulatory effect of S. australis Cham. & Schltdl on the production of NO by a standard culture model of macrophages stimulated with LPS (Chandrasekaran et al. 2010CHANDRASEKARAN CV, GUPTA A and AGARWAL A. 2010. Effect of an extract of Andrographis paniculata leaves on inflammatory and allergic mediators in vitro. J Ethnopharmacol 129: 203-207.). Our results suggest that the S. australis Cham. & Schltdl-mediated decrease in NO production may be due at least in part to the reduced levels of inflammatory cytokines. These data support the antioxidant activity of S. australis, which has been described in other species of Sambucus (Kim and Moon 2015, Nascimento et al. 2014, Dawidowicz et al. 2006DAWIDOWICZA AL, WIANOWSKAA D and BARANIAKB B. 2006. The antioxidant properties of alcoholic extracts from Sambucus nigra L. (antioxidant properties of extracts). LWT Food Sci Technol 39: 308-315.).
The release of chemical mediators in injured tissues and migrating cells cause disturbances in the cell membrane, resulting in the activation of lysosomal enzymes with potent cytotoxic activity, which can destroy neighboring cells and are responsible for various symptoms and signs of inflammation. Anti-inflammatory nonsteroidal drugs can inhibit the disruption of the membrane, blocking the release of inflammatory mediators (Amann and Peskar 2002AMANN R and PESKAR BA. 2002. Anti-inflammatory effects of aspirin and sodium salicylate. Eur JPharmacol 447: 1-9., Mounnissamy et al. 2007MOUNNISSAMY VM, KAVIMANI S, BALU V and QUINE SD. 2007. Evaluation of anti-inflammatory and membrane stabilizing properties of ethanol extract of Cansjera rheedii J.Gmelin (Opiliaceae). IJPT 6: 235-237.).
Therefore, we used a membrane stabilization assay of erythrocytes to analyze the anti-inflammatory activity of S. australis in vitro in this context, as the membrane of the erythrocytes is analogous to the lysosomal membrane, and anti-inflammatory drugs can act by stabilizing the lysosomal membrane (Leelaprakash and Dass 2011, Okoye and Osadebe 2010, Yoganandam et al. 2010). Different concentrations of EMSA (12.5, 25, 50, and 100 μg/ml) inhibited the disruption of erythrocyte membranes in a concentration-dependent manner when compared to a classical standard drug. This behavior demonstrates the potential capacity of S. australis in protecting the membrane of lysosomes and preventing the release of toxic enzymes during inflammation and activation of other mediators, further demonstrating a potential anti-inflammatory effect.
To confirm the possible mechanism whereby S. australis Cham. & Schltdl has anti-inflammatory activity, we analyzed its effect on the transcription factor NF-kB, which regulates the transcription of a number of proinflammatory factors, such as cytokines and inflammatory mediators (Fröde-Saleh and Calixto 2000FRÖDE-SALEHTS and CALIXTO JB. 2000. Synergistic antiinflammatory effect of NF-kappaB inhibitors and steroidal or non steroidal antiinflammatory drugs in the pleural inflammation induced by carrageenan in mice. Inflamm Res 49: 330-337.). Corticosteroids are NF-kB inhibitors because they bind to specific receptors for GCs, forming complexes that act as second messengers that regulate/activate the expression of genes involved in the immune response. These complexes have high binding affinity with DNA and enter the nucleus and bind to specific gene promoters, causing gene transcription. Thus, one of the pathways modulated by steroids is the activation of IκB transcription. IκBB maintains the inactive NF-kB in the cytoplasm by preventing the translocation of this factor to the nucleus (D’acquisto 2002, Barnes and Karin 1997BARNES PJ and KARIN M. 1997. Nuclear factor-kappaB: a pivotal transcription factor in chronic inflammatory diseases. N Engl J Med 336: 1066-1071.). Therefore, the inhibition of NF-kB can modulate inflammatory resolution by inhibiting the secretion of inflammatory cytokines and factors (Vendramini-Costa and Carvalho 2012VENDRAMINI-COSTA DB and CARVALHO JE. 2012. Molecular Link Mechanisms between Inflammation and Cancer. Curr Pharm Des 18: 3831-3852.).
In this context, EMSA statistically decreased NF-kB expression, explaining the reduction in levels of inflammatory cytokines and NO when different concentrations of EMSA in assays were tested. The EMSA mechanism of action on NF-kB may be associated with the presence of UA in S. australis Cham. & Schltdl, confirming recent reports that showed UA, the major component in S. australis, possesses anti-inflammatory activity via the inactivation of NF-kB (Kim and Moon 2015, Jiang 2015, Ma et al 2014). Still, further studies should be conducted to elucidate the mechanisms of the inhibition of this pathway by S. australis Cham. & Schltdl.
CONCLUSIONS
We suggest that S. australis Cham. & Schltdl has anti-inflammatory potential by inhibiting the activation of the transcription factor NF-kB and consequently by reducing levels of inflammatory cytokines and NO, which justifies its use in popular medicine for the treatment of inflammation. Further in vivo studies are needed to better characterize the underlying mechanisms of EMSA.
ACKNOWLEGMENTS
The authors want to thank the Brazilian agencies Fundação de Amparo a Pesquisa do Estado da Bahia (FAPESB) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for financial support and for Carneiro NVQ’s scholarship, respectively. NMAN and CAV are Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) scholarship recipients.
REFERENCES
- ALICE CB, SIQUEIRA NCS, HALBIG JC, MENTZ LA, SILVA GAA and GOETTEMSEI. 1990. Diagnose comparativa de compostos polifenólicos de Sambucus nigra L. e Sambucus australis Cham. & Schldht. Rev Bras Farm 71: 88-90.
- ALQAHTANI A, HAMID K, KAM A, WONG KH, ABDELHAK Z, RAZMOVSKI-NAUMOVSKI V, CHAN K, LI KM, GROUNDWATER PW and LI GQ. 2013. The pentacyclic triterpenoids in herbal medicines and their pharmacological activities in diabetes and diabetic complications. Curr Med Chem 20: 908-931.
- AMANN R and PESKAR BA. 2002. Anti-inflammatory effects of aspirin and sodium salicylate. Eur JPharmacol 447: 1-9.
- APPLIED BIOSYSTEMS. 1997. User Bulletin. Applied Biosystems Boston, MA.
- ARIEL A and SERHAN CN. 2012. New Lives Given by Cell Death: Macrophage Differentiation Following Their Encounter with Apoptotic Leukocytes during the Resolution of Inflammation. Front Immunol 31: 3-4.
- BACIGALUPO NM. 1974. Sambucaceae. In: Burkart A (Ed), Flora Ilustrada de Entre Rios (Argentina), Caprifoliaceae. INTA 1974: 50-52.
- BADESCU L, BADULESCU O, BADESCU M and CIOCOIU M. 2012. Mechanism by Sambucus nigra Extract Improves Bone Mineral Density in Experimental Diabetes.Evid. Based Complement Alternat Med, 6 p.
- BARNES PJ and KARIN M. 1997. Nuclear factor-kappaB: a pivotal transcription factor in chronic inflammatory diseases. N Engl J Med 336: 1066-1071.
- CHANDRASEKARAN CV, GUPTA A and AGARWAL A. 2010. Effect of an extract of Andrographis paniculata leaves on inflammatory and allergic mediators in vitro. J Ethnopharmacol 129: 203-207.
- CHUN J, LEE C, HWANG SW, IM JP AND KIM JS. 2014. Ursolic acid inhibits nuclear factor-κB signaling in intestinal epithelial cells and macrophages, and attenuates experimental colitis in mice. Life Sci 110: 23-34.
- DAWIDOWICZA AL, WIANOWSKAA D and BARANIAKB B. 2006. The antioxidant properties of alcoholic extracts from Sambucus nigra L. (antioxidant properties of extracts). LWT Food Sci Technol 39: 308-315.
- FRÖDE-SALEHTS and CALIXTO JB. 2000. Synergistic antiinflammatory effect of NF-kappaB inhibitors and steroidal or non steroidal antiinflammatory drugs in the pleural inflammation induced by carrageenan in mice. Inflamm Res 49: 330-337.
- GOVINDAPPA M, NAGA SRAVYA S, POOJASHRI MN, SADANANDA TS, CHANDRAPPA CP, SANTOYO G, SHARANAPPA P and ANIL KUMAR NV. 2011. Antimicrobial, antioxidant and in vitro anti-inflammatory activity and phytochemical screening of water extract of Wedelia trilobata (L.) Hitchc. J Med Plants Res 5: 5718-5729.
- GREEN LC, WAGNER DA, GLOGOWSKI J, SKIPPER PL, WISHNOK JS and TANNENBAUM SR. 1982. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem 126: 131-138.
- HANADA T and YOSHIMURA A. 2002. Regulation of cytokine signaling and inflammation. Cytokine Growth Factor Rev 13: 413-421.
- IALENTI A, IANARO A, MONCADA S and DI ROSA M. 1992. Modulation of acute inflammation by endogenous nitric oxid. Eur J Pharmacol 211: 177-182.
- JUDD WS, CAMPBELL CS, KELLOG EA and STEVENS PF. 1999. Plant Systematics: a Phylogenetic Approach. Massachusetts: Sinauer Associates.
- KIM ES and MOON A. 2015. Ursolic acid inhibits the invasive phenotype of SNU-484 human gastric cancer cells. Oncol Lett 9: 897-902.
- KIM HP, SON KH, CHANG HW and KANG SS. 2004. Anti-inflammatory plant flavonoids and cellular action mechanisms. J Pharmacol Sci 96: 229-245.
- LAMAISON JL, PETIT JEAN-FREYTET C and CARNAT A. 1991. Présence de 3-glucoside et de 3-rutinoside dʼisorhamnétine dans les fleurs de Sambucus nigra L. Ann Pharm Fr 49: 258-262.
- LIOBIKAS J, MAJIENE D, TRUMBECKAITE S, KURSVIETIENE L, MASTEIKOVA R, KOPUSTINSKIENE DM, SAVICKAS A and BERNATONIENE J. 2011. Uncoupling and antioxidante effects of ursolic acid in isolated rat heart mitochondria. J Nat Prod 74: 1640-1644.
- MA JQ, DING J, ZHANG L AND LIU CM. 2014. Ursolic acid protects mouse liver against CCl4-induced oxidative stress and inflammation by the MAPK/NF-κB pathway. Environ Toxicol Pharmacol 37: 975-983.
- MOSMANN T. 1983. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65(1-2): 55-63.
- MOUNNISSAMY VM, KAVIMANI S, BALU V and QUINE SD. 2007. Evaluation of anti-inflammatory and membrane stabilizing properties of ethanol extract of Cansjera rheedii J.Gmelin (Opiliaceae). IJPT 6: 235-237.
- NASCIMENTO PG, LEMOS TL, BIZERRA AM, ARRIAGA ÂM, FERREIRA DA, SANTIAGO GM, BRAZ-FILHO R AND COSTA JG. 2014. Antibacterial and antioxidant activities of ursolic acid and derivatives. Molecules 19: 1317-1327.
- NORLING LV and SERHAN CN. 2010. Profiling in resolving inflammatory exudates identifies novel anti-inflammatory and pro-resolving mediators and signals for termination. J Intern Med 268: 15-24.
- RAO VS, DE MELO CL, QUEIROZ MGR, LEMOS TLG, MENEZES DB, MELO TS and SANTOS FA. 2011. Ursolic Acid, a pentacyclic triterpene from Sambucus australis, prevents abdominal adiposity in mice fed a high-fat diet. J Med Food 14: 1375-1382.
- SCOPEL M, NUNES E, VIGNOLI-SILVA M, VENDRUSCOLO GS, HENRIQUES AT and MENTZ LA. 2007. Caracterização farmacobotânica das espécies de Sambucus (Caprifoliaceae) utilizadas como medicinais no Brasil. Parte I. Sambucus nigra L. Rev Bras Farmacogn 17: 249-261.
- SERHAN CN, WARD PA and GILROY DW. 2010. Fundamentals of Inflammation. Cambridge University Press, EUA, 473 p.
- SERHAN CN, BRAIN SD, BUCKLEY CD, GILROY DW, HASLETT C, O’NEILL LA, PERRETTI M, ROSSI AG and WALLACE JL. 2007. Resolution of inflammation: state of the art, definitions and terms. FASEB J 21: 325-332.
- SHINDE UA, PHADKE AS, NARI AM, MUNGANTIWAR AA, DIKSHIT VJ and SARAF MN. 1999. Membrane stabilization activity - a possible mechanism of action for the anti-inflammatory activity of Cedrus deodara wood oil. Fitoterapia 70: 251-257.
- SOCCA EAR and BRITO ARMS. 2010. Effects of Sambucus nigra L. monomeric anthocyanins on TNBS induced ulcerative colitis. Dissertação de Mestrado. (Unpublished data)
- STAHN C, LÖWENBERG M, HOMMES DW and BUTTGEREIT F. 2007. Molecular mechanisms of glucocorticoid action and selective glucocorticoid receptor agonists. Mol Cell Endocrinol 275: 71-78.
- VENDRAMINI-COSTA DB and CARVALHO JE. 2012. Molecular Link Mechanisms between Inflammation and Cancer. Curr Pharm Des 18: 3831-3852.
- YOON JH, YOUN K, HOCT, KARWE MV, JEONG WS AND JUN MP. 2014. Coumaric acid and ursolic acid from Corni fructus attenuated β-amyloid(25-35)-induced toxicity through regulation of the NF-κB signaling pathway in PC12 cells. J Agric Food Chem 62: 4911-4916.
- ZHANG JF, GAO L, DUAN HQ, WANG W, LI Y and MA YG. 2014. Mechanism of antitumor effect of ursolic acid on T (8;21) leukemia cell kasumi-1. Zhongguo Shi Yan Xue Ye Xue Za Zhi 22: 687-691.
- ZHAO J, CHEN J, LIU T, FANG J, WAN J, ZHAO J, LI W, LIU J, ZHAO X and CHEN S. 2012. Anti-viral effects of urosolic acid on guinea pig cytomegalovirus in vitro. J Huazhong Univ Sci Technolog Med Sci 32: 883-887.
Publication Dates
-
Publication in this collection
21 Mar 2019 -
Date of issue
2019
History
-
Received
17 Oct 2017 -
Accepted
17 Apr 2018