Abstract
Abstract: The first pterosaur bone bed from Brazil was reported in 2014 at the outskirts of the town Cruzeiro do Oeste, Paraná State, in the Southern region of the country. Here named 'cemitério dos pterossauros' site, these outcrops were referred to the Goio-Erê Formation (Turonian-Campanian) of the Caiuá Group (Bauru Basin) and revealed the presence of hundreds of isolated or partially articulated elements of the tapejarine pterosaur Caiuajara and fewer amounts of a theropod dinosaur. Here we present a new tapejaromorph flying reptile from this site, Keresdrakon vilsoni gen. et sp. nov., which shows a unique blunt ridge on the dorsal surface of the posterior end of the dentary. Morphological and osteohistological features indicate that all recovered individuals represent late juveniles or sub-adults. This site shows the first direct evidence of sympatry in Pterosauria. The two distinct flying reptiles coexisted with a theropod dinosaur, providing a rare glimpse of a paleobiological community from a Cretaceous desert.
Key words
Paleoecology; Pterosauria; Pterodactyloidea; Keresdrakon vilsoni; Paraná; Cretaceous
INTRODUCTION
Pterosaurs are the first vertebrates to conquer the air by developing powered flight (e.g., KellnerKELLNER AWA. 2006. Pterossauros: os senhores dos ceús do Brasil. Rio de Janeiro: Vieira & Lent, 175 p. 2006). Despite having been recovered in all continents (HammerHAMMER WR and HICKERSON WJ. 1994. A crested theropod dinosaur from Antarctica. Science 264: 828-830. and Hickerson 1994, BarrettBARRETT PM, BUTLER RJ, EDWARDS NP and MILNER AR. 2008. Pterosaur distribution in time and space: an atlas. Zitteliana B 28: 61-108. et al. 2008), their fossil record can be considered as patchy, with only few deposits having presented complete or nearly complete skeletons (e.g., EatonEATON GF. 1910. Osteology of Pteranodon. Memoirs of the Connecticut Academy of Arts and Sciences 2: 1-38. 1910, TischlingerTISCHLINGER H and FREY E. 2002. Ein Rhamphorhynchus (Pterosauria, Reptilia) mit ungewöhnlicher Flughauterhaltung aus dem Solnhofener Plattenkalk. Archaeopteryx 20: 1-20. and Frey 2002, Barrett et al. 2008, BantimBANTIM RAM, SARAIVA AAF, OLIVEIRA G and SAYÃO JM. 2014. A new toothed pterosaur (Pterodactyloidea: Anhangueridae) from the Early Cretaceous Romualdo Formation, NE Brazil. Zootaxa 3869: 201-223. et al. 2014, Kellner 2017). Even in the stratigraphic units that have yielded large quantities of these volant archosaurs, there is a general lack of pterosaur bone beds (e.g., WellnhoferWELLNHOFER P. 1975. Die Rhamphorhynchoidea (Pterosauria) der Oberjura-Plattenkalke Süddeutschlands. Teil I: Allgemaine Skelettmorphologie. Palaeontogr Abt A 148: 1-33. 1975, WildWILD R. 1978. Die Flugsaurier (Reptilia, Pterosauria) aus der Oberen Trias von Cene bei Bergamo, Italien. Boll Soc Paleont It 17(2): 176-256. 1978, SaraivaSARAIVA AAF, HESSEL MH, GUERRA NC and FARA E. 2008. Concreções calcárias da Formação Santana, Bacia do Araripe: uma proposta de classificação. Estudos Geológicos 17(1): 40-57. et al. 2008, DallaDALLA VECCHIA FM. 2014. Gli pterosauri triassici. Udine: Pubblicazione del Museo Friulano di Storia Naturale, 319 p. Vecchia 2014, ChengCHENG X, JIANG X, WANG X and KELLNER AWA. 2017. Premaxillary crest variation withing the Wukongopteridae (Reptilia, Pterosauria) and comments on cranial structures in pterosaurs. An Acad Bras Cienc 89: 119-130. et al. 2017, PinheiroPINHEIRO FL and RODRIGUES T. 2017. Anhanguera taxonomy revisited: is our understanding of Santana Group pterosaur diversity biased by poor biological and stratigraphic control? PeerJ 5: e3285. and Rodrigues 2017), so far restricted to only three occurrences: the Lagarcito Formation (Lower Cretaceous, Albian) in San Luis, Argentina (YrigoyenYRIGOYEN M. 1975. La edad Cretácica del Grupo del Gigante (San Luis) y su relación con cuencas circunvecinas. Actas Congreso Geológico Argentino de Paleontología y Bioestratigrafía 2: 29-56. 1975, ChiappeCHIAPPE LM, RIVAROLA D, ROMERO E, DAVILA S and CODORNIÚ L. 1998. Recent advances in the paleontology of the Lower Cretaceous Lagarcito Formation (Parque Nacional Sierra de Las Quijadas, San Luis, Argentina). In: Lucas SG, Kirkland JI and Estep JW (Eds), Lower and Middle Cretaceous Terrestrial Ecosystems, Albuquerque: New Mexico Museum of Natural History and Science, p. 187-192. et al. 1998), the Lower Cretaceous deposits of the Tugulu Group in Hami, Xinjiang, China (EberthEBERTH DA, BRINKMAN DB, CHEN PJ, YUAN FT, WU SZ, LI G and CHENG XS. 2001. Sequence stratigraphy, paleoclimate patterns, and vertebrate fossil preservation in Jurassic-Cretaceous strata of the Junggar Basin, Xinjiang Autonomous Region, People’s Republic of China. Can J Earth Sci 38: 1627-1644. et al. 2001, WangWANG XL et al. 2014. Sexually dimorphic tridimensionally preserved pterosaurs and their eggs from China. Curr Biol 24: 1323-1330. et al. 2014), and the strata of the Upper Cretaceous Goio-Erê Formation (Bauru Group) that outcrop at Cruzeiro do Oeste, Paraná State, Brazil (BasiliciBASILICI G, SGARBI GN and DAL'BÓ PFF. 2012. A Sub-bacia Bauru: um sistema continental entre deserto e cerrado. In: Hasui Y, Carneiro CDR, Almeida FFM and Bartorelli A (Eds), Geologia do Brasil, São Paulo: Beca, p. 520-543. et al. 2012, ManzigMANZIG PC, KELLNER AWA, WEINSCHÜTZ LC, FRAGOSO CE, VEGA CS, GUIMARÃES GB, GODOY LC, LICCARDO A, RICETTI JHC and MOURA CC. 2014. Discovery of a rare pterosaur bone bed in a Cretaceous Desert with insights on ontogeny and behavior of flying reptiles. PloS One 9(8): e100005. et al. 2014). The latter is the first occurrence of pterosaurs in the Bauru Group that has yielded a comparatively large number of other fossil vertebrates such as dinosaurs (e.g., BrusatteBRUSATTE SL, CANDEIRO CRA and SIMBRAS FM. 2017. The last dinosaurs of Brazil: The Bauru Group and its implication for the end-Cretaceous mass extinction. An Acad Bras Cienc 89: 1465-1485. et al. 2017, BandeiraBANDEIRA KLN, BRUM AS, PÊGAS RV, CIDADE GM, HOLGADO B, CIDADE A and SOUZA RG. 2018. The Baurusuchidae vs Theropoda record in the Bauru Group (Upper Cretaceous, Brazil): a taphonomic perspective. J Iber Geol 44: 25-54. et al. 2018) and crocodylomorphs (e.g., RiffRIFF D, SOUZA RG, CIDADE GM, MARTINELI AG and SOUZA FILHO JP. 2012. Crocodilomorfos: a maior diversidade de répteis fósseis do Brasil. Terrae 9: 12-40. et al. 2012).
The outcrops at the region of Cruzeiro do Oeste have a peculiar history (Fig. 1). The site, here named 'cemitério dos pterossauros ' (pterosaur graveyard), was discovered some 40 years ago by the Dobruski family. No fieldwork was carried out in the area over these years until it was rediscovered in 2011, when some specimens that had been eroding on the surface were collected without fine stratigraphic control (see Manzig and WeinschützMANZIG PC and WEINSCHÜTZ LC. 2012. Museus e fósseis da Região Sul do Brasil: uma experiência visual com a paleontologia. Curitiba: Germânica, 306 p. 2012). Later (October/ 2012) one of the authors (AWAK) was invited by LCW and Paulo C. Manzig (at that time associated with CENPALEO - Centro Paleontológico of the Universidade do Contestado) to examine this material and recognized the presence of at least two pterosaur taxa and one theropod dinosaur. He has encouraged CENPALEO to organize fieldwork in the region, which was carried out in cooperation with the Museu Nacional/UFRJ during July/2012, January-February/2013, July/2013, and July/2014. This activity resulted in thousands of fossil bones, mostly found isolated that were split between CENPALEO and the Municipality of Cruzeiro do Oeste by a written agreement signed some years later. Some blocks (still unprepared) are housed in the Museu Nacional and were not affected by the recent fire that has consumed the main building of the institution (e.g., ZamudioZAMUDIO KR et al. 2018. Lack of science support fails Brazil. Science 361: 1322-1323. et al. 2018). Training for the preparation of the material was performed by the Museu Nacional, first at CENPALEO (July/2012) and then at Cruzeiro do Oeste (August/2016).
The more numerous bones recovered so far belong to a small tapejarine pterosaur that was named Caiuajara dobruskii Manzig et al. 2014. The previously identified dinosaur was recently named Vespersaurus paranaensisLangerLANGER MC et al. 2019. A new desert-dwelling dinosaur (Theropoda, Noasaurinae) from the Cretaceous of south Brazil. Sci Rep 9: 9379. et al. 2019, also known by hundreds of isolated elements. An acrodont lizard, Gueragama sulamericanaSimõesSIMÕES TR, WILNER E, CALDWELL MW, WEINSCHÜTZ LC and KELLNER AWA. 2015. A stem acrodontan lizard in the Cretaceous of Brazil, revises early lizard evolution in Gondwana. Nat Commun 6: 8149. et al. 2015, which was described based on fragmentary material (Simões et al. 2015), complete the known reptilian fauna from this site. Lizard remains tend to be quite incomplete (e.g., Chavarría-ArellanoCHAVARRÍA-ARELLANO ML, SIMÕES TR and MONTELLANO-BALLESTEROS MM. 2018. New data on the Late Cretaceous lizard Dicothodon bajaensis (Squamata, Borioteiioidea) from Baja California, Mexico reveals an unusual tooth replacement pattern in squamates. An Acad Bras Cienc 90: 2781-2795. et al. 2018), for all those representing ancient desertic environments.
Map showing the location of the 'cemitério dos pterossauros' site in Cruzeiro do Oeste (red arrow), Paraná State, southern Brazil. (a) The outcrops of the Bauru Basin (light grey) in Brazil, (b) detail of the Bauru basin with the Rio Paraná (light green), Goio-Erê (light blue), Santo Anastácio (grey) formations, and the Bauru Group (dark grey), and (c) stratigraphic chart showing the relations of the former, including the underlying Serra Geral Formation (green).
In this study we present the description of the second toothless pterosaur taxon from this deposit, Keresdrakon vilsoni gen. et sp. nov., which is known by far less specimens than Caiuajara and Vespersaurus. We also explore the paleoecological relationships of the known taxa of this Cretaceous desert environment, which has the potential for unveiling many new findings (Kellner 2012KELLNER AWA. 2012. Pterossauros: os primeiros répteis alados. In: Hasui Y, Carneiro CDR, Almeida FFM and Bartorelli A (Eds), Geologia do Brasil, São Paulo: Beca, p. 735-746.).
GEOLOGICAL SETTING
The stratigraphic assignment of the deposits in the outskirts of Cruzeiro do Oeste has been put under dispute recently (Figs. 1-2). The 'cemitério dos pterossauros' site was originally regarded by Manzig et al. (2014) as belonging to the Caiuá Group, more specifically to the Goio-Erê Formation. Langer et al. (2019) agreed with the assignment to the Caiuá Group but argued that the site is located in an area already mapped as the Rio Paraná Formation (FernandesFERNANDES LA. 2004. Mapa litoestratigráfico da parte oriental da Bacia Bauru (PR, SP, MG), escala 1: 1.000.000. Boletim Paranaense de Geociências 55. 2004).
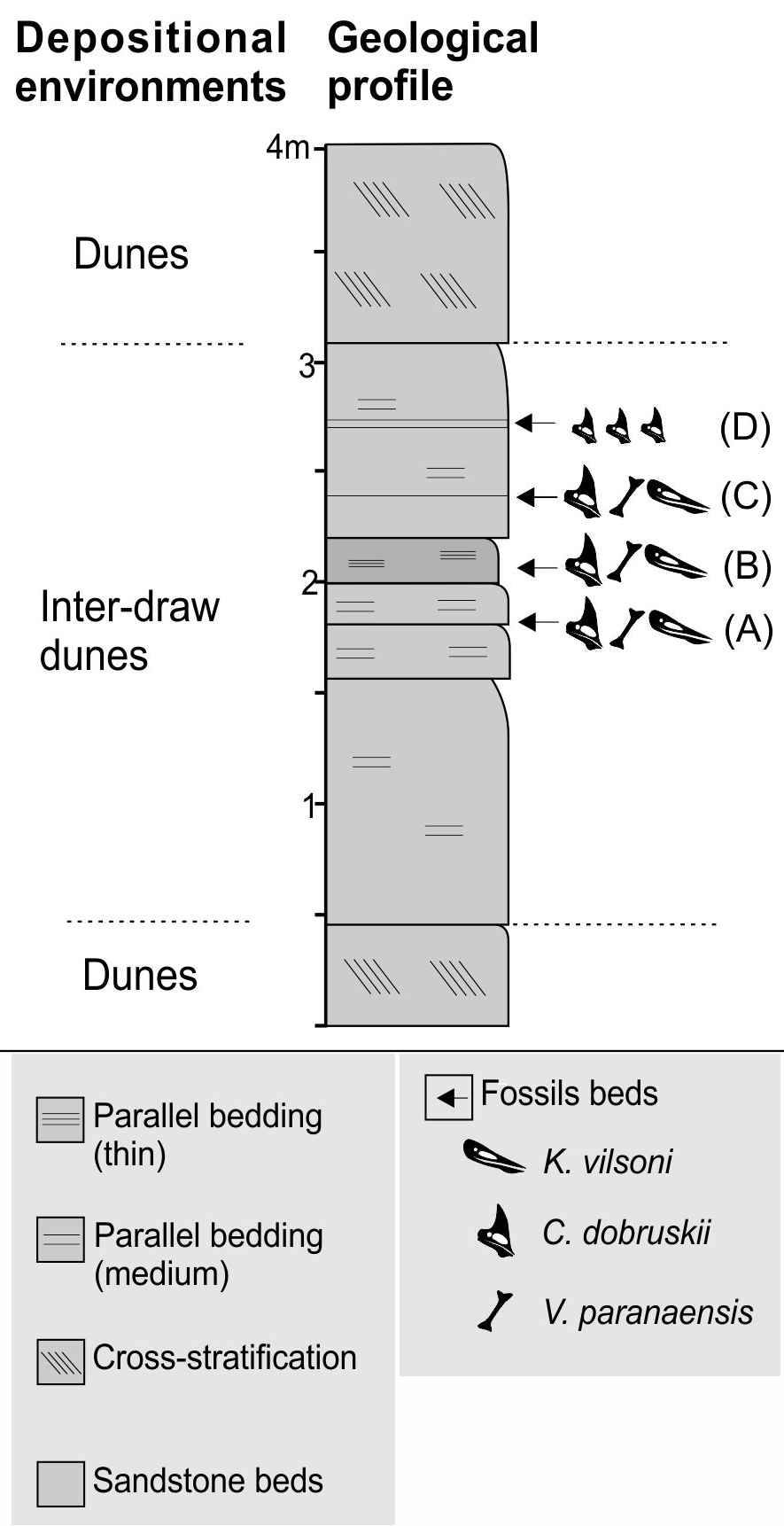
Stratigraphic sequence of the 'cemitério dos pterossauros' site, showing the recorded taxa (Caiuajara dobruskii, Vespersaurus paranaensis and Keresdrakon vilsoni) in the four (A-D) bonebeds, the correspondent depositional environments, and the sedimentary structures. Note that in bonebed D only small individuals of Caiuajara were collected.
The Bauru Basin is formed by the Bauru and the Caiuá groups (FernandesFERNANDES LA and COIMBRA AM. 2000. Revisão estratigráfica da parte oriental da Bacia Bauru (Neocretáceo). Revista Brasileira de Geociências 30: 717-728. and Coimbra 2000) and has its development linked to the isostatic compensation related to the large basaltic lava accumulation of the Eocretaceous Serra Geral magmatic event (Fernandes 2004). BatezelliBATEZELLI A. 2010. Arcabouço tectono-estratigráfico e evolução das Bacias Caiuá e Bauru no Sudeste brasileiro. Revista Brasileira de Geociências 40: 265-285. (2010) showed that the Caiuá Group was formed by a depocenter located in the southernmost part of the basin (in relation to the Bauru Group) directed towards the Paraná State and whose northern limit extended to the southeastern part of the Minas Gerais State. Based on the eolian sedimentary rocks, the paleoclimate at that time of deposition is regarded as arid (Fernandes 2004).
The Caiuá Group is characterized by predominantly desert-type red beds and divided into the Rio Paraná, Goio-Erê and Santo Anastácio formations (Fernandes and Coimbra 2000). All three units are interpreted as chronocorrelated and interdigitated, whose limits are transitional (Fernandes 2004). Their contact with the basalts of the underlying Serra Geral Formation is marked by an unconformity (Fig. 1).
The Rio Paraná Formation is composed of fine to very fine to medium grained sandstones, forming large eolian structures with medium to large cross stratification that, according to Fernandes (2004), correspond to the central region of the “sand sea”. The Goio-Erê Formation consists of fine and very fine (sometimes medium) grained sandstones, rich in quartz and whose color ranges from reddish brown to purplish gray. These rocks are mineralogically and textually supermature. They form tabular layers with cross-stratification, sometimes with flat-parallel lamination, and deposits that correspond to wet interdunes (Fernandes and Coimbra 2000). Lastly, the Santo Anastácio Formation fills shallow troughs of the valleys formed by the Paraná river tributaries. The deposits constitute massive tabular sandstones and correspond to sand sheets present on the extensive desertic marginal plains of the former “sand sea” (Fernandes 2004).
Although the 'cemitério dos pterossauros' site is showed to correspond to the Rio Paraná Formation in the current geologic map encompassing the region of Cruzeiro do Oeste, the lithological characteristics of the outcrop with the bonebeds are more consistent with the description of the deposits of the Goio-Erê Formation. The fossils themselves indicate the presence of humid inter-dunes where life could thrive. Therefore, we interpret the 'cemitério dos pterossauros' site as a non-mappable occurrence of the Goio-Erê Formation. Otherwise, it would indicate the presence of inter-dune deposits within the Rio do Paraná Formation, which contradicts its original interpretation.
The age of those layers is also controversial. Basilici et al. (2012) favor a Turonian - Campanian age, with the lower limits even being considered as Coniacian by MilaniMILANI EJ, MELO JH, SOUZA PA, FERNANDES LA and FRANÇA AB. 2007. Bacia do Paraná. Bol Geoc Petrobras 15: 265-287. et al. (2007), while BatezelliBATEZELLI A. 2015. Continental systems tracts of the Brazilian Cretaceous Bauru Basin and their relationship with the tectonic and climatic evolution of South America. Basin Research 27: 1-25. (2015) argues for a lower Cretaceous age (Aptian-Albian). No definitive conclusion can be reached at the present stage of knowledge, particularly concerning the 'cemitério dos pterossauros' site.
Regarding the bonebeds, they occur on a secondary road floor leading from the center of the town Cruzeiro do Oeste to the rural areas nearby, as well as on the right ravine (direction city - rural areas). As pointed out before (Manzig et al. 2014), there are three distinct lithological units formed by massive, fine to very fine sandstones (Fig. 2). The surface of the grains is opaque and covered with iron oxide. On the top of the sequence there are very fine sandstones. Four bonebeds have been observed, with the lower one bearing many disarticulated remains of pterosaurs (Caiuajara and Keresdrakon) and one theropod dinosaur (Vespersaurus). The same fossil assembly was recovered in the subsequent two other bonebeds, although elements tend to be sparser. The last bonebed consists mainly of hundreds of very small bones attributed to Caiuajara.
MATERIALS AND METHODS
PHYLOGENETIC ANALYSIS
In order to determine the phylogenetic position of Keresdrakon vilsoni gen. et sp. nov., we performed a phylogenetic analysis using the software TNT 1.5 (GoloboffGOLOBOFF PA and CATALANO SA. 2016. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics 32(3): 221-238. and Catalano 2016). This analysis is based on Holgado et al.HOLGADO B, PÊGAS RV, CANUDO JI, FORTUNY J, RODRIGUES T, COMPANY J and KELLNER AWA. 2019. On a new crested pterodactyloid from the Early Cretaceous of the Iberian Peninsula and the radiation of the clade Anhangueria. Sci Rep 9: 4940. (2019 - see data matrix there). We have used all taxa of that study (144) adding only the new species. All characters are discrete and the original coding was maintained. Search for the most parsimonious trees (MPTs) was conducted via Traditional Search (TBR swapping algorithm), 10,000 replicates, random seed, and collapsing trees after search. We also conducted the analysis via New Technology using Sect. Search, Ratchet (parameters: 20 substitutions made, or 99% swapping completed, 6 up-weighting prob., 6 down-weighting prob., and a total number of iterations of 10), Tree fusing, Driven search (15 initial addseqs., 15 times of min. length), random seed, and without collapsing trees after search. Subsequently, the results of the New Technology were analyzed via Traditional Search (TBR swapping algorithm), starting trees for RAM, and without collapsing trees after search.
The coding of Keresdrakon vilsoni in the data matrix published by Holgado et al. (2019) is as follows:
101????????????????00?120000-00?0--00--??0-???????????2????????????000???13200 101?30-00010000--0000?111?00????? 00020011?0?01520??1???????????????.
SLIDE PREPARATION
Two bones of Keresdrakon vilsoni gen. et sp. nov. were sectioned for a preliminary histological analysis (CP.V 2077 and CP.V 2374). All elements were measured, photographed and described for bone microstructure investigation before they were sectioned, according to the methodology proposed by LammLAMM ET. 2013. Bone histology of fossil tetrapods. In: Padian K and Lamm ET (Eds), Preparation and sectioning of specimens, Oakland: University of California Press, p. 55-160. (2013). Bones were sectioned in the diaphysis portion, and a sample of about 1 cm thick from each was obtained for histological slide preparation. These specimens were immersed in clear epoxy resin Resapol T-208 catalyzed with Butanox M50, cut with the aid of a micro rectify (Dremel 4000 with extender cable 225) coupled to a diamond disk. The section assembly side was ground and polished in a metal polishing machine (AROPOL-E, AROTEC LTDA) using AROTEC abrasive grit (grit size 60 / P60, 120 / P120, 320 / P400, 1200 / P2500). Finally, the sections were examined and photographed in a light microscope (Zeiss Inc. Barcelona, Spain) equipped with an AxioCam camera with Axio Imager, after the histological slides were prepared. The M2 imaging software was used in the examination procedure.
NOMENCLATURAL ACTS
This published work and the nomenclatural acts it contains have been registered in ZooBank, the proposed online registration system for the International Code of Zoological Nomenclature. The Zoobank Life Science Identifiers (LSIDs) to the prefix http://zoobank.org/. The LSID for this publication is urn:lsid:zoobank.org:pub:C4E8E040-D158-42C3-A91F-E432CDEAB424, and the LSIDs for the new erected groups and taxa are: urn:lsid:zoobank.org:act:E26A65E0-2859-4CD3-B773-B99D568D366C (Keresdrakon) and urn:lsid:zoobank.org:act:8358C917-6C12-4390-AAB8-37D82723BBCD (Keresdrakon vilsoni).
The following pterosaur genera and species are mentioned in this study: ArgentinadracoKellnerKELLNER AWA and CALVO J. 2017. New azhdarchoid pterosaur (Pterosauria, Pterodactyloidea) with an unusual lower jaw from the Portezuelo Formation (Upper Cretaceous), Neuquén Group, Patagonia, Argentina. An Acad Bras Cienc 89: 2003-2012. and Calvo 2017, CaiCAI Z and WEI F. 1994. On a new pterosaur (Zhejiangopterus linhaiensis gen. et sp. nov.) from Upper Cretaceous in Linhai, Zhejiang, China. Vert Palas 32: 181-194.uajara dobruskii Manzig et al. 2014, CaupedactylusKellner 2013KELLNER AWA. 2013. A new unusual tapejarid (Pterosauria, Pterodactyloidea) from the Early Cretaceous Romualdo Formation, Araripe Basin, Brazil. Earth Env Sci T R So 103: 1-14., EurazhdarchoVremirVREMIR M, KELLNER AWA, NAISH D and DYKE GJ. 2013. A new azhdarchid pterosaur from the Late Cretaceous of the Transylvanian Basin, Romania: implications for azhdarchid diversity and distribution. PloS One 8(1): e54268. et al. 2013, Hamipterus Wang et al. 2014, Pteranodon Marsh 1876, Quetzalcoatlus Lawson 1975, TapejaraKellner 1989KELLNER AWA. 1989. A new edentate pterosaur of the Lower Cretaceous from the Araripe Basin, Northeast Brazil. An Acad Bras Cienc 61: 439-446., ThalassodromeusKellner and Campos 2002KELLNER AWA and CAMPOS DA. 2002. The function of the cranial crest and jaws of a unique pterosaur from the Early Cretaceous of Brazil. Science 297: 389-392., Tupuxuara leonardii Kellner 1994, and Zhejiangopterus linhaiensis Cai and Wei 1994.
SYSTEMATIC PALEONTOLOGY
PTEROSAURIA Kaup 1834
PTERODACTYLOIDEA Plieninger 1901
AZHDARCHOIDEA NesovNESOV LA. 1984. Upper Cretaceous pterosaurs and birds from central Asia. Paleontol J 1984(1): 38-49. 1984
TAPEJAROMORHA AndresANDRES B, CLARK J and XU X. 2014. The earliest pterodactyloid and the origin of the group. Curr Biol 24(9): 1011-1016. et al. 2014
Keresdrakon gen. nov.
Etymology: A combination of Keres, death-spirits who personified violent death in Greek mythology and are associated to doom and/or plunder; and drakon, which is the Ancient Greek word for dragon or huge serpent.
Type species: Keresdrakon vilsoni, type by monotypy.
Diagnosis: The same as for the type species.
Keresdrakon vilsoni gen. et sp. nov.
Etymology. In honor to Mr. Vilson Greinert, a volunteer who dedicated hundreds of hours preparing most of the specimens from 'cemitério dos pterossauros' site housed in CENPALEO and in the town of Cruzeiro do Oeste.
Holotype. Partial skeleton including the anterior portion of the skull, quadrate, almost complete lower jaw, two cervical vertebrae, four dorsal vertebrae, sternum, elements of the gastralia, right scapula, incomplete humeri, proximal portion of a radius, incomplete femora, incomplete tibia, left ilium, right pubis, left ischium and other fragmentary elements (CP.V 2069) housed at the Centro Paleontológico (CENPALEO) of the Universidade do Contestado, Mafra, Santa Catarina, Brazil (Fig. 3, Table I). It was recovered from bonebed C (Fig. 2).
Paratypes. Rostral end of a skull, including the anterior part of the nasoantorbital fenestra (CP.V 2068, Fig. 4) and the rostral end of lower jaw (CP.V 2070, Fig. 4); incomplete mid-cervical vertebra (CP.V 2445); mid-cervical vertebra, tibia (shaft) and rib (CP.V 2317); scapulae (CP.V 2004 - right, 2075 - left, 2339 - right); coracoid (CP.V 2083 - left); humerus (CP.V 1011 - right); ulnae (CP.V 2073 right, 2074 left, 2077 right, the latter used for osteohistological analysis), radii (CP.V 2251 and 2247); and first phalanges of the fourth wing finger (CP.V 2242 right, 2248 left, 2373 right) (Table II).
Referred specimens. Fragment of two jaws (CP.V 2254; CP.V 2343); two incomplete mandibular rami (CP.V 2071); one mid-cervical vertebra (CP.V 2398), incomplete dorsal vertebrae (CP.V 4181, CP.V 2234, 2316); elements of a large wing (CP.V 5797); femora (CP.V 2081, 2245); tibia (CP.V 2246); one large incomplete long bone interpreted as the first wing phalanx (CP.V. 2374) used for osteohistological purposes (see osteohistology section) (Table III).
Measurements (in mm) of the referred specimens of Keresdrakon vilsoni nov. gen., nov. sp.** estimated;
Type locality, horizon and age. 'cemitério dos pterossauros' site (53° 03′ 53,4″W; 23° 45′ 34,5″S - contra Langer et al. 2019), Cruzeiro do Oeste, Paraná State, Brazil; Bauru Basin, Caiuá Group, Goio-Erê Formation Cretaceous (Milani et al. 2007, Basilici et al. 2012, Batezelli 2015).
Diagnosis. Azhdarchoid pterodactyloid with the following autapomorphies: short blunt ridge on the dorsal surface of the posterior end of the dentary; foramen on the ventral surface at the anterior half of the proximal articulation of the first phalanx of digit IV; and foramen on lateral surface of the ischium.
The new species can be further distinguished from other azhdarchoid pterosaurs by the following combination of characters: dorsal margin of the premaxillae above the nasoantorbital fenestra rounded; sagittal groove on the dorsal surface of the premaxillae above the nasoantorbital fenestra; ridge on the medial surface of the splenial; ventral bar of the nasoantorbital fenestra thick; lateral pneumatic foramen on mid-cervical vertebra large; and strongly asymmetrical sternal articulation of the coracoid.
DESCRIPTION AND COMPARISON
The material of Keresdrakon vilsoni is preserved in a red coarse and friable sandstone (Figs. 3-9, Tables I-III). The bones show different degrees of compression, but most maintain at least some three-dimensionality, an uncommon condition regarding pterosaurs. Although some specimens were damaged due to weathering, there are several elements with breakages that have occurred prior or during the fossilization process. Based on the skulls and the first wing phalanges, at least three individuals are recognized so far but the actual number must be higher, particularly due to the fact that elements of the new pterosaur were found in three different levels throughout the section (Fig. 2).
The holotype (CP.V 2069) is the most complete specimen (Fig. 3). The bones were found associated but not articulated except for the upper and lower jaw. Most elements are not fused such as the scapula (found isolated and not fused to the coracoid), the distal epiphysis of the right humerus, and the pelvic elements. The following bones are fused: premaxillae with each other and with the maxillae, the splenial with the dentary, the opposing dentaries, and the centrum with the neural arch of the preserved mid-cervical vertebrae. As far as we know from pterosaur ontogeny, CP.V 2069 likely represents an animal that was between ontogenetic stage (OS) 2 and 3 sensuKellnerKELLNER AWA. 2015. Comments on Triassic perosaurs with discussion about ontogeny and description of new taxa. An Acad Bras Cienc 87: 669-689. (2015).
Holotype of Keresdrakon vilsoni gen. et sp. nov. (CP.V 2069). Skull and lower jaw are presented in right lateral view. Abbreviations: cra - skull, cv - cervical vertebra, fe - femur, gas - gastralia, hu - humerus, man - mandible, q - quadrate, ra - radius, ri - ribs, sca - scapula, st - sternum, ti - tíbia; l - left, r - right. Scale bar = 100mm.
The holotype (CP.V 2069) also shows the best preserved skull of the new species, formed by the rostral end until part of the nasoantorbital fenestra and a lower jaw that lacks the most distal portion of the mandibular rami (Figs. 3-4a, c). Additional cranial material consists of the anterior portion of an upper jaw (CP.V 2068, Fig. 4a) and part of a mandibular symphysis (CP.V 2070, Figs. 4d, e). Two additional jaw fragments (CP.V 2254; CP.V 2343) and incomplete mandibular rami (CP.V 2071) are also tentatively referred to this species.
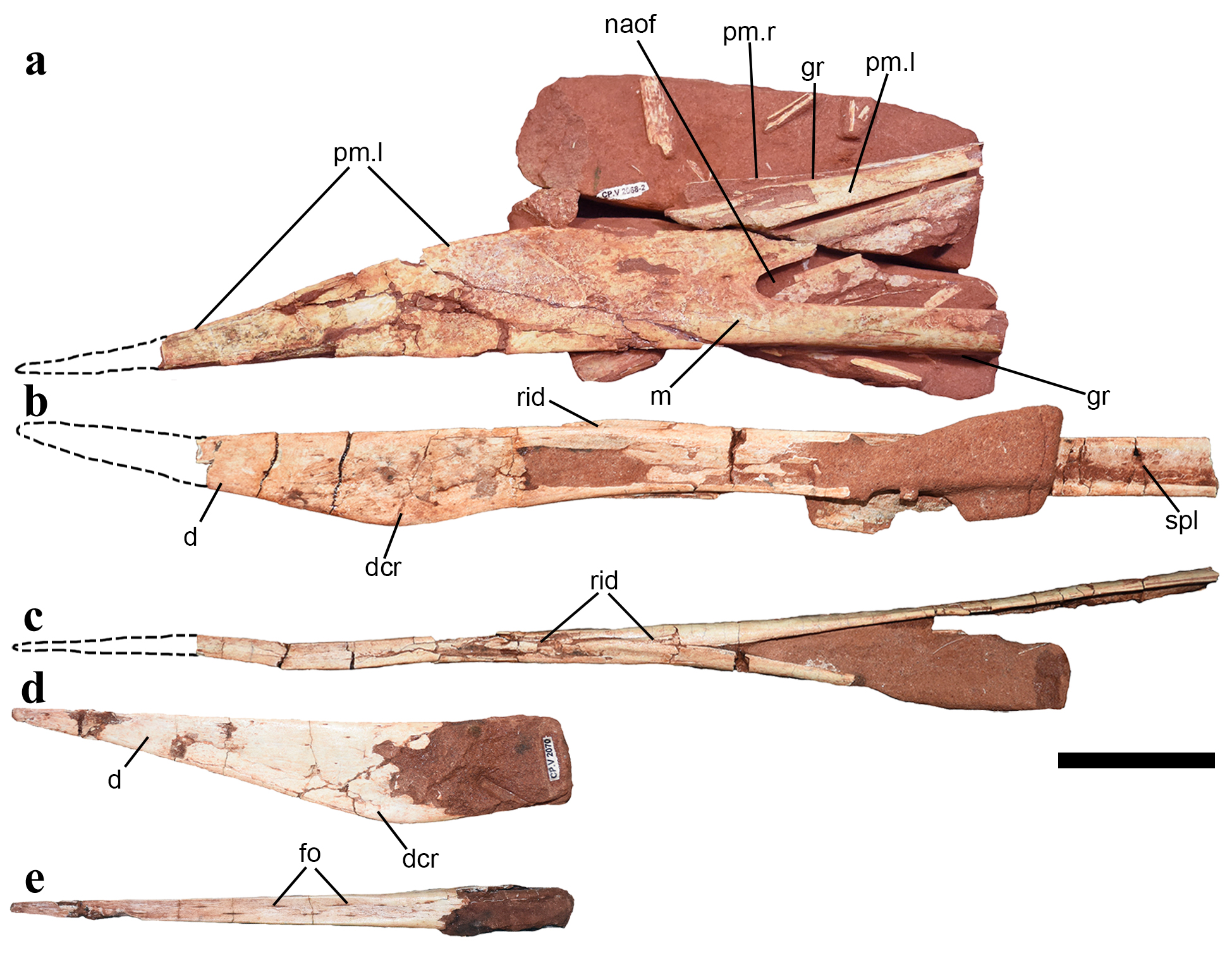
Skull and lower jaws of Keresdrakon vilsoni gen. et sp. nov. (a) Incomplete skull showing the rostral part, in left lateral view (CP.V 2068), (b) incomplete lower jaw (CP.V 2069 part of the holotype) in left lateral and (c) dorsal views, (d) anterior portion of a dentary (CP.V 2070) in left lateral and (e) dorsal views. Abbreviations: d - dentary, dcr - dentary crest, fo - foramina, gr - groove, m - maxilla, naof - nasoantorbital fenestra, pm - premaxilla, rid - ridge, spl - splenial; l - left, r - right. Scale bar = 50mm.
Based on these specimens, Keresdrakon vilsoni is toothless and bears a low skull. The rostrum is elongated, longer than that of any tapejarid (WangWANG XL and ZHOU ZH. 2003a. A new pterosaur (Pterodactyloidea, Tapejaridae) from the Early Cretaceous Jiufotang Formation of western Liaoning, China and its implications for biostratigraphy. Chin Sci Bull 48: 16-23. and Zhou 2003a, LüLÜ JC, JIN XS, UNWIN DM, ZHAO LJ, AZUMA Y and JI Q. 2006. A new species of Huaxiapterus (Pterosauria: Pterodactyloidea) from the Lower Cretaceous of western Liaoning, China with comments on the systematics of tapejarid pterosaurs. Acta Geol Sin 80: 315-326. et al. 2006, KellnerKELLNER AWA and CAMPOS DA. 2007. Short note on the ingroup relationships of the Tapejaridae (Pterosauria, Pterodactyloidea). Bol Mus Nac N S Geol Rio de Janeiro 75: 1-14. and Campos 2007, PinheiroPINHEIRO FL, FORTIER DC, SCHULTZ CL, ANDRADE JAFG and BANTIM RAM. 2011. New information on the pterosaur Tupandactylus imperator, with comments on the relationships of Tapejaridae. Acta Palaeontol Pol 56: 567-580. et al. 2011, Kellner 2013), chaoyangopterid (Wang and Zhou 2003bWANG XL and ZHOU ZH. 2003b. Two new pterodactyloid pterosaurs from the Early Cretaceous Jiufotang Formation of western Liaoning, China. Vert Palas 41: 34-41., Lü et al. 2008LÜ JC, UNWIN DM, XU L and ZHANG XL. 2008. A new azhdarchoid pterosaur from the Lower Cretaceous of China and its implications for pterosaur phylogeny and evolution. Naturwissenschaften 95: 891-897.) and the azhdarchid Zhejiangopterus linhaiensis Cai and Wei 1994, but shorter than that of the azhdarchid Quetzalcoatlus sp. (Kellner and Langston 1996KELLNER AWA and LANGSTON W. 1996. Cranial remains of Quetzalcoatlus (Pterosauria, Azhdarchidae) from the Late Cretaceous sediments of Big Bend National Park, Texas. J Vert Paleontol 16(2): 222-231.). The rostrum ends in a pointed tip. The dorsal margin of the rostrum until the anterior half of the nasoantorbital fenestra is slightly sigmoid, differing in this respect from all other pterodactyloids. The ventral margin is slightly curved downwards but not as in tapejarines (Kellner and Campos 2007). It lacks the large quantities of foramina that are commonly observed in tapejarids, including Caiuajara (Manzig et al. 2014). Occlusion of jaws form no gap, a different condition from tapejarines (Kellner and Campos 2007).
The anterior angle of the nasoantorbital fenestra (CP.V 2068 ~19.3°; CP.V 2069 ~22.5°) is more acute than in other azhdarchoids except for Quetzalcoatlus sp. (~15.8°, see Kellner and Langston 1996). In lateral view, the ventral bar of this opening formed by the premaxillae/maxillae is thicker than in other azhdarchoids, except in Thalassodromeus (Kellner and Campos 2002, PêgasPÊGAS RV, COSTA FR and KELLNER AWA. 2018. New information on the osteology and a taxonomic revision of the genus Thalassodromeus (Pterodactyloidea, Tapejaridae, Thalassodrominae). J Vert Paleontol 38(2): e1443273. et al. 2018). Although not complete, it is unlikely that this cranial opening is as large as observed in the tapejarids (Kellner and Campos 2007) or high as in chaoyangopterids (Lü et al. 2008).
There is no evidence of a premaxillary sagittal crest until the anterior margin of the nasoantorbital fenestra, and it is unknown if such a structure, which is a common feature among azhdarchoids (Kellner and Langston 1996, Wang and Zhou 2003a, Kellner and Campos 2007, Lü et al. 2008), was present more posteriorly. Above the anterior portion of the nasoantobital fenestra, the premaxillae have a rounded dorsal margin and display a sagittal groove (Figs. 3-4a). Inside this cranial opening, the premaxillae are fused, have laterally raised borders and form medially a flat surface, lacking the ridge present in Caiuajara (Manzig et al. 2014). No suture with the maxillae is visible. The palate is slightly concave and gets more flattened posteriorly, lacking any ventral ridge as in thalassodromines (Kellner and Campos 2007) or an expansion close to the anterior opening of the nasoantorbital, which is diagnostic for tapejarines (Kellner and Campos 2007). On the lateral side, a longitudinal groove is observed.
An incomplete right quadrate (CP.V 2069) is preserved, showing that it is a long rod-like bone that has a thin medial flange (incomplete) and laterally shows an elongated groove probable the articulation for the quadratojugal.
The lower jaw is long and ends in a pointed tip (Figs. 3, 4). As common in pterodactyloids, the main element is the dentary, which fuses into a symphysis. The occlusal (=dorsal) surface is pierced by several foramina, which are more than those of the upper jaw, but much fewer than those in tapejarines (Kellner and Campos 2007, Andres et al. 2014, Manzig et al. 2014) and similar to the low number present in basal tapejarines (Kellner 2013, PêgasPÊGAS RV, LEAL MEC and KELLNER AWA. 2016. A basal tapejarine (Pterosauria; Pterodactyloidea; Tapejaridae) from the Crato Formation, Early Cretaceous of Brazil. PloS One 11(9): e0162692. et al. 2016). The ventral surface of the symphysis is blunt and bears a small and thick dentary crest. It differs from the deeper lower jaw at the posterior end of the mandibular symphysis of Argentinadraco (Kellner and Calvo 2017). The occlusal surface is anteriorly flattened and becomes slightly concave posteriorly. A medial ridge starts at the region corresponding to the end of the dentary crest and runs at least for 40 mm (up to 65 mm) until almost reaching the posterior dorsal end of the symphysis. Such a dentary dorsal ridge has never been reported before in pterosaurs so far. Argentinadraco bears two mandibular ridges separated by a depression and laterally boarded by a sulcus, differing from the condition of Keresdrakon. Although the mandible is not complete, the symphysis in the holotype (CP.V 2069) has reached about 220 mm and corresponded up to 46% of the estimated mandible length (~470mm). Despite the fact that the exact posterior limit of the dorsal portion of the mandibular symphysis cannot be established, it seems not to extend further posteriorly relative to the ventral end, neither to develop a symphyseal shelf, differing from several azhdarchoids (Kellner and Langston 1996, VulloVULLO R, MARUGÁN-LOBÓN JS, KELLNER AWA, BUSCALIONI AD, GOMEZ B, DE LA FUENTE M and MORATALLA JJ. 2012. A new crested pterosaur from the Early Cretaceous of Spain: the first European tapejarid (Pterodactyloidea: Azhdarchoidea). PLoS ONE 7: e38900. et al. 2012, Kellner 2013, Pêgas et al. 2016). The typical tapejarine step-like dorsal margin of the mandibular symphysis (Kellner and Campos 2007) is absent. At the most posterior preserved end of the right mandibular ramus (CP.V 2069), the medial portion of the lower jaw can be observed showing the splenial. This bone is very thin and shows a blunt longitudinal ridge that does not extend anteriorly.
Few cervical vertebrae are preserved and might belong to distinct individuals (Fig. 5). The elements from the middle series are sub-equal in length, with the centrum longer than wide but not to the same degree as in archaeopterodactyloids, chaoyangopterids and azhdarchids (Nesov 1984, HowseHOWSE SCB. 1986. On the cervical vertebrae of the Pterodactyloidea (Reptilia: Archosauria). Zool J Linn Soc 88: 307-328. 1986, Kellner 2003KELLNER AWA. 2003. Pterosaur phylogeny and comments on the evolutionary history of the group. In: Buffetaut E and Mazin JM (Eds), Evolution and palaeobiology of pterosaurs, London: Geological Society, Special Publications, 105-137 (Geol Soc London Spec Pub 217)., AndresANDRES B and JI Q. 2008. A new pterosaur from the Liaoning Province of China, the phylogeny of the Pterodactyloidea, and the convergence in their cervical vertebrae. Palaeontology 51: 453-469. and Ji 2008, Lü et al. 2008, LealLEAL MEC, PÊGAS RV, BONDE N and KELLNER AWA. 2018. Cervical vertebrae of an enigmatic pterosaur from the Crato Formation (Lower Cretaceous, Araripe Basin, NE Brazil). In: Hone DWE, Witton MP and Martill DM (Eds), New perspectives on pterosaur palaeobiology, London: Geological Society, Special Publications, 195-208 (Geol Soc London Spec Pub 455). et al. 2018). They bear developed postexapophyses and hypapophysis. On each side of the hypapophysis, small projections - the preexapophysis - are present but not as developed as in the azhdarchid Eurazhdarcho (Vremir et al. 2013). This structure corresponds to a ridge identified in an azhdarchid middle cervical vertebra from Morroco (RodriguesRODRIGUES T, KELLNER AWA, MADER BJ and RUSSELL D. 2011. New pterosaur specimens from the Kem Kem beds (Upper Cretaceous, Cenomanian) of Morocco. Riv Ital Paleontol S 117(1): 149-160. et al. 2011) and the preexapophyseal articulations reported in the pteranodontoid Pteranodon (BennettBENNETT SC. 2001. The osteology and functional morphology of the Late Cretaceous pterosaur Pteranodon part 1 - General description and osteology. Palaeontographica 260: 1-112. 2001). Although not reported, they are present in at least some tapejarines, including Tapejara (WellnhoferWELLNHOFER P and KELLNER AWA. 1991. The skull of Tapejara wellnhoferi Kellner (Reptilia, Pterosauria) from the Lower Cretaceous Santana Formation of the Araripe Basin, Northeastern Brazil. Mitt Bayer Staatsslg Paläont Hist Geol 31: 89-106. and Kellner 1991) and Caiuajara (Manzig et al. 2014) (AWAK pers. obs.). They have not been reported in thalassodromines (AiresAIRES ASS, KELLNER AWA, MÜLLER RT, DA SILVA LR, PACHECO CP and DIAS-DA-SILVA S. 2013. New postcranial elements of the Thalassodrominae (Pterodactyloidea, Tapejaridae) from the Romualdo Formation (Aptian-Albian), Santana Group, Araripe Basin, Brazil. Palaeontology 57: 343-355. et al. 2013) and anhanguerids (Wellnhofer 1985WELLNHOFER P. 1985. Neue Pterosaurier aus der Santana-Formation (Apt) der Chapada do Araripe, Brasilien. Palaeontogr Abt A 187: 105-182., 1991WELLNHOFER P. 1991. Weitere Pterosanrierfunde aus der Santana-Formation (Apt) der Chapada do Araripe, Brasilien. Palaeontogr Abt A 215: 43-101.).
- Cervical vertebrae of Keresdrakon vilsoni gen. et sp. nov., CP.V 6495 in (a) ventral, (b) right lateral, (c) dorsal and (d) anterior views; CP.V 2445 in (e) ventral, (f) dorsolateral, (g) posterior views. Note the ossified tube that encloses the neural cannal in (F) marked by an arrow. Abbreviations: con - condyle, fo - foramen, fopn - foramen pneumaticum, hyp - hypapophysis, nc - neural canal, ns - neural spine, poex - postexapophysis, poz - postzygapophysis, prex - preexapophysis, prz - prezygapophysis, tra - trabeculae. Scale bar = 20mm.
The neural spine is tall and blade-like (Fig. 5). The centrum is laterally pierced by a pneumatic foramen, which is absent in chaoyangopterids (Lü et al. 2008) and azhdarchids (Howse 1986). The size of this pneumatic foramen is comparable to the one found in anhanguerids (Wellnhofer 1985, 1991, Buchmann and Rodrigues 2019), and is much larger than that in the pteranodontoid Pteranodon (Bennett 2001) and tapejarids (e.g., Aires et al. 2013). There are also three pneumatic openings on the anterior and posterior articulation surfaces in some vertebrae – one above and one on each side of the neural canal (CP.V. 2445, CP.V 2317; Fig. 5g). In one mid-cervical element, the lateral pneumatic openings at the anterior articulation are reduced or absent (Fig. 5d). In CP.V 2445, the neural arch on the left side is broken, revealing that the inner part of the vertebra is hollow and has the neural canal encased by a bony tube supported by trabeculae (Fig. 5f). Such a construction has been previously observed in one undescribed tapejarid (Kellner 2006) and might be a general feature of at least derived pterosaurs. This vertebra (CP.V 2445) also displays one foramen on each side of the hypapophysis. No cervical rib was found in the mid-cervical elements.
The sternum (CP.V 2069) is a large plate-like and thin quadrangular bone (Figs. 3, 6). During the collecting process the sternal plate (Fig. 6a) was separated from the anterior portion of the sternum (Figs. 6b, c). The cristospine is shallow and elongated, with a blunt anterior and ventral margin. At the base of the cristospine on the dorsal surface is a large foramen (Figs. 6b, c). Several elements of the thin “V” shaped gastralia are preserved. The scapula is an elongated bone with a straight shaft and a flattened and elongated proximal articulation (Fig. 7j). It shows a well-developed tuberculum at the lateroposterior margin, similar to that of Caupedactylus (Kellner 2013). On the lateral surface above the glenoid this bone shows a marked depression. The sole coracoid (CP.V 2083) attributed to this species is incomplete, lacking the glenoid portion (Figs. 7h-i). The developed coracoidal flange as the one reported in some azhdarchids (Kellner and Langston 1996) is absent. However, there is a broad tubercle on the ventroposterior margin (Fig. 7h), a feature commonly observed in tapejarids (Kellner 2013). The articulation with the sternum is dorsoventrally flattened, lacks a posterior expansion and is strongly concave. It further has the posterior portion more developed than the anterior one, giving it an asymmetric outline that, to our knowledge, has not been reported in any other pterosaur before (Figs. 7h-i).
Sternum of Keresdrakon vilsoni gen. et sp. nov. (CP.V 2069, part of the holotype). (a) ventral, cristospine in dorsal (b) and (c) right lateral views. Abbreviations: afc - articular facet for coracoid, cs - cristospine, fo - foramen, stp - sternal plate Scale bars = (a) and (b) 50mm, (c) 20mm.
Keresdrakon vilsoni gen. et sp. nov. (a) complete left first phalanx of manual digit IV (CP.V 2248), (b) proximal articulation of left first phalanx of manual digit IV (CP.V 2248), (c) proximal articulation of right first phalanx of manual digit IV (CP.V 2373); right humerus (CP.V 1011) in (d) medial, (e) dorsal, (f) ventral and (g) lateral views; left incomplete coracoid in (h) ventral and (i) dorsal views, (j) scapula in dorsal view. Abbreviations: dpc - deltopectoral crest, fopn - foramen pneumaticum, tucor - coracoidal tuberculum. Scale bars = (b) and (c) 10mm, all others 50mm.
Overall, the humerus (Figs. 7d-g) shows a similar shape to the one of Caupedactylus (Kellner 2013). The deltopectoral crest (CP.V 1011) is long and quadrangular in lateral view, with parallel margins. It is proximally placed and directed ventrally, curving gently inwards. While there is no pneumatic foramen on the dorsal surface of the proximal portion, a large one can be found piercing the ventral surface, placed at the base of the deltopectoral crest (Fig. 7f), which is more developed than in Caupedactylus (Kellner 2013).
The ulna is a long bone with the proximal and distal articulations slightly twisted relative each other. In all of these elements attributed to this species, there is a marked depression on the posterior surface at the proximal articulation (CP.V 2073, CP.V 2077) showing that the ulnar epiphysis was unfused in these specimens (Fig. 9c). A developed foramen pierces the anterior surface of the proximal articulation. The distal articulation is flattened anteroposteriorly and shows a small ridge along the dorsal margin of the anterior surface. Although no radius and ulna can be confidently attributed to the same individual, in those specimens where both bones have about the same length, the diameter of the radius is about half that of the ulna, a common feature within azhdarchoids (Kellner 2003). The distal articulation of the radius has a flattened posterior surface, which is pierced by a pneumatic foramen, and a more irregular anterior surface that shows a long blunt ridge (anterior tubercle) at the lower ventral portion.
The first phalanges of the fourth wing finger that are attributed to Keresdrakon lack the extensor tendon process, which shows that it was unfused in all specimens (CP.V 2242, 2248, 2373; Figs. 7a-c). The shaft of the phalanges is slightly bowed anteriorly, with a concave anterior margin and a convex posterior one (Fig. 7a), a feature that has been observed in dsungaripterids (Wellnhofer 1978WELLNHOFER P. 1978. Pterosauria. Handbuch der Paläoherpetologie, Teil 19. Stuttgart: Gustav Fischer Verlag, 82 p.) and some tapejarines (EckECK K, ELGIN RA and FREY E. 2011. On the osteology of Tapejara wellnhoferi Kellner 1989 and the first occurrence of multiple specimen assemblage from the Santana Formation, Araripe Basin, NE-Brazil. Swiss J Palaeontol 130: 277-296. et al. 2011). These elements show a pneumatic foramen on the ventral side of the proximal articulation situated close to the posterior margin. All three also show a second pneumatic foramen closer to the anterior margin (Figs. 7b, c), which has never been reported in any pterosaur before. In some specimens this foramen is quite clear, while in others it is more difficult to observe due to compression.
The holotype (CP.V 2069) also shows the remains of a pelvic girdle (Fig. 8). The ilium is an elongated bone with a developed postacetabular process that has the anterior projection particularly expanded, as observed in some tapejarids (AWAK pers. obs.). This bone forms most of the acetabulum, a common feature in derived pterosaurs (Fig. 8a). The pubis is a dorsoventrally elongated bone with a stout dorsal portion that contacts the ilium, followed by a thin bonny plate that gently curves medially (Figs. 8b, c). The anterior portion of this bony plate further bends laterally to form the anterior opening of the pelvic canal. The ventral margin of the pubis is concave, while the posterior margin that forms the contact surface with the ischium is gently convex. This bone is perforated by a well-developed oval obturator foramen whose distal margin is formed by the ischium. Dorsal to this foramen the pubis forms a comparatively thick bony bar that has a convex posterior articulation where it joins the ischium. The obturator foramen in this species is similar in size at least to some tapejarines (Eck et al. 2011), but larger compared to most other groups of pterosaurs as, for example, anhanguerids and pteranodontids (Wellnhofer 1978, 1991, Bennett 2001, NaishNAISH D, SIMPSON M and DYKE G. 2013. A new small-bodied azhdarchoid pterosaur from the Lower Cretaceous of England and its implications for pterosaur Anatomy, diversity and phylogeny. PloS One 8(3): e58451. et al. 2013). The ischium is a subtriangular plate that gently curves medially (Figs. 8d, e). The anterior and posterior margins are almost straight, while the ventral border is slightly concave. The anterodorsal corner has a concave articulation surface that receives the dorsal part of the pubis, while the posterodorsal corner is displaced medially. On the lateral surface there is a small foramen, which has not been reported in other pterosaurs (Wellnhofer 1978, 1991, Bennett 2001, Naish et al. 2013).
Keresdrakon vilsoni gen. et sp. nov., pelvis (CP.V 2869, part of the holotype), (a) left ischium in lateral view; right pubis in (b) medial and (c) lateral views; right ischium in (d) lateral and (e) medial views. Abbreviations: ac - acetabulum, fo - foramen, gas - gastralia, obfo - obturator foramen. Scale bar = 50mm.
The femur is bowed anteriorly (Fig. 3). Its proximal articulation is made by a well-developed femur head with a constricted neck that is set from the shaft at ~135°. The femur head forms a convex rounded surface with a suboval outline and lacks a medial notch observed in some pterodactyloids. A pneumatic foramen is present on the posterior surface between the femur head and the triangular greater trochanter. The tibia is a long and much thinner element than the femur. No fibula was found suggesting that this bone was not fused in the material recovered from the new species.
OSTEOHISTOLOGY
In order to assess a preliminary histological analysis of Keresdrakon vilsoni, two bones were sectioned: one ulna (CP.V 2077) and one incomplete portion of a large element lacking articulations (CP.V 2374), interpreted as the first wing phalanx (Fig. 9). Overall, the osteohistological pattern is very similar in both samples, showing fibrolamellar primary tissue, a feature observed in most pterosaurs (de Ricqlès et al. 2000DE RICQLÈS A, PADIAN K, HORNER JR and FRANCILLON-VIEILLOT H. 2000. Palaeohistology of the bones of pterosaurs (Reptilia: Archosauria): anatomy, ontogeny, and biomechanical implications. Zool J Linn Soc 129: 349-385., ProndvaiPRONDVAI E, STEIN K, ÖSI A and SANDER MP. 2012. Life history of Rhamphorhynchus inferred from bone histology and the diversity of Pterosaurian growth strategies. PloS ONE 7: e31392. et al. 2012, Eleutério et al. 2016). The cortex thickness varies from 410 μm in the first wing phalanx (CP.V 2374) to 850 μm in the ulna (CP.V 2077). Bone microstructure shows dispersed osteocytes lacunae in the cortex with some plexiform and anastomosed canals randomly deposited.
Osteohistological sections ofKeresdrakon vilsonigen. et sp. nov. appendicular elements. Incomplete first wing phalanx of digit four (CP.V 2374) (a) and osteohistological cross section of the diaphysis; left ulna (CP. V 2077) (c) and respective osteohistological cross section of the diaphysis. Abbreviations: lag - line of arrested growth, vc - vascular canal. The yellow arrows point where the samples were taken. Scale bar = (a) and (c) 50mm, (b) and (d) 200µm.
The ulna (CP.V 2077) shows some vascular canals accessing the periosteal surface (Fig. 9d), a specific condition for juvenile to sub-adult individuals (SteelSTEEL L. 2008. The palaeohistology of pterosaur bone: an overview. Zitteliana B 28: 109-125. 2008). There are no structures throughout the entire bone circumference that point to an advanced maturity of this individual, such as growth marks, bone remodeling structures or an external fundamental system (Steel 2008, Kellner et al. 2013KELLNER AWA, CAMPOS DA, SAYÃO JM, SARAIVA AAF, RODRIGUES T, OLIVEIRA G, CRUZ LA, COSTA FR, SILVA HP and FERREIRA JS. 2013. The largest flying reptile from Gondwana: a new specimen of Tropeognathus cf. T. mesembrinus Wellnhofer, 1987 (Pterodactyloidea, Anhangueridae) and other large pterosaurs from the Romualdo Formation, Lower Cretaceous, Brazil. An Acad Bras Cienc 85: 113-135., AndradeANDRADE RCLP, BANTIM RAM, LIMA FJ, CAMPOS LS, ELEUTÉRIO LHS and SAYÃO JM. 2015. New data about the presence and absence of the external fundamental system in Archosaurs. Cad Cult Ciênc 14(1): 200-211. et al. 2015). Furthermore, the absence of the endosteal lamella suggests that this bone belongs to a juvenile. InHamipterus and some anhanguerid pterosaurs the size variation is reflected in the osteohistological pattern of each bone (SayãoSAYÃO JM. 2003. Histovariability in bones of two pterodactyloid pterosaurs from the Santana Formation, Araripe Basin, Brazil: preliminary results. In: Buffetaut E and Mazin JM (Eds), Evolution and palaeobiology of pterosaurs, London: Geological Society, Special Publications, p. 335-342 (Geol Soc London Spec Pub 217). 2003, EleutérioELEUTÉRIO LHS, BANTIM RAM, LIMA FJ, ANDRADE RCLP, SARAIVA AAF, KELLNER AWA and SAYÃO JM. 2015. Biomechanical and physiological influences on the osteohistological deposition of Anhangueria (Pterosauria, Pterodactyloidea). Rev Bras Paleo 18(3): 403-412. et al. 2015, Wang et al. 2014), which establishes a correlation between size difference and ontogenetic development (Wang et al. 2014, 2017WANG XL et al. 2017. Egg accumulation with 3D embryos provides insight into the life history of a pterosaur. Science 358: 1197-1201. DOI: 10.1126/science.aan2329.). The osteohistological features of this specimen are consistent with a juvenile but not a neonate (see Wang et al. 2017 for histological structures of neonates and embryos). Several grains of silica and quartz fill the inner medullary cavity. These grains have possibly been deposited during the bone transport process, pointing to bone fragmentation prior to fossilization.
The most different pattern among samples belongs to the first wing phalanx (CP.V 2374), which has a unique growth mark located in the deep portion of the cortex (Fig. 9b). The presence of cyclical growth marks is continuous and partly caused by endogenous physiological processes (Eleutério et al. 2015), with lines of arrested growth being common in vertebrates as general. However, these marks are hardly found in pterosaurs, but have been recorded in Cretaceous anhanguerids (Sayão 2003, Eleutério et al. 2015) and Jurassic pterosaurs (PadianPADIAN K, HORNER JR and DE RICQLÈS A. 2004. Growth in small dinosaurs and pterosaurs: The evolution of archosaurian growth strategies. J Vert Paleont 24: 555-571. et al. 2004). The presence of these growth marks suggests that this bone belongs to an ontogenetically less developed individual compared to others.
PHYLOGENETIC RESULTS
Phylogenetic analysis resulted in 6 most-parsimonious trees (MPTs), with 341 steps, a consistency index of 0.66 and retention index of 0.87. Azhdarchoidea is supported by the following synapomorphies: character state 7(2) - a dorsoventrally elongated orbit (piriform); 12(3) - slit-like lower temporal fenestra; 56(2) - a cranio-mandibular articulation under the anterior margin of the orbit; and 75(3) - a long extension of the contact surface of opposing dentaries, up to 55% the mandible length. Furthermore, Keresdrakon vilsoni gen. et sp. nov. was recovered as a sister taxon of the Tapejaridae (Fig. 10), which would enable to recognize a non-tapejarid lineage within the Tapejaromorpha (Andres et al. 2014). Tapejaromorphs are characterized by two synapomorphies: character state 79(1) - a dentary ossified sagittal crest; and 118(1) - coracoid with developed tuberculum on the posteroventral margin. The Tapejaridae (sensu Kellner 2003) is supported by character state 27(1) - anterior portion of the rostrum forming a high ossified plate, which is absent in Keresdrakon.
Time-calibrated phylogenetic tree showing the relationships of Keresdrakon vilsoni gen. et sp. nov. within the Pterodactyloidea. Intermittent bars show uncertain temporal range. Letters in intermittent bars indicate controversial age hypotheses of the Goio-Erê Formation: (a) Albian-Aptian; (b) Turonian-Campanian. Outgroup relationships are not shown (see Holgado et al. 2019 - Supplementary Information for further details). Stratigraphic chart modified from Cohen et al. (2013COHEN KM, FINNEY SM, GIBBARD PL and FAN JX. 2013. The ICS International Chronostratigraphic Chart. Episodes 36: 199-204.).
DISCUSSION
Two species are considered sympatric when they show the same or overlapping geographical distribution, irrespective of their macrohabitat (RivasRIVAS LR. 1964. A reinterpretation of the concepts “sympatric” and “allopatric” with proposal of the additional terms “syntopic” and “allotopic”. Syst Biol 13: 42-43. 1964), and there are several examples in modern environments (e.g., LealLEAL M and FLEISHMAN LJ. 2002. Evidence for habitat partitioning based on adaptation to environmental light in a pair of sympatric lizard species. Proc Royal Soc B 269: 351-359. and Fleishman 2002, LuiselliLUISELLI L. 2006. Resource partitioning in the communities of terrestrial turtles: a review of the evidences. Rev Ecol-Terre Vie 61(4): 353-365. 2006, PigotPIGOT AL and TOBIAS JA. 2015. Dispersal and the transition to sympatry in vertebrates. Proc Royal Soc B 282(1799): 1-8. and Tobias 2015). Even though sympatry is normally used for closely related species and associated with speciation (e.g. MayrMAYR E. 1942. Systematics and the origin of species. New York: Columbia University Press, 334 p. 1942, DiamondDIAMOND JM. 1975. Assembly of species communities. In: Cody ML and Diamond JM (Eds), Ecology and evolution of Communities, Cambridge: Harvard University Press, p. 342-444. 1975, VanVAN VALEN L. 1976. Ecological species, multispecies, and oaks. Taxon 25(2/3): 233-239. Valen 1976, Pigot and Tobias 2015), there are cases in which this concept is applied to phylogenetically more distant taxa (e.g., MagnussonMAGNUSSON WE. 1985. Habitat selection, parasites, and injuries in Amazonian crocodiles. Amazoniana 9: 193-204. 1985, RaoRAO RJ and CHOUDHURY BC. 1990. Sympatric distribution of gharial and mugger in India. J Bombay Nat Hist Soc 89: 312-315. and Choudhury 1990, SolerSOLER M and MØLLER AP. 1990. Duration of sympatry and coevolution between the great spotted cuckoo and its magpie host. Nature 343: 748-750. and Møller 1990, ChoudharyCHOUDHARY S, CHOUDHURY BC and GOPI GV. 2018. Spatio-temporal partitioning between two sympatric crocodilians (Gavialis gangeticus & Crocodylus palustris) in Katarniaghat Wildlife Sanctuary, India. Aquatic Conserv: Mar Freshw Ecosyst 2018: 1-10. et al. 2018).
Sympatry in the fossil record, particularly considering extinct vertebrates, is difficult to be established since different individuals that have never shared the same habitat might be preserved together in the same deposit as a result of time averaging (e.g., BehrensmeyerBEHRENSMEYER AK, KIDWELL SM and GASTALDO RA. 2000. Taphonomy and paleobiology. Paleobiology 26: 103-147. et al. 2000). Bone beds or the occurrence of specimens in close associations are occasionally used as indication of coexistence (e.g., CabreiraCABREIRA SF et al. 2016. A unique Late Triassic dinosauromorph assemblage reveals dinosaur ancestral anatomy and diet. Curr Biol 26: 1-6. et al. 2016), although caution is always needed (e.g., RogersROGERS RR and KIDWELL SM. 2000. Associations of vertebrate skeletal concentrations and discontinuity surfaces in continental and shallow marine records: a test in the Cretaceous of Montana. J Geol 108: 131-154. and Kidwell 2000). This situation is even more complicated in pterosaurs due to the lack of bonebeds (e.g., Kellner 2017KELLNER AWA. 2017. Rebuttal of Martin-Silverstone et al. 2017, 'Reassessment of Dawndraco kanzai Kellner 2010 and reassignment of the type specimen to Pteranodon sternbergi Harksen, 1966'. Vertebrate Anatomy Morphology Palaeontology 3: 81-89. DOI 10.18435/B54D49.). So far, some authors assume sympatry for these flying reptiles if species or genera occur in the same stratigraphic unit (e.g., UpchurchUPCHURCH P, ANDRES B, BUTLER RJ and BARRETT PM. 2015. An analysis of pterosaurian biogeography: implications for the evolutionary history and fossil record quality of the first flying vertebrates. Hist Biol 27: 697-717. et al. 2015, LongrichLONGRICH NR, MARTILL DM and ANDRES B. 2018. Late Maastrichtian pterosaur from North Africa and mass extinctions of Pterosauria at the Cretaceous-Paleogene boundary. PLoS Biol 16: e2001663. et al. 2018), but direct and more conclusive evidences are difficult to obtain.
The bonebeds of Cruzeiro do Oeste, however, represent a particular case that provides, to our knowledge, the first direct evidence for sympatry in Pterosauria. YoungYOUNG CC. 1973. Reports of Paleontological Expedition to Sinkiang (II). Pterosaurian Fauna from Wuerho, Sinkiang. Memoirs of the Institute of Vertebrate Palaeontology and Paleoanthropology, Academy Sinica 11: 18-35. (1973) has reported the occurrence of two taxa in the same deposits in two localities in Wuerhe, but it is not clear if they actually came from the same horizon, as is the case of the material studied here. The 'cemitério dos pterossauros' site also shows the co-occurrence of a theropod dinosaur and two flying reptile species that can confidently be regarded as part of the same paleobiological community (e.g., Figs. 11-16).
Sample (CP.V 6559) from bonebed A of Figure 2, showing on the left (a) side a partial skeleton of the pterosaur Caiuajara dobruskii and on the right (b) the remains of the dinosaur Vespersaurus paranaensis. White line shows the limits between the two specimens. Scale bar = 100mm.
Sample (CP.V 7228) from bonebed A of Figure 2 showing the close association of a femur of Caiuajara dobruskii (a) and the incomplete tail and forelimb (including partial manus) of Vespersaurus paranaensis (b), separated by the white line. Scale bar = 100mm.
Paratype of Caiuajara dobruskii (CP.V 1003) (a) and a partial articulated hind limb of Vespersaurus paranaensis with tibia, fibula, tarsals (including astragalus) and metatarsals on the upper right corner (b). This specimen was collected from bonebed C of Figure 2. White line shows the limits of the two specimens. Scale bar = 100mm.
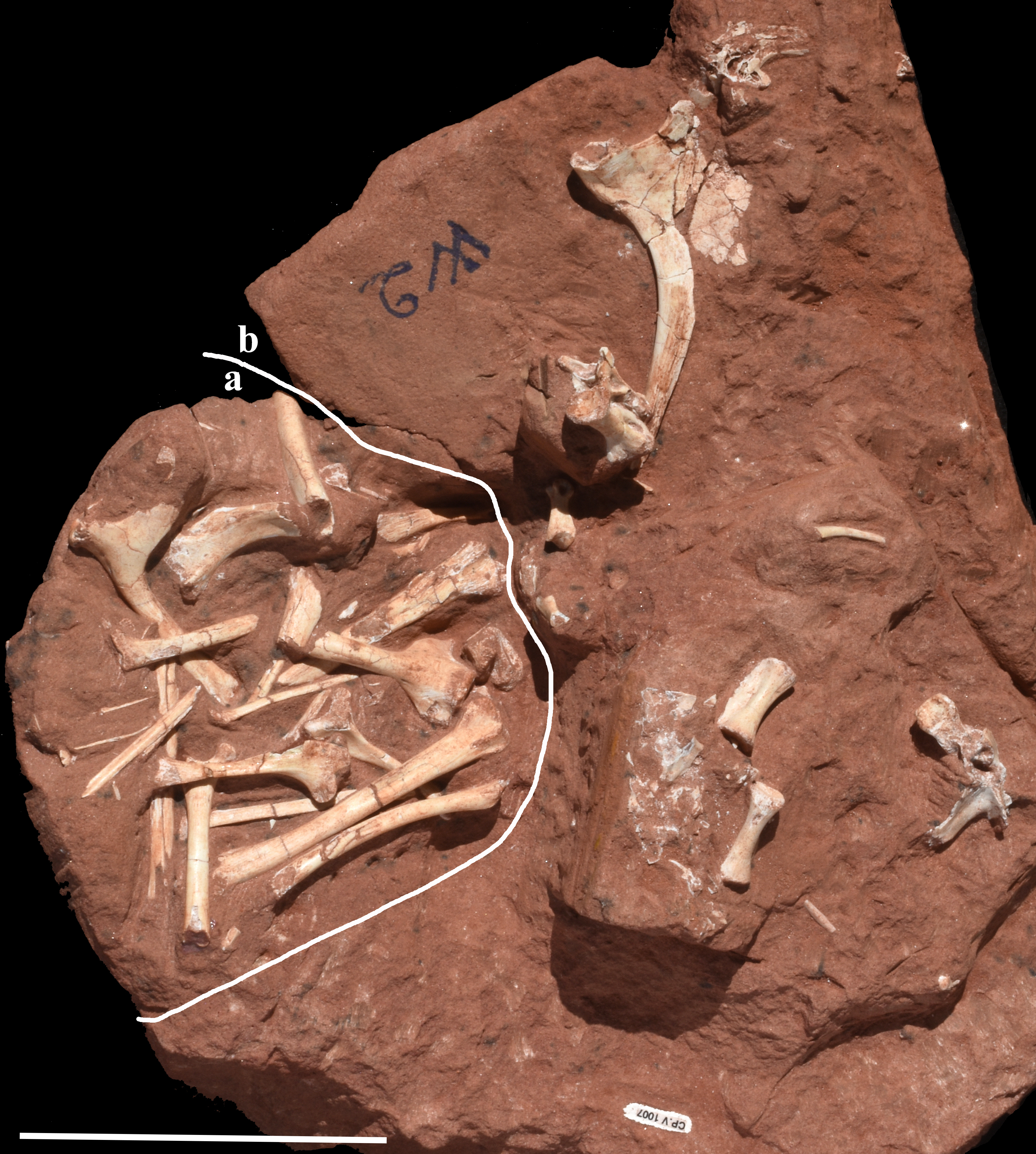
Sample (CP.V 1007) from bonebed level C of Figure 2, showing the close association of Caiuajara dobruskii (a) and Vespersaurus paranaensis (b) separated by the white line. Scale bar = 100mm.
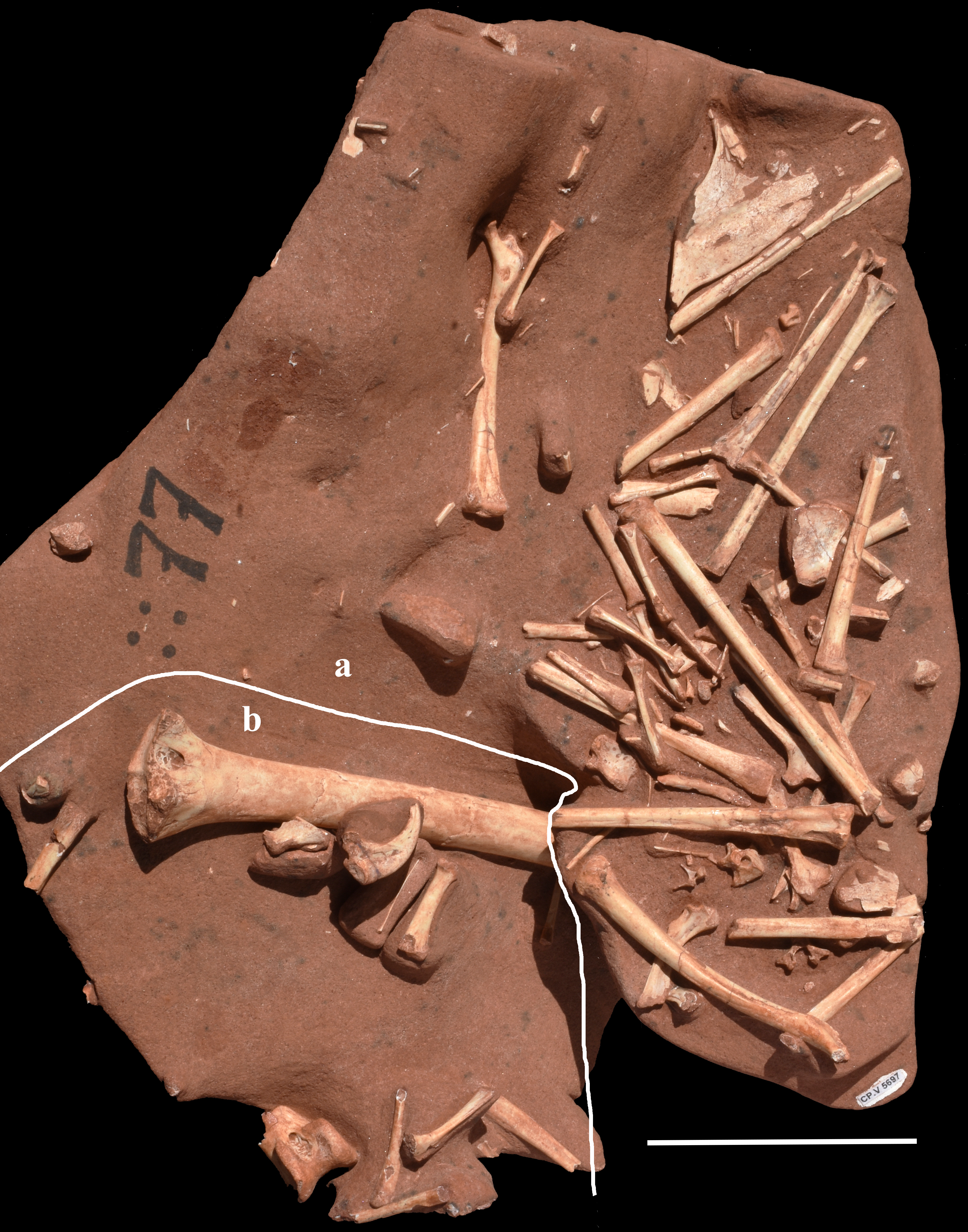
Sample (CP.V 5697) from bonebed C of Figure 2, showing on the right (a) a partial skeleton of the Caiuajara dobruskii the left (b) elements of Keresdrakon vilsoni gen. et sp. nov. separated by the white line. Scale bar = 100mm.
Sample (CP.V 2374) from bonebed C of Figure 2, showing two caudal vertebrae from Vespersaurus paranaensis (a), the diaphysis of a large first phalanx of manual digit IV of Keresdrakon vilsoni gen. et sp. nov. (b), and bones of Caiuajara dobruskii (c), separated by the white line. Abbreviations: cdr - caudal vertebrae, ph1d4 - first phalanx of manual digit IV. Scale bar = 100mm.
The majority of bones collected at the 'cemitério dos pterossauros' site was found isolated, and only very few can be assigned to the same individual. This is also the case of Caiuajara dobruskii, with only one articulated specimen and few others presenting skeletal elements in close association. The same condition is observed for the dinosaur Vespersaurus, for which at least 12 individuals have been discovered. Except for some feet, two arms and few pelvic elements, all specimens known to us were found disarticulated, albeit in close association (AWAK and LCW pers. obs., specimens housed in CENPALEO and in Cruzeiro do Oeste). The holotype of Vespersaurus paranaensis is no exception, for which the assignment to the same individual of a few vertebrae from different parts of the body, pelvic elements and pes seems to be unlikely.
The material of Keresdrakon vilsoni also shows this taphonomic pattern: most elements were found isolated and only a few were associated. The great exception is the holotype (CP.V 2069). In terms of paleoecological interpretations, the disarticulated state of the material suggests that pterosaurs and dinosaurs died around an oasis, with their skeleton exposed for some time that leaded to disarticulation, and then accumulated due to episodic events of high energy such as desert storms.
At least four of such events could be identified and resulted in bone beds. In three of them (from base to top, A, B, and C, Fig. 2), the pterosaurs Caiuajara dobruskii and Keresdrakon vilsoni, as well as the dinosaur Vespersaurus paranaensis, were found in close association (Figs. 11-16). The repeating pattern of this close association argues that Keresdrakon, Caiuajara and the dinosaur were coeval. As was pointed out before (e.g., KellnerKELLNER AWA. 2010. Comments on the Pteranodontidae (Pterosauria, Pterodactyloidea) with the description of two new species. An Acad Bras Cienc 82: 1063-1084. 2010, 2017), several pterosaurs were found in the same deposit such as the Solnhofen Limestone (Wellnhofer 1975, BennettBENNETT SC. 1996. Year-classes of pterosaurs from the Solnhofen Limestone of Germany: taxonomic and systematic implications. J Vert Paleontol 16(3): 432-444. 1996), Yixian and Jiufotang Formations (e.g., Wang and Zhou 2006WANG XL and ZHOU ZH. 2006. Pterosaur assemblages of the Jehol Biota and their implication for the Early Cretaceous pterosaur radiation. Geol J 41(3-4): 405-418.), Crato and Romualdo formations (e.g., MaiseyMAISEY JG. 1991. Santana Fossils: An Illustrated Atlas. Neptune City: T.F.H. Publications, 459 p. 1991, Kellner 2006, UnwinUNWIN DM and MARTILL DM. 2007. Pterosaurs from the Crato Formation. In: Martill DM, Bechly G and Loveridge RF (Eds), The Crato fossil beds of Brazil: window into an ancient world, Cambridge: Cambridge University Press, p. 475-524. and Martill 2007, VilaVILA NOVA BC, SARAIVA AAF, MOREIRA JKR and SAYÃO JM. 2011. Controlled excavations in the Romualdo Formation lagerstätte (Araripe Basin, Brazil) and pterosaur diversity: remarks based on new findings. Palaios 26(3): 173-179. Nova et al. 2011), Cambridge Greensand (e.g., Unwin 2001UNWIN DM. 2001. An overview of the pterosaur assemblage from the Cambridge Greensand (Cretaceous) of Eastern England. Mitt Mus Natkd Berl, Geowiss Reihe 4: 189-221., Rodrigues and Kellner 2013RODRIGUES T and KELLNER AWA. 2013. Taxonomic review of the Ornithocheirus complex (Pterosauria) from the Cretaceous of England. ZooKeys (Monographs) 308: 1-112.), and Niobrara Chalk (Bennett 2001, 2003BENNETT SC. 2003. New crested specimens of the Late Cretaceous pterosaur Nyctosaurus. Pal Zeit 77: 61-75., Kellner 2010). However, the lack of stratigraphic control and the absence of specimens showing more than one individual preserved in close association, conclusively demonstrating that different pterosaurs were recovered from the exact same level within these deposits, is a problem that has hampered several biological questions regarding these flying reptiles (e.g., Kellner 2010, Wang et al. 2014).
Still considering taphonomy, the number of elements referable to Caiuajara by far outnumber all others. There are well over 100 individuals identified from this small pterosaur and although some specimens clearly show the orientation of elements indicating the action of water currents, there is no specific predominance of one bone over another. Only pelvic elements are scarcer, to which there is no particular explanation.
The second more common fossil is Vespersaurus, known by at least 12 individuals (based on stratigraphy and number of elements of the hind limb). Contrary to Caiuajara, most specimens of this dinosaur recovered so far belong to the hind limb (including feet) and the pelvis, with rare remains of the skull and arms. Metatarsals II and IV, which in the original description were described based on pictures (Langer et al. 2019) indicating that the specimen is lost or was destroyed, are very compressed lateromedially (Fig. 13). There are also many caudal vertebrae, several articulated (Figs. 11, 12). The amount of elements of Vespersaurus paranaensis that occurs in three bonebeds, suggests that this species might have been gregarious.
Bones of Keresdrakon vilsoni are the rarest of the three species. Referring isolated bones to a specific taxon can be difficult, as well as distinguishing diagnostic from ontogenetic features, which is a problem not restricted to pterosaurs (e.g., MüllerMÜLLER RT. 2017. Are the dinosauromorph femora from the Upper Triassic of Hayden Quarry (New Mexico) three stages in a growth series of a single taxon? An Acad Bras Cienc 89: 835-839. 2017). Due to the morphological similarity of postcranial elements of pterosaur groups in general (e.g., Kellner 2003, Unwin 2003UNWIN DM. 2003. On the phylogeny and evolutionary history of pterosaurs. In: Buffetaut E and Mazin JM (Eds), Evolution and palaeobiology of pterosaurs, London: Geological Society, Special Publications, 139-190 (Geol Soc London Spec Pub 217)., VilaVILA NOVA BC, SAYÃO JM, LANGER MC and KELLNER AWA. 2015. Comments on the cervical vertebrae of the Tapejaridae (Pterosauria, Pterodactyloidea) with description of new specimens. Hist Biol 27: 770-780. Nova et al. 2015), it is possible that difficulties might arise in separating larger individuals (sub-adults) of Caiuajara dobruskii from smaller ones (juveniles) of Keresdrakon vilsoni. The anatomical differences in the cranial anatomy between both pterosaurs of the 'cemitério dos pterossauros' site are striking, but the main differences in the postcranial skeleton are primarily restricted to the presence of pneumatic foramina. While Keresdrakon vilsoni possesses an additional pneumatic foramen on the ventral surface at the anterior half of the proximal articulation of the first wing phalanx (absent in Caiuajara dobruskii), it lacks a pneumatic foramen at the dorsal surface of the proximal articulation of the humerus (present in tapejarine pterosaurs, including Caiuajara). Both species show the lateral surface of the mid-cervical vertebrae pierced by a pneumatic foramen (sometimes two in Caiuajara), which in Keresdrakon is much larger. Another difference is the strongly asymmetrical sternal articulation of the coracoid in Keresdrakon vilsoni, not observed in several coracoids recovered from Caiuajara dobruskii. Nonetheless, the minimum number of Keresdrakon individuals recorded so far is three. Based on the distribution of the bones throughout the section the actual number must be higher. Despite the small sample, no preservation bias of any particular portion of the skeleton is detectable, with cranial and postcranial elements being recovered. There is also no specific taphonomic reason for the comparatively low number of preserved elements of this pterosaur, which might have been the least represented of the three species that occurred in this environment (not including the lizard Gueragama). Based on the present number of specimens, Keresdrakon vilsoni might have had a solitary behavior and was not gregarious as inferred for the dinosaur and Caiuajara.
Lastly, the lizard Gueragama sulamericana, known from two fragmentary specimens only (Simões et al. 2015), is the sole other taxon recognized so far from the 'cemitério dos pterossauros' site. Even though the exact stratigraphic level from where this material came from is unknown, this lizard was most likely part of the same paleobiological community of the dinosaur and the pterosaurs.
Sedimentological data supports the interpretation that the Goio-Erê Formation was formed in a desert environment with interdunal wetland (Milani et al. 2007, FernandesFERNANDES LA, SEDOR FA, SILVA RC, SILVA LR, AZEVEDO AA and SIQUEIRA AG. 2009. Icnofósseis da Usina Porto Primavera, SP: rastros de dinossauros e de mamíferos em rochas do deserto neocretáceo. In: Winge M, Schobbenhaus C, Souza CRG, Fernandes ACS, Bebert-Born M, Queiroz ET and Campo DA (Eds), Sítios Geológicos e Paleontológicos do Brasil, Brasília: CPRM - Serviço Geológico do Brasil p. 479-488. et al. 2009). Whilst Caiuajara dobruskii most likely was a frugivore, a feeding behavior suggested for tapejarines in general (Kellner 1989, Wellnhofer and Kellner 1991WELLNHOFER P and KELLNER AWA. 1991. The skull of Tapejara wellnhoferi Kellner (Reptilia, Pterosauria) from the Lower Cretaceous Santana Formation of the Araripe Basin, Northeastern Brazil. Mitt Bayer Staatsslg Paläont Hist Geol 31: 89-106., Wang and Zhou 2003a, Vullo et al. 2012), this does not seem to have been the case of Keresdrakon vilsoni. Taken into account that deserts tend to have limited resources, niche partitioning concerning these pterosaurs would be feasible, particularly if they were sympatric, which is considered here. Therefore, we can infer that Keresdrakon vilsoni might have behaved as an opportunistic predator or a scavenger (Fig. 17). According to previous studies, a long and laterally flattened rostrum like the one of Keresdrakon is appropriate to support low bite pressures restricted to the jaw tips and the straight jaw margins, as has been proposed for azhdarchids (WittonWITTON MP and NAISH D. 2008. A reappraisal of azhdarchid pterosaur functional morphology and paleoecology. PloS One 3(5): e2271. and Naish 2008). Therefore, subjugation of larger prey, such as the dinosaur Vespersaurus, might have been more difficult for this pterosaur despite its larger size, which could better handle smaller or immobile food items such as carcasses. This feeding behavior is observed in the extant marabou stork (KahlKAHL MP. 1966. Comparative ethology of the Ciconiidae. Part 1. The Marabou Stork, Leptoptilos crumeniferus. Behaviour 27(1/2): 76-106. 1966) Leptoptilos crumenifer (Lesson, 1831). The feeding preference of this bird comprises mainly carrion and scraps, but it will opportunistically eat almost any animal matter that it can swallow, including small mammals and reptiles such as crocodile hatchlings and eggs (HancockHANCOCK JA, KUSHLAN JA and KAHL MP. 1992. Storks, ibises and spoonbills of the World. Princeton: Princeton University Press, 385 p. et al. 1992). Considering the little diverse fossiliferous record of the Caiuá Group at the 'cemitério dos pterossauros' site, Keresdrakon could probably prey on small lizards such as Gueragama sulamericana. According to Simões et al. (2015) the latter is assigned to have presented the same habit as that of modern agamid lizards in arid regions, which is to live in burrows to avoid the arid weather. Additionally, Keresdrakon might have also feed on eggs, younglings or even juveniles of Caiuajara dobruskii, which was the predominant species in this region during part of the Cretaceous.
Reconstruction of the paleoenvironment showing the possible interaction of the vertebrate fauna recovered from the 'cemitério dos pterossauros' site. See text for details. Artwork by Maurilio Oliveira.
Regarding carrion feeding hypothesis, so far there have been only Caiuajara and Vespersaurus coming from these layers. Although admittedly speculative, it is most likely that Keresdrakon might have fed on their carcasses, particularly on that of the dinosaur. As for Vespersaurus, its primary food resource might have been Caiuajara, which is found in large numbers in this deposit.
The lateral surface of the rostral end of the skull of CP.V 2068 is quite rough (unlike the mild condition observed in CP.V 2069), suggesting that this specimen could have been exposed before burial. The roughness aspect of the bone surface is considered as an indicator of ontogeny in pterosaurs (Bennett 1993, Steel 2008). In the Pteranodon specimens, this roughness (grain pattern for Bennett 1993) is only present in some specimens with non-ossified epiphyses, reinforcing the hypothesis that this characteristic is present in juveniles. Both samples analyzed for histological studies in Keresdrakon had open vascular canals in the periosteal region (CP.V 2077 and CP.V 2374), which is also an osteohistological characteristic pointing to juveniles (de Ricqlès et al. 2000, Steel 2008). Therefore, features such as open canals in the periosteum and the roughness or granulation patterns endorse the interpretation of these specimens between ontogenetic stages OS2 and OS3 of Kellner (2015).
The few elements of Keresdrakon vilsoni analyzed show a distinct osteohistological pattern from that of Caiuajara dobruskii. The latter present a highly vascularized plexiform tissue (SayãoSAYÃO JM, BANTIM RAM, WEINSCHÜTZ LC, ARAUJO EV and KELLNER AWA. 2018. Osteohistological characterization as a support of Caiuajra dobruskii population in the Cenomanian of Brazil. Flugsaurier 2018: the 6th international symposium on pterosaurs. Los Angeles, USA. Abstracts, p. 77-80. et al. 2018), absent in the bones of Keresdrakon studied here, which have a low vascularized cortex, with few vascular canals connected (anastomosed). Although both taxa present fibrolamellar complexes, the plexiform tissue of Caiuajara has additional radial connections between vascular canals. Both types of histological features are indicative of fast growth rates (Chinsamy-TuranCHINSAMY-TURAN A. 2005. The Microstructure of Dinosaur Bone. The Johns Hopkins University Press, 195 p. 2005), although the plexiform tissue is considered to have slightly denser vascular canals than those of the laminar tissue (Francillon-VieillotFRANCILLON-VIEILLOT H, DE BUFFRÉNIL V, CASTANET J, GÉRAUDIE J, MEUNIER FJ, SIRE JY, ZYLBERBERG L and DE RICQLÈS A. 1990. Microstructure and mineralization of vertebrate skeletal tissues. In: Carter JG (Ed), Skeletal biomineralization: patterns, processes and evolutionary trends, New York: vol. I. Van Nostrand Reinhold, p. 471-530. et al. 1990). Thus, it is not possible to establish differences in growth rates between the specimens analyzed of both pterosaur species based only on their vascular arrangement. In any case, the bones studied here indicate high growth rates for both species.
Regarding bone maturity, Caiuajara shows some signs of remodeling (Sayão et al. 2018) represented by secondary osteons in the fourth metacarpal (CP.V 2061), which are absent in both Keresdrakon samples. Although this difference may be related to a sampling problem, it suggests that the population of Caiuajara may present individuals slightly more ontogenetically developed than Keresdrakon. Fused scapulocoracoids and several first wing phalanx with the extensor tendon process fused agrees with this supposition. Further osteohistological analyzes are needed to support this observation.
Comparisons with Tupuxuara leonardii (Iwaki specimen) and Caupedactylus (Kellner 2013), particularly regarding the humerus length, suggest that Keresdrakon vilsoni had a wingspan of approximately 3 m, quite larger than that of Caiuajara dobruskii (Manzig et al. 2014: 0.65 to 2.35 m). Taking into account that they are young individuals this proportion could be even higher.
CONCLUSIONS
The coexistence of fossil vertebrates, including pterosaurs, are hard to be proven in the fossil record, specially cases of sympatry. Regarding pterosaurs, there must have been several places along deep time where closely related species might have overlapped and shared similar geographic distribution. The challenge is to find direct evidences of this. The 'cemitério dos pterossauros' site represents the co-occurrence of distinct pterosaur species in the same bone beds, showing that Keresdrakon and Caiuajara were coeval. This site also shows that the dinosaur Vespersaurus paranaensis shared this ancient desert environment with these two flying reptiles. While Caiuajara, despite being small and therefore potentially having a more fragile skeleton, by far outnumber the other taxa and is one of the few examples in the fossil record that might argue for gregarious behavior in pterosaurs, Keresdrakon vilsoni was scarcer and apparently represents a species with solitary behavior. We advocate that both specialized in different feeding items of this most likely depauperate environment. Caiuajara is regarded as being frugivorous and Keresdrakon might have been an opportunistic predator or a scavenger feeding at small animals or carcasses such as that of Vespersaurus. The latter might have made a living by hunting individuals of Caiuajara dobruskii that were abundant. All three were part of a paleobiological community that existed in this region during part of the Cretaceous.
As has been pointed out before (Kellner 2012), the outcrops in the region of Cruzeiro do Oeste, more specifically deposits from the Caiuá Group as the Goio-Erê formation, might turn out to become the `Brazilian Mongolia´ in terms of fossil vertebrates. The hundreds of fossils from two pterosaur species and one theropod dinosaur, as well as the occurrence of a lizard, endorse this potential. It is also likely that the future might show additional taxonomic groups like mammals and other reptiles. More fieldwork and careful collection of specimens might contribute to a better understanding of the ecosystem of ancient Cretaceous deserts that, despite its general depauperate conditions, had areas where life could prosper.
ACKNOWLEGMENTS
We would like to thank Valter Pereira da Rocha (former Mayor of Municipality of Cruzeiro do Oeste), João Gustavo Dobruski and his family, Maristela Sanches Morcelli and Neurides de Oliveira Martins, all residents of Cruzeiro do Oeste, for their help during fieldwork. Solange Salete Sprandel da Silva, Luciano Bendlin, and Gabriel Bonetto Bampi from the Universidade do Contestado are thanked for supporting the research at CENPALEO. Vilson Greinert is acknowledged for the preparation of several specimens housed in CENPALEO and in the Municipality of Cruzeiro do Oeste that greatly contributed to several studies of fossil vertebrates from this region. Kamila Bandeira (Museu Nacional/UFRJ) has helped in the discussion of Greek mythology. Maurilio Oliveira is thanked for the reconstruction of the paleoenvironment of the 'cemitério dos pterossauros' site. AWAK would like to acknowledge Neurides de Oliveira Martins for inviting him to teach the fossil vertebrate course in August 2016 at Cruzeiro do Oeste. This course trained several employees of the municipality as well as teachers and students from local universities such as the Universidade Estadual do Maringá (Paraná State) and was the basis of a fossil preparation lab in the city. Lastly, we would like to acknowledge three anonymous reviewers for their comments and suggestions that greatly improved the final version of this paper. This study was partially financed by the Fundação de Desenvolvimento Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ #E-26/202.893/2015 to AWAK), the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq #420687/2016-5 and #313461/2018-0 to AWAK; #140789/2016-2 to BH, #311715/2017-6 to JMS), the Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico (FUNCAP #BMD-0124-00302.01.01/19 to RAMB) and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001 (CAPES #88887.162865/2018-00 to RAMB).
REFERENCES
- AIRES ASS, KELLNER AWA, MÜLLER RT, DA SILVA LR, PACHECO CP and DIAS-DA-SILVA S. 2013. New postcranial elements of the Thalassodrominae (Pterodactyloidea, Tapejaridae) from the Romualdo Formation (Aptian-Albian), Santana Group, Araripe Basin, Brazil. Palaeontology 57: 343-355.
- ANDRADE RCLP, BANTIM RAM, LIMA FJ, CAMPOS LS, ELEUTÉRIO LHS and SAYÃO JM. 2015. New data about the presence and absence of the external fundamental system in Archosaurs. Cad Cult Ciênc 14(1): 200-211.
- ANDRES B, CLARK J and XU X. 2014. The earliest pterodactyloid and the origin of the group. Curr Biol 24(9): 1011-1016.
- ANDRES B and JI Q. 2008. A new pterosaur from the Liaoning Province of China, the phylogeny of the Pterodactyloidea, and the convergence in their cervical vertebrae. Palaeontology 51: 453-469.
- BANDEIRA KLN, BRUM AS, PÊGAS RV, CIDADE GM, HOLGADO B, CIDADE A and SOUZA RG. 2018. The Baurusuchidae vs Theropoda record in the Bauru Group (Upper Cretaceous, Brazil): a taphonomic perspective. J Iber Geol 44: 25-54.
- BANTIM RAM, SARAIVA AAF, OLIVEIRA G and SAYÃO JM. 2014. A new toothed pterosaur (Pterodactyloidea: Anhangueridae) from the Early Cretaceous Romualdo Formation, NE Brazil. Zootaxa 3869: 201-223.
- BARRETT PM, BUTLER RJ, EDWARDS NP and MILNER AR. 2008. Pterosaur distribution in time and space: an atlas. Zitteliana B 28: 61-108.
- BASILICI G, SGARBI GN and DAL'BÓ PFF. 2012. A Sub-bacia Bauru: um sistema continental entre deserto e cerrado. In: Hasui Y, Carneiro CDR, Almeida FFM and Bartorelli A (Eds), Geologia do Brasil, São Paulo: Beca, p. 520-543.
- BATEZELLI A. 2010. Arcabouço tectono-estratigráfico e evolução das Bacias Caiuá e Bauru no Sudeste brasileiro. Revista Brasileira de Geociências 40: 265-285.
- BATEZELLI A. 2015. Continental systems tracts of the Brazilian Cretaceous Bauru Basin and their relationship with the tectonic and climatic evolution of South America. Basin Research 27: 1-25.
- BEHRENSMEYER AK, KIDWELL SM and GASTALDO RA. 2000. Taphonomy and paleobiology. Paleobiology 26: 103-147.
- BENNETT SC. 1996. Year-classes of pterosaurs from the Solnhofen Limestone of Germany: taxonomic and systematic implications. J Vert Paleontol 16(3): 432-444.
- BENNETT SC. 2001. The osteology and functional morphology of the Late Cretaceous pterosaur Pteranodon part 1 - General description and osteology. Palaeontographica 260: 1-112.
- BENNETT SC. 2003. New crested specimens of the Late Cretaceous pterosaur Nyctosaurus. Pal Zeit 77: 61-75.
- BRUSATTE SL, CANDEIRO CRA and SIMBRAS FM. 2017. The last dinosaurs of Brazil: The Bauru Group and its implication for the end-Cretaceous mass extinction. An Acad Bras Cienc 89: 1465-1485.
- CABREIRA SF et al. 2016. A unique Late Triassic dinosauromorph assemblage reveals dinosaur ancestral anatomy and diet. Curr Biol 26: 1-6.
- CAI Z and WEI F. 1994. On a new pterosaur (Zhejiangopterus linhaiensis gen. et sp. nov.) from Upper Cretaceous in Linhai, Zhejiang, China. Vert Palas 32: 181-194.
- CHAVARRÍA-ARELLANO ML, SIMÕES TR and MONTELLANO-BALLESTEROS MM. 2018. New data on the Late Cretaceous lizard Dicothodon bajaensis (Squamata, Borioteiioidea) from Baja California, Mexico reveals an unusual tooth replacement pattern in squamates. An Acad Bras Cienc 90: 2781-2795.
- CHENG X, JIANG X, WANG X and KELLNER AWA. 2017. Premaxillary crest variation withing the Wukongopteridae (Reptilia, Pterosauria) and comments on cranial structures in pterosaurs. An Acad Bras Cienc 89: 119-130.
- CHIAPPE LM, RIVAROLA D, ROMERO E, DAVILA S and CODORNIÚ L. 1998. Recent advances in the paleontology of the Lower Cretaceous Lagarcito Formation (Parque Nacional Sierra de Las Quijadas, San Luis, Argentina). In: Lucas SG, Kirkland JI and Estep JW (Eds), Lower and Middle Cretaceous Terrestrial Ecosystems, Albuquerque: New Mexico Museum of Natural History and Science, p. 187-192.
- CHINSAMY-TURAN A. 2005. The Microstructure of Dinosaur Bone. The Johns Hopkins University Press, 195 p.
- CHOUDHARY S, CHOUDHURY BC and GOPI GV. 2018. Spatio-temporal partitioning between two sympatric crocodilians (Gavialis gangeticus & Crocodylus palustris) in Katarniaghat Wildlife Sanctuary, India. Aquatic Conserv: Mar Freshw Ecosyst 2018: 1-10.
- COHEN KM, FINNEY SM, GIBBARD PL and FAN JX. 2013. The ICS International Chronostratigraphic Chart. Episodes 36: 199-204.
- DALLA VECCHIA FM. 2014. Gli pterosauri triassici. Udine: Pubblicazione del Museo Friulano di Storia Naturale, 319 p.
- DE RICQLÈS A, PADIAN K, HORNER JR and FRANCILLON-VIEILLOT H. 2000. Palaeohistology of the bones of pterosaurs (Reptilia: Archosauria): anatomy, ontogeny, and biomechanical implications. Zool J Linn Soc 129: 349-385.
- DIAMOND JM. 1975. Assembly of species communities. In: Cody ML and Diamond JM (Eds), Ecology and evolution of Communities, Cambridge: Harvard University Press, p. 342-444.
- EATON GF. 1910. Osteology of Pteranodon. Memoirs of the Connecticut Academy of Arts and Sciences 2: 1-38.
- EBERTH DA, BRINKMAN DB, CHEN PJ, YUAN FT, WU SZ, LI G and CHENG XS. 2001. Sequence stratigraphy, paleoclimate patterns, and vertebrate fossil preservation in Jurassic-Cretaceous strata of the Junggar Basin, Xinjiang Autonomous Region, People’s Republic of China. Can J Earth Sci 38: 1627-1644.
- ECK K, ELGIN RA and FREY E. 2011. On the osteology of Tapejara wellnhoferi Kellner 1989 and the first occurrence of multiple specimen assemblage from the Santana Formation, Araripe Basin, NE-Brazil. Swiss J Palaeontol 130: 277-296.
- ELEUTÉRIO LHS, BANTIM RAM, LIMA FJ, ANDRADE RCLP, SARAIVA AAF, KELLNER AWA and SAYÃO JM. 2015. Biomechanical and physiological influences on the osteohistological deposition of Anhangueria (Pterosauria, Pterodactyloidea). Rev Bras Paleo 18(3): 403-412.
- FERNANDES LA. 2004. Mapa litoestratigráfico da parte oriental da Bacia Bauru (PR, SP, MG), escala 1: 1.000.000. Boletim Paranaense de Geociências 55.
- FERNANDES LA and COIMBRA AM. 2000. Revisão estratigráfica da parte oriental da Bacia Bauru (Neocretáceo). Revista Brasileira de Geociências 30: 717-728.
- FERNANDES LA, SEDOR FA, SILVA RC, SILVA LR, AZEVEDO AA and SIQUEIRA AG. 2009. Icnofósseis da Usina Porto Primavera, SP: rastros de dinossauros e de mamíferos em rochas do deserto neocretáceo. In: Winge M, Schobbenhaus C, Souza CRG, Fernandes ACS, Bebert-Born M, Queiroz ET and Campo DA (Eds), Sítios Geológicos e Paleontológicos do Brasil, Brasília: CPRM - Serviço Geológico do Brasil p. 479-488.
- FRANCILLON-VIEILLOT H, DE BUFFRÉNIL V, CASTANET J, GÉRAUDIE J, MEUNIER FJ, SIRE JY, ZYLBERBERG L and DE RICQLÈS A. 1990. Microstructure and mineralization of vertebrate skeletal tissues. In: Carter JG (Ed), Skeletal biomineralization: patterns, processes and evolutionary trends, New York: vol. I. Van Nostrand Reinhold, p. 471-530.
- GOLOBOFF PA and CATALANO SA. 2016. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics 32(3): 221-238.
- HAMMER WR and HICKERSON WJ. 1994. A crested theropod dinosaur from Antarctica. Science 264: 828-830.
- HANCOCK JA, KUSHLAN JA and KAHL MP. 1992. Storks, ibises and spoonbills of the World. Princeton: Princeton University Press, 385 p.
- HOLGADO B, PÊGAS RV, CANUDO JI, FORTUNY J, RODRIGUES T, COMPANY J and KELLNER AWA. 2019. On a new crested pterodactyloid from the Early Cretaceous of the Iberian Peninsula and the radiation of the clade Anhangueria. Sci Rep 9: 4940.
- HOWSE SCB. 1986. On the cervical vertebrae of the Pterodactyloidea (Reptilia: Archosauria). Zool J Linn Soc 88: 307-328.
- KAHL MP. 1966. Comparative ethology of the Ciconiidae. Part 1. The Marabou Stork, Leptoptilos crumeniferus. Behaviour 27(1/2): 76-106.
- KELLNER AWA. 1989. A new edentate pterosaur of the Lower Cretaceous from the Araripe Basin, Northeast Brazil. An Acad Bras Cienc 61: 439-446.
- KELLNER AWA. 2003. Pterosaur phylogeny and comments on the evolutionary history of the group. In: Buffetaut E and Mazin JM (Eds), Evolution and palaeobiology of pterosaurs, London: Geological Society, Special Publications, 105-137 (Geol Soc London Spec Pub 217).
- KELLNER AWA. 2006. Pterossauros: os senhores dos ceús do Brasil. Rio de Janeiro: Vieira & Lent, 175 p.
- KELLNER AWA. 2010. Comments on the Pteranodontidae (Pterosauria, Pterodactyloidea) with the description of two new species. An Acad Bras Cienc 82: 1063-1084.
- KELLNER AWA. 2012. Pterossauros: os primeiros répteis alados. In: Hasui Y, Carneiro CDR, Almeida FFM and Bartorelli A (Eds), Geologia do Brasil, São Paulo: Beca, p. 735-746.
- KELLNER AWA. 2013. A new unusual tapejarid (Pterosauria, Pterodactyloidea) from the Early Cretaceous Romualdo Formation, Araripe Basin, Brazil. Earth Env Sci T R So 103: 1-14.
- KELLNER AWA. 2015. Comments on Triassic perosaurs with discussion about ontogeny and description of new taxa. An Acad Bras Cienc 87: 669-689.
- KELLNER AWA. 2017. Rebuttal of Martin-Silverstone et al. 2017, 'Reassessment of Dawndraco kanzai Kellner 2010 and reassignment of the type specimen to Pteranodon sternbergi Harksen, 1966'. Vertebrate Anatomy Morphology Palaeontology 3: 81-89. DOI 10.18435/B54D49.
- KELLNER AWA and CALVO J. 2017. New azhdarchoid pterosaur (Pterosauria, Pterodactyloidea) with an unusual lower jaw from the Portezuelo Formation (Upper Cretaceous), Neuquén Group, Patagonia, Argentina. An Acad Bras Cienc 89: 2003-2012.
- KELLNER AWA and CAMPOS DA. 2002. The function of the cranial crest and jaws of a unique pterosaur from the Early Cretaceous of Brazil. Science 297: 389-392.
- KELLNER AWA and CAMPOS DA. 2007. Short note on the ingroup relationships of the Tapejaridae (Pterosauria, Pterodactyloidea). Bol Mus Nac N S Geol Rio de Janeiro 75: 1-14.
- KELLNER AWA, CAMPOS DA, SAYÃO JM, SARAIVA AAF, RODRIGUES T, OLIVEIRA G, CRUZ LA, COSTA FR, SILVA HP and FERREIRA JS. 2013. The largest flying reptile from Gondwana: a new specimen of Tropeognathus cf. T. mesembrinus Wellnhofer, 1987 (Pterodactyloidea, Anhangueridae) and other large pterosaurs from the Romualdo Formation, Lower Cretaceous, Brazil. An Acad Bras Cienc 85: 113-135.
- KELLNER AWA and LANGSTON W. 1996. Cranial remains of Quetzalcoatlus (Pterosauria, Azhdarchidae) from the Late Cretaceous sediments of Big Bend National Park, Texas. J Vert Paleontol 16(2): 222-231.
- LAMM ET. 2013. Bone histology of fossil tetrapods. In: Padian K and Lamm ET (Eds), Preparation and sectioning of specimens, Oakland: University of California Press, p. 55-160.
- LANGER MC et al. 2019. A new desert-dwelling dinosaur (Theropoda, Noasaurinae) from the Cretaceous of south Brazil. Sci Rep 9: 9379.
- LEAL M and FLEISHMAN LJ. 2002. Evidence for habitat partitioning based on adaptation to environmental light in a pair of sympatric lizard species. Proc Royal Soc B 269: 351-359.
- LEAL MEC, PÊGAS RV, BONDE N and KELLNER AWA. 2018. Cervical vertebrae of an enigmatic pterosaur from the Crato Formation (Lower Cretaceous, Araripe Basin, NE Brazil). In: Hone DWE, Witton MP and Martill DM (Eds), New perspectives on pterosaur palaeobiology, London: Geological Society, Special Publications, 195-208 (Geol Soc London Spec Pub 455).
- LONGRICH NR, MARTILL DM and ANDRES B. 2018. Late Maastrichtian pterosaur from North Africa and mass extinctions of Pterosauria at the Cretaceous-Paleogene boundary. PLoS Biol 16: e2001663.
- LUISELLI L. 2006. Resource partitioning in the communities of terrestrial turtles: a review of the evidences. Rev Ecol-Terre Vie 61(4): 353-365.
- LÜ JC, JIN XS, UNWIN DM, ZHAO LJ, AZUMA Y and JI Q. 2006. A new species of Huaxiapterus (Pterosauria: Pterodactyloidea) from the Lower Cretaceous of western Liaoning, China with comments on the systematics of tapejarid pterosaurs. Acta Geol Sin 80: 315-326.
- LÜ JC, UNWIN DM, XU L and ZHANG XL. 2008. A new azhdarchoid pterosaur from the Lower Cretaceous of China and its implications for pterosaur phylogeny and evolution. Naturwissenschaften 95: 891-897.
- MAGNUSSON WE. 1985. Habitat selection, parasites, and injuries in Amazonian crocodiles. Amazoniana 9: 193-204.
- MAISEY JG. 1991. Santana Fossils: An Illustrated Atlas. Neptune City: T.F.H. Publications, 459 p.
- MANZIG PC, KELLNER AWA, WEINSCHÜTZ LC, FRAGOSO CE, VEGA CS, GUIMARÃES GB, GODOY LC, LICCARDO A, RICETTI JHC and MOURA CC. 2014. Discovery of a rare pterosaur bone bed in a Cretaceous Desert with insights on ontogeny and behavior of flying reptiles. PloS One 9(8): e100005.
- MANZIG PC and WEINSCHÜTZ LC. 2012. Museus e fósseis da Região Sul do Brasil: uma experiência visual com a paleontologia. Curitiba: Germânica, 306 p.
- MAYR E. 1942. Systematics and the origin of species. New York: Columbia University Press, 334 p.
- MILANI EJ, MELO JH, SOUZA PA, FERNANDES LA and FRANÇA AB. 2007. Bacia do Paraná. Bol Geoc Petrobras 15: 265-287.
- MÜLLER RT. 2017. Are the dinosauromorph femora from the Upper Triassic of Hayden Quarry (New Mexico) three stages in a growth series of a single taxon? An Acad Bras Cienc 89: 835-839.
- NAISH D, SIMPSON M and DYKE G. 2013. A new small-bodied azhdarchoid pterosaur from the Lower Cretaceous of England and its implications for pterosaur Anatomy, diversity and phylogeny. PloS One 8(3): e58451.
- NESOV LA. 1984. Upper Cretaceous pterosaurs and birds from central Asia. Paleontol J 1984(1): 38-49.
- PADIAN K, HORNER JR and DE RICQLÈS A. 2004. Growth in small dinosaurs and pterosaurs: The evolution of archosaurian growth strategies. J Vert Paleont 24: 555-571.
- PÊGAS RV, COSTA FR and KELLNER AWA. 2018. New information on the osteology and a taxonomic revision of the genus Thalassodromeus (Pterodactyloidea, Tapejaridae, Thalassodrominae). J Vert Paleontol 38(2): e1443273.
- PÊGAS RV, LEAL MEC and KELLNER AWA. 2016. A basal tapejarine (Pterosauria; Pterodactyloidea; Tapejaridae) from the Crato Formation, Early Cretaceous of Brazil. PloS One 11(9): e0162692.
- PIGOT AL and TOBIAS JA. 2015. Dispersal and the transition to sympatry in vertebrates. Proc Royal Soc B 282(1799): 1-8.
- PINHEIRO FL, FORTIER DC, SCHULTZ CL, ANDRADE JAFG and BANTIM RAM. 2011. New information on the pterosaur Tupandactylus imperator, with comments on the relationships of Tapejaridae. Acta Palaeontol Pol 56: 567-580.
- PINHEIRO FL and RODRIGUES T. 2017. Anhanguera taxonomy revisited: is our understanding of Santana Group pterosaur diversity biased by poor biological and stratigraphic control? PeerJ 5: e3285.
- PRONDVAI E, STEIN K, ÖSI A and SANDER MP. 2012. Life history of Rhamphorhynchus inferred from bone histology and the diversity of Pterosaurian growth strategies. PloS ONE 7: e31392.
- RAO RJ and CHOUDHURY BC. 1990. Sympatric distribution of gharial and mugger in India. J Bombay Nat Hist Soc 89: 312-315.
- RIFF D, SOUZA RG, CIDADE GM, MARTINELI AG and SOUZA FILHO JP. 2012. Crocodilomorfos: a maior diversidade de répteis fósseis do Brasil. Terrae 9: 12-40.
- RIVAS LR. 1964. A reinterpretation of the concepts “sympatric” and “allopatric” with proposal of the additional terms “syntopic” and “allotopic”. Syst Biol 13: 42-43.
- RODRIGUES T and KELLNER AWA. 2013. Taxonomic review of the Ornithocheirus complex (Pterosauria) from the Cretaceous of England. ZooKeys (Monographs) 308: 1-112.
- RODRIGUES T, KELLNER AWA, MADER BJ and RUSSELL D. 2011. New pterosaur specimens from the Kem Kem beds (Upper Cretaceous, Cenomanian) of Morocco. Riv Ital Paleontol S 117(1): 149-160.
- ROGERS RR and KIDWELL SM. 2000. Associations of vertebrate skeletal concentrations and discontinuity surfaces in continental and shallow marine records: a test in the Cretaceous of Montana. J Geol 108: 131-154.
- SARAIVA AAF, HESSEL MH, GUERRA NC and FARA E. 2008. Concreções calcárias da Formação Santana, Bacia do Araripe: uma proposta de classificação. Estudos Geológicos 17(1): 40-57.
- SAYÃO JM. 2003. Histovariability in bones of two pterodactyloid pterosaurs from the Santana Formation, Araripe Basin, Brazil: preliminary results. In: Buffetaut E and Mazin JM (Eds), Evolution and palaeobiology of pterosaurs, London: Geological Society, Special Publications, p. 335-342 (Geol Soc London Spec Pub 217).
- SAYÃO JM, BANTIM RAM, WEINSCHÜTZ LC, ARAUJO EV and KELLNER AWA. 2018. Osteohistological characterization as a support of Caiuajra dobruskii population in the Cenomanian of Brazil. Flugsaurier 2018: the 6th international symposium on pterosaurs. Los Angeles, USA. Abstracts, p. 77-80.
- SIMÕES TR, WILNER E, CALDWELL MW, WEINSCHÜTZ LC and KELLNER AWA. 2015. A stem acrodontan lizard in the Cretaceous of Brazil, revises early lizard evolution in Gondwana. Nat Commun 6: 8149.
- SOLER M and MØLLER AP. 1990. Duration of sympatry and coevolution between the great spotted cuckoo and its magpie host. Nature 343: 748-750.
- STEEL L. 2008. The palaeohistology of pterosaur bone: an overview. Zitteliana B 28: 109-125.
- TISCHLINGER H and FREY E. 2002. Ein Rhamphorhynchus (Pterosauria, Reptilia) mit ungewöhnlicher Flughauterhaltung aus dem Solnhofener Plattenkalk. Archaeopteryx 20: 1-20.
- UNWIN DM. 2001. An overview of the pterosaur assemblage from the Cambridge Greensand (Cretaceous) of Eastern England. Mitt Mus Natkd Berl, Geowiss Reihe 4: 189-221.
- UNWIN DM. 2003. On the phylogeny and evolutionary history of pterosaurs. In: Buffetaut E and Mazin JM (Eds), Evolution and palaeobiology of pterosaurs, London: Geological Society, Special Publications, 139-190 (Geol Soc London Spec Pub 217).
- UNWIN DM and MARTILL DM. 2007. Pterosaurs from the Crato Formation. In: Martill DM, Bechly G and Loveridge RF (Eds), The Crato fossil beds of Brazil: window into an ancient world, Cambridge: Cambridge University Press, p. 475-524.
- UPCHURCH P, ANDRES B, BUTLER RJ and BARRETT PM. 2015. An analysis of pterosaurian biogeography: implications for the evolutionary history and fossil record quality of the first flying vertebrates. Hist Biol 27: 697-717.
- VAN VALEN L. 1976. Ecological species, multispecies, and oaks. Taxon 25(2/3): 233-239.
- VILA NOVA BC, SARAIVA AAF, MOREIRA JKR and SAYÃO JM. 2011. Controlled excavations in the Romualdo Formation lagerstätte (Araripe Basin, Brazil) and pterosaur diversity: remarks based on new findings. Palaios 26(3): 173-179.
- VILA NOVA BC, SAYÃO JM, LANGER MC and KELLNER AWA. 2015. Comments on the cervical vertebrae of the Tapejaridae (Pterosauria, Pterodactyloidea) with description of new specimens. Hist Biol 27: 770-780.
- VREMIR M, KELLNER AWA, NAISH D and DYKE GJ. 2013. A new azhdarchid pterosaur from the Late Cretaceous of the Transylvanian Basin, Romania: implications for azhdarchid diversity and distribution. PloS One 8(1): e54268.
- VULLO R, MARUGÁN-LOBÓN JS, KELLNER AWA, BUSCALIONI AD, GOMEZ B, DE LA FUENTE M and MORATALLA JJ. 2012. A new crested pterosaur from the Early Cretaceous of Spain: the first European tapejarid (Pterodactyloidea: Azhdarchoidea). PLoS ONE 7: e38900.
- WANG XL et al. 2014. Sexually dimorphic tridimensionally preserved pterosaurs and their eggs from China. Curr Biol 24: 1323-1330.
- WANG XL and ZHOU ZH. 2003a. A new pterosaur (Pterodactyloidea, Tapejaridae) from the Early Cretaceous Jiufotang Formation of western Liaoning, China and its implications for biostratigraphy. Chin Sci Bull 48: 16-23.
- WANG XL and ZHOU ZH. 2003b. Two new pterodactyloid pterosaurs from the Early Cretaceous Jiufotang Formation of western Liaoning, China. Vert Palas 41: 34-41.
- WANG XL and ZHOU ZH. 2006. Pterosaur assemblages of the Jehol Biota and their implication for the Early Cretaceous pterosaur radiation. Geol J 41(3-4): 405-418.
- WANG XL et al. 2017. Egg accumulation with 3D embryos provides insight into the life history of a pterosaur. Science 358: 1197-1201. DOI: 10.1126/science.aan2329.
- WELLNHOFER P. 1975. Die Rhamphorhynchoidea (Pterosauria) der Oberjura-Plattenkalke Süddeutschlands. Teil I: Allgemaine Skelettmorphologie. Palaeontogr Abt A 148: 1-33.
- WELLNHOFER P. 1978. Pterosauria. Handbuch der Paläoherpetologie, Teil 19. Stuttgart: Gustav Fischer Verlag, 82 p.
- WELLNHOFER P. 1985. Neue Pterosaurier aus der Santana-Formation (Apt) der Chapada do Araripe, Brasilien. Palaeontogr Abt A 187: 105-182.
- WELLNHOFER P. 1991. Weitere Pterosanrierfunde aus der Santana-Formation (Apt) der Chapada do Araripe, Brasilien. Palaeontogr Abt A 215: 43-101.
- WELLNHOFER P and KELLNER AWA. 1991. The skull of Tapejara wellnhoferi Kellner (Reptilia, Pterosauria) from the Lower Cretaceous Santana Formation of the Araripe Basin, Northeastern Brazil. Mitt Bayer Staatsslg Paläont Hist Geol 31: 89-106.
- WILD R. 1978. Die Flugsaurier (Reptilia, Pterosauria) aus der Oberen Trias von Cene bei Bergamo, Italien. Boll Soc Paleont It 17(2): 176-256.
- WITTON MP and NAISH D. 2008. A reappraisal of azhdarchid pterosaur functional morphology and paleoecology. PloS One 3(5): e2271.
- YOUNG CC. 1973. Reports of Paleontological Expedition to Sinkiang (II). Pterosaurian Fauna from Wuerho, Sinkiang. Memoirs of the Institute of Vertebrate Palaeontology and Paleoanthropology, Academy Sinica 11: 18-35.
- YRIGOYEN M. 1975. La edad Cretácica del Grupo del Gigante (San Luis) y su relación con cuencas circunvecinas. Actas Congreso Geológico Argentino de Paleontología y Bioestratigrafía 2: 29-56.
- ZAMUDIO KR et al. 2018. Lack of science support fails Brazil. Science 361: 1322-1323.
Publication Dates
-
Publication in this collection
19 Aug 2019 -
Date of issue
2019
History
-
Received
5 July 2019 -
Accepted
6 Aug 2019