Abstracts
PURPOSE: To evaluate the safety, efficacy and predictability after refractive lens exchange with multifocal diffractive aspheric intraocular lens implantation. METHODS: Sixty eyes of 30 patients underwent bilateral implantation with AcrySof® ReSTOR® SN6AD3 intraocular lens with +4.00 D near addition. Patients were divided into myopic and hyperopic groups. Monocular best corrected visual acuity at distance and near and monocular uncorrected visual acuity at distance and near were measured before and 6 months postoperatively. RESULTS: After surgery, uncorrected visual acuity was 0.08 ± 0.15 and 0.11 ± 0.14 logMAR for the myopic and hyperopic groups, respectively (50% and 46.67% of patients had an uncorrected visual acuity of 20/20 or better in the myopic and hyperopic groups, respectively). The safety and efficacy indexes were 1.05 and 0.88 for the myopic and 1.01 and 0.86 for the hyperopic groups at distance vision. Within the myopic group, 20 eyes remained unchanged after the surgery, and 3 gained >2 lines of best corrected visual acuity. For the hyperopic group, 2 eyes lost 2 lines of best corrected visual acuity, 21 did not change, and 3 eyes gained 2 lines. At near vision, the safety and efficacy indexes were 1.23 and 1.17 for the myopic and 1.16 and 1.13 for the hyperopic groups. Best corrected near visual acuity improved after surgery in both groups (from 0.10 logMAR to 0.01 logMAR in the myopic group, and from 0.10 logMAR to 0.04 logMAR in the hyperopic group). CONCLUSIONS: The ReSTOR® SN6AD3 intraocular lens in refractive lens exchange demonstrated good safety, efficacy, and predictability in correcting high ametropia and presbyopia.
Lens implantation, intraocular; Lens, crystalline; Myopia; Hyperopia; Phacoemulsification; Prosthesis design; Tonometry, ocular; Visual acuity
INTRODUÇÃO: O objetivo deste estudo foi avaliar a segurança, eficácia e efetividade da extração de cristalino transparente com implantação de lente intraocular multifocal difrativa. MÉTODOS: Sessenta olhos de 30 pacientes foram submetidos à cirurgia bilateral de retirada de cristalino transparente com colocação de lente intraocular ReSTOR® SN6AD3 com adição de +4,00 D para perto. Os pacientes foram divididos em dois grupos: miopia e hipermetropia. Em ambos os grupos foram medidas a acuidade visual monocular de longe e de perto, com e sem a melhor correção, antes e 6 meses após a cirurgia. RESULTADOS: Após a cirurgia, os resultados de acuidade visual não corrigida para longe foram 0,08 ± 0,15 e 0,11 ± 0,14 logMAR para o grupo de míopes e hipermétropes, respectivamente (50% do grupo de míopes e 46,67% do grupo de hipermétropes tiveram uma acuidade visual para longe maior ou igual a 20/20). Os índices de segurança e eficácia foram 1,05 e 0,88 para o grupo de míopes e 1,01 e 0,86 para o grupo de hipermétropes, para a visão de distância. Dentro do grupo de míopes, 20 olhos permaneceram inalterados após a cirurgia, e 3 ganharam mais de 2 linhas da melhor acuidade visual corrigida. Para o grupo de hipermétropes, 2 olhos perderam 2 linhas da melhor acuidade visual corrigida, 21 não se alteraram e 3 ganharam 2 linhas. Na visão de perto, os índices de segurança e eficácia foram 1,23 e 1,17 para o grupo de míopes, e 1,16 e 1,13 para o grupo de hipermétropes, respectivamente. A acuidade visual binocular de perto com a melhor correção melhorou após a cirurgia nos dois grupos, de 0,10 logMAR para 0,01 logMAR no grupo de míopes e de 0,10 logMAR para 0,04 logMAR no grupo de hipermétropes. CONCLUSÕES: Podemos concluir que a lente intraocular ReSTOR® SN6AD3 demonstrou eficácia, segurança e previsibilidade na cirurgia de altas ametropias e presbiopia.
Implantação de lentes intraoculares; Cristalino; Miopia; Hipermetropia; Hipermetropia; Desenho de próteses; Acuidade visual
ORIGINAL ARTICLE ARTIGO ORIGINAL
Refractive lens exchange with a multifocal diffractive aspheric intraocular lens
Cirurgia de cristalino transparente com lente intraocular multifocal asférica difrativa
Teresa Ferrer-Blasco; Santiago García-Lázaro; César Albarrán-Diego; Lurdes Belda-Salmerón; Robert Montés-Micó
Physician, Optics Department, University of Valencia, Spain
Correspondence address Correspondence address: Robert Montés-Mico PhD. Optometry Research Group Optics Department, Faculty of Physics. University of Valencia. C/ Dr. Moliner, 50. 46100. Burjassot (Valencia). SPAIN. E-mail: robert.montes@uv.es
ABSTRACT
PURPOSE: To evaluate the safety, efficacy and predictability after refractive lens exchange with multifocal diffractive aspheric intraocular lens implantation.
METHODS: Sixty eyes of 30 patients underwent bilateral implantation with AcrySof® ReSTOR® SN6AD3 intraocular lens with +4.00 D near addition. Patients were divided into myopic and hyperopic groups. Monocular best corrected visual acuity at distance and near and monocular uncorrected visual acuity at distance and near were measured before and 6 months postoperatively.
RESULTS: After surgery, uncorrected visual acuity was 0.08 ± 0.15 and 0.11 ± 0.14 logMAR for the myopic and hyperopic groups, respectively (50% and 46.67% of patients had an uncorrected visual acuity of 20/20 or better in the myopic and hyperopic groups, respectively). The safety and efficacy indexes were 1.05 and 0.88 for the myopic and 1.01 and 0.86 for the hyperopic groups at distance vision. Within the myopic group, 20 eyes remained unchanged after the surgery, and 3 gained >2 lines of best corrected visual acuity. For the hyperopic group, 2 eyes lost 2 lines of best corrected visual acuity, 21 did not change, and 3 eyes gained 2 lines. At near vision, the safety and efficacy indexes were 1.23 and 1.17 for the myopic and 1.16 and 1.13 for the hyperopic groups. Best corrected near visual acuity improved after surgery in both groups (from 0.10 logMAR to 0.01 logMAR in the myopic group, and from 0.10 logMAR to 0.04 logMAR in the hyperopic group).
CONCLUSIONS: The ReSTOR® SN6AD3 intraocular lens in refractive lens exchange demonstrated good safety, efficacy, and predictability in correcting high ametropia and presbyopia.
Keywords: Lens implantation, intraocular; Lens, crystalline/surgery; Myopia/surgery; Hyperopia/physiopathology; Hyperopia/surgery; Phacoemulsification; Prosthesis design; Tonometry, ocular; Visual acuity
RESUMO
INTRODUÇÃO: O objetivo deste estudo foi avaliar a segurança, eficácia e efetividade da extração de cristalino transparente com implantação de lente intraocular multifocal difrativa.
MÉTODOS: Sessenta olhos de 30 pacientes foram submetidos à cirurgia bilateral de retirada de cristalino transparente com colocação de lente intraocular ReSTOR® SN6AD3 com adição de +4,00 D para perto. Os pacientes foram divididos em dois grupos: miopia e hipermetropia. Em ambos os grupos foram medidas a acuidade visual monocular de longe e de perto, com e sem a melhor correção, antes e 6 meses após a cirurgia.
RESULTADOS: Após a cirurgia, os resultados de acuidade visual não corrigida para longe foram 0,08 ± 0,15 e 0,11 ± 0,14 logMAR para o grupo de míopes e hipermétropes, respectivamente (50% do grupo de míopes e 46,67% do grupo de hipermétropes tiveram uma acuidade visual para longe maior ou igual a 20/20). Os índices de segurança e eficácia foram 1,05 e 0,88 para o grupo de míopes e 1,01 e 0,86 para o grupo de hipermétropes, para a visão de distância. Dentro do grupo de míopes, 20 olhos permaneceram inalterados após a cirurgia, e 3 ganharam mais de 2 linhas da melhor acuidade visual corrigida. Para o grupo de hipermétropes, 2 olhos perderam 2 linhas da melhor acuidade visual corrigida, 21 não se alteraram e 3 ganharam 2 linhas. Na visão de perto, os índices de segurança e eficácia foram 1,23 e 1,17 para o grupo de míopes, e 1,16 e 1,13 para o grupo de hipermétropes, respectivamente. A acuidade visual binocular de perto com a melhor correção melhorou após a cirurgia nos dois grupos, de 0,10 logMAR para 0,01 logMAR no grupo de míopes e de 0,10 logMAR para 0,04 logMAR no grupo de hipermétropes.
CONCLUSÕES: Podemos concluir que a lente intraocular ReSTOR® SN6AD3 demonstrou eficácia, segurança e previsibilidade na cirurgia de altas ametropias e presbiopia.
Descritores: Implantação de lentes intraoculares; Cristalino/cirurgia; Miopia/cirurgia; Hipermetropia/fisiopatologia; Hipermetropia/cirurgia, facoemulsificação; Desenho de próteses; Acuidade visual
INTRODUCTION
The main goal of a presbyopic patient is to reduce dependence on optical aids and improve both distance and near vision as well as the range of clear vision. Several studies report good safety, predictability, and efficacy outcomes in the implantation with monofocal(1-5), toric(6) and multifocal intraocular lenses (IOLs)(7-9) after refractive lens exchange (RLE).
Patients with hybrid(10-13) spherical IOLs show good distance and near visual acuity with some restrictions at intermediate distances, and visual complaints regarding glare and haloes(14). Aspheric IOLs were designed to introduce negative spherical aberration to compensate for the positive spherical aberration of the cornea(15). These new IOL designs with aspheric profiles were developed with the goal of reducing unwanted visual phenomena associated with multifocal IOL performance. Preliminary reports(16-20) show good distance and near vision as well as functional intermediate vision.
The purpose of this study was to assess the efficacy, safety, and predictability in patients who had undergone bilateral implantation of the AcrySof® ReSTOR® SN6AD3 IOL in the capsular bag after RLE.
METHODS
Study design
The present prospective study involved 60 consecutive eyes of 30 patients bilaterally implanted with of the AcrySof® ReSTOR® SN6AD3 IOL (Alcon Inc., Fort Worth, Texas). Inclusion criteria were age between 45 and 70 years and the motivation to reduce the dependency of any form of spectacle or contact lens correction for distance and near. Exclusion criteria included ≥1 diopters (D) of corneal astigmatism, history of glaucoma or retinal detachment (RD), corneal disease, and previous corneal or intraocular surgery. The tenets of the Declaration of Helsinki were followed in this research. Informed consent was obtained from all patients after the nature and possible consequences of the study were explained. Approval from the Institutional Review Board was obtained.
Before the RLE procedure, patients underwent a complete ophthalmologic examination. Ocular biometry was performed with the IOLMaster biometer (Carl Zeiss Meditec, Inc., Dublin, CA). The SRK/T formula was used for eyes with axial length (AXL) of 22.00 mm or higher and Holladay II formula for eyes with AXL lower than 22.00 mm. The targeted refraction was emmetropia in all cases. The same surgeon performed all surgeries in this study by phacoemulsification with the Infiniti Vision System (Alcon, Fort Worth, TX) using topical anaesthesia and a clear corneal 2.2-3.2 mm incision. There were no complications in any of the cases.
IOL specifications
The AcrySof® ReSTOR® SN6AD3 pseudoaccommodating aspheric IOL uses apodization, diffraction, and refraction. The IOL power used in this study varied from +10.00 to +30.00 D and incorporated a +4.00 D near addition (add). The IOL has an aspheric profile to correct positive spherical aberration of the cornea. The IOL material includes a blue light-filtering chromophore. It has been suggested that the use of a blue-light filter is advisable because it prevents ultraviolet light alterations to the retina without disturbing contrast sensitivity and chromatic vision(21,22).
Visual performance measures
Monocular best corrected visual acuity at distance and near (BCVA and BCNVA, respectively) and monocular uncorrected visual acuity (UCVA and UCNVA, respectively) were measured before and after 6 months post-surgery. Visual acuity was measured as logMAR units using for 100% contrast early treatment diabetic retinopathy study charts (ETDRS) (Optec 6500, Stereo Optical Co, Chicago, IL) under photopic conditions (85 cd/m2) at 4 m. Monocular UCNVA and BCNVA were measured using the Logarithmic Visual Acuity Chart 2000 New ETDRS (Precision Vision, La Salle, IL) at 40 cm under photopic conditions (85 cd/m2).
The safety index (calculated as the ratio between postoperative BCVA and preoperative BCVA), the efficacy index (calculated as the ratio between postoperative UCVA and preoperative BCVA), as well as the predictability (expressed as the percentage of eyes within ±1.00 D and within ±0.50 D of the intended refraction) and correlation coefficient (R) for both groups were calculated at 6 month after surgery.
Data analysis
All examinations were performed preoperatively and 6 months after IOL implantation.
Normality distribution of data was evaluated by the Shapiro-Wilk test. Paired t-test was used to look for differences in visual acuity between pre surgery and post-surgery outcomes. Two sample t-test was used to look for differences between myopic and hyperopic groups. Fisher's exact test was used to look for differences in percentages. Differences were considered to be statistically significant when the P value was less than 0.05.
RESULTS
Sixty eyes of 30 patients had clear lens exchanged with ReSTOR® SN6AD3 IOL. Patients' demographics are shown in table 1. Patients were divided into two groups, myopic (n=15, 10 female, 5 male) with an average preoperative SE refraction of -6.87 ± 2.03 D (range -4.25 to -11.00 D), and hyperopic (n=15, 9 female, 6 male) with an average preoperative SE of +4.52 ± 1.14 D (range +3.00 to +7.00 D). All patients were available for examination at 6 months. No statistically significant differences were found between groups for age (p=0.28) and gender distribution (p>0.99).
Predictability
Six months after surgery, 96.67% of eyes in both groups were within ±1.00 D of the desired refraction; 6 eyes (20%) from the myopic group and 10 eyes (33.33%) from the hyperopic group were within ±0.50 D of the desired refraction (p=0.38).
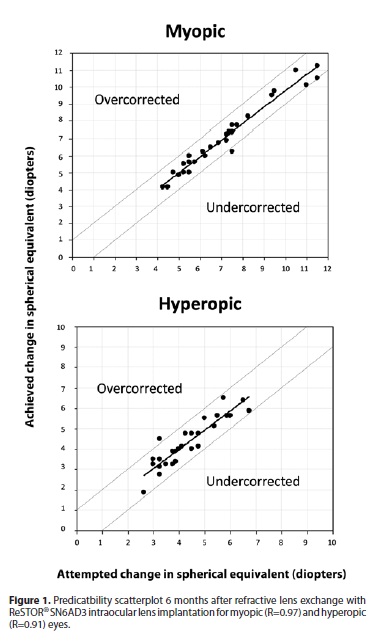
Figure 1 shows the predictability scatterplots for both groups, with the R=0.97 for myopic group and R=0.91 for hyperopic group.
The mean postoperative SE for the myopic and hyperopic groups were -0.19 ± 0.40 D and -0.04 ± 0.46 D, respectively (p=0.18).
Distance visual acuity
Table 2 summarizes monocular visual acuity results obtained for distance vision. There were no statistically significant differences between pre and postoperative BCVA in the hyperopic group (p=0.49), but significant differences were found for the myopic group (p=0.02).
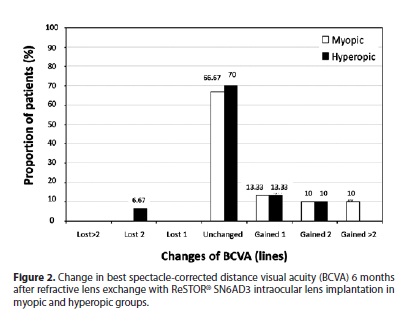
The number of eyes gaining and losing lines of BCVA after surgery is shown in figure 2, both for myopic and hyperopic groups. Six eyes in the myopic group (20%) and 3 eyes in the hyperopic group (10%) gained two or more lines (p=0.47).
Mean safety resulted to be 1.05 ± 0.07 for the myopic group and 1.01 ± 0.05 for the hyperopic group. Mean efficacy was 0.88 ± 0.15 for the myopic group and 0.86 ± 0.09 for the hyperopic group.
Near visual acuity
Table 3 summarizes monocular visual acuity results obtained for near vision. None of the examined eyes in both groups lost lines of BCNVA 6 months after the surgery (see Figure 3). Twelve eyes (40%) in the myopic group and 7 eyes (23.3%) in the hyperopic group gained lines of BCNVA (p=0.27).
Mean safety was 1.23 ± 0.11 for the myopic group and 1.16 ± 0.19 for the hyperopic group, whereas mean efficacy resulted in 1.17 ± 0.11 for the myopic group and 1.13 ± 0.12 for the hyperopic group.
Complications/re-intervention
None of the eyes examined required a second intervention. No potentially sight-threatening complications such as persistent corneal edema, pupillary block, RD, or endophthalmitis were observed during the postoperative period. In addition, no eye was in the need of posterior capsulotomy up to the last postoperative visit.
DISCUSSION
Several studies have shown that the RLE and IOL implantation in the posterior chamber gives good safety, predictability, and efficacy outcomes and that the postoperative improvements in visual acuity are stable throughout a long follow-up period(2-9). In the current study, the safety index was good and similar for both the myopic and hyperopic groups (1.05 and 1.01, respectively). Only two eyes (6.67% of the eyes) in the hyperopic group lost 2 lines of BCVA, all the others maintaining or improving BCVA in both groups. There was no statistically significant difference between pre and postoperative BCVA in hyperopic group (p=0.49), however we found statistically significant difference in the myopic group (p=0.02). Magnification and minimization of the retinal image in myopic and hyperopic patients, respectively, may play a significant role in this difference. Fernandez-Vega et al.(9), studied the spherical ReSTOR® SA60D3 IOL in 224 eyes of 112 consecutive patients and found similar results of safety indexes in myopic and hyperopic groups (1.05 and 1.02, respectively). Thirty per cent and 23% of patients gained ≥1 lines of BCVA in the myopic and hyperopic groups, respectively. In a previous clinical study in which the authors studied 68 eyes implanted with Array multifocal IOL, they found that no eye in the myopic group and 3 eyes in the hyperopic group sustained a loss of 1 line of BCVA. Eighty-two percent of patients (41 eyes) remained unchanged, 10% (5 eyes) had a gain of 1 line, and 2% (1 eye) had a gain of 2 lines of BCVA(8).
The efficacy indexes were good and comparable in both groups (0.88 and 0.86 in myopic and hyperopic groups, respectively). The patients obtained an UCVA of 0.08 logMAR and 0.11 logMAR that improved to 0.01 logMAR and 0.04 logMAR in myopic and hyperopic groups with the best distance correction (Table 2). In the Fernandez-Vega et al.(9) study, the mean UCVA (Snellen decimal) was 0.90 in the myopic group and 0.95 in the hyperopic group and these values remained similar with the best distance correction. Some authors(7)studied 68 eyes implanted with Array multifocal IOL and reported that the 100% of the patients achieved binocular UCVA of 0.5.
The predictability of the RLE treatment was high. The 96.67% of the eyes of myopic and hyperopic groups were within ±1.00 D and 6 eyes (20%) of the myopic group and 10 eyes (33.33%) of the hyperopic group were within ±0.50 D of the desired refraction at 6 months. For both groups, the preoperative SE correlated highly with the amount of change in the postoperative SE (Figure 1; R=0.97 for myopic patients and R=0.91 for hyperopic patients). These results agree with another research in which the authors obtained 100% of the patients within ±1.00 D of the desired refraction 6 months after surgery. Previous studies found better results, with 80.9% of patients(7) and 90.9% for the myopic group and 88.6% for the hyperopic group(9) with a postoperative refractive SE within ±0.50 D of the target refraction in both studies.
Regarding near vision, none of the examined eyes lost ≥2 lines of BCNVA. In the myopic and hyperopic groups respectively, the 60% and 76.67% of the patients had no change in their BCNVA, and the 40% and 23.33% of the patients gained 2 lines (Figure 3). The safety index was 1.23 and 1.16 in myopic and hyperopic groups, respectively. In the Fernandez-Vega et al.(9), study the 15% and 5% of the myopic and hyperopic patients, respectively, lost 1 or 2 lines of BCNVA. About 60% of the patients had no change in their BCNVA and the about 25% of the patients gained 1 or 2 lines of BCNVA. The efficacy indexes in near vision were good and comparable in both groups (1.17 and 1.13 in myopic and hyperopic groups, respectively). The patients obtained an UCNVA of 0.03 logMAR and 0.08 logMAR that improved to 0.01 logMAR and 0.04 logMAR in myopic and hyperopic groups with the best distance correction (Table 2).
No eye lost vision as a result of RD, and no Nd:YAG capsulotomy was necessary in our patients with a 6-month follow-up. Nevertheless, RLE remains a controversial technique because it is an invasive procedure and carries an increased risk of RD(23-26). Other authors(25) showed an RD rate of 8.1% after 7 years. A recent research reported that in 62 myopic cases of RLE over an 11-year period, a 3.2% RD rate occurred at intervals of 2 and 5 months after surgery(5). If refractive lens exchange is performed in patients with high myopia, they must give extensive informed consent regarding the long-term risks for RD. In addition, the risks for RD, endophthalmitis, glaucoma, corneal decompensation, and posterior capsule opacification should be discussed with all patients regardless of their preoperative refractive error. Preoperative and long-term retinal evaluation is imperative previous to this procedure to control and treat any possible retinal complication.
One of the most important assessments for successful RLE with multifocal IOLs use involves precise preoperative measurement of axial length and accurate IOL power calculation. We chose the SRK/T formula for eyes with AXL≥22.00 mm and the Holladay II formula for eyes with AXL<22.00 mm based on previous reports(27-28). Minimal deviation from target refraction was obtained using these formulas (0.04 ± 0.39 and 0.17 ± 0.40 D for myopic and hyperopic groups, respectively). Current customized calculation using individual computer models for each eye(29) would be helpful in this type of refractive surgery patients.
Results in the present study reveal good predictability, safety and efficacy for the RLE with ReSTOR® SN6AD3 IOL implantation. However, some limitations in our study must be taken into account. First, spectacle independence and photic phenomena have not been investigated, so future studies are needed, considering subjective questionnaires for those purposes. Our main interest in the present work was to report distance and near visual acuity, and intermediate vision has not been measured, being this the second limitation. A recent study showed that intermediate vision is improved with the aspheric ReSTOR® SN6AD1 IOL with +3.00 D of add model in relation to that found with the spherical ReSTOR® SA60D3 IOL with +4.00 D of add model and the aspheric ReSTOR® SN6AD3 IOL with +4.00 D of add model(20). The role of pupil size also needs to be addressed in future studies, and so the changes in contrast sensitivity.
ACKNOWLEDGEMENT
We thank Cristina Puchades for her help in different aspects of the study.
Submitted for publication: January, 11, 2012
Accepted for publication: May, 15, 2012
Study was carried out at Optics Department, University of Valencia, Spain.
Disclosure of potential conflicts of interest: T.Ferrer-Blasco, None; S.García-Lázaro, None; C.Albarrán-Diego, None; L.Belda-Salmerón, None; R.Montés-Micó, None.
This research was supported in part by Ministerio de Ciencia e Innovación Research Grants to Robert Montés-Micó (#SAF2008-01114# and #SAF2009-13342#)
-
1Goldberg MF. Clear lens extraction for axial myopia: an appraisal. Ophthalmology. 1987;94(5):571-82.
-
2Colin J, Robinet A. Clear lensectomy and implantation of low-power posterior chamber intraocular lens for the correction of high myopia: a four-year follow-up. Ophthalmology. 1997;104(1):73-7; discussion 77-8. Comment in: Ophthalmology. 1998; 105(6):944-5. Ophthalmology. 1997;104(10):1525.
-
3Pucci V, Morselli S, Romanelli F, Pignatto S, Scandellari F, Bellucci R. Clear lens phacoemulsification for correction of high myopia. J Cataract Refract Surg 2001;27(6): 896 -900. Comment in: J Cataract Refract Surg. 2001;27(12):1901-2.
-
4Fernandez-Vega L, Alfonso JF, Villacampa T. Clear lens extraction for the correction of high myopia. Ophthalmology. 2003;110(12):2349 -54. Comment in: Ophthalmology. 2004;111(6):1263.
-
5Horgan N, Condon PI, Beatty S. Refractive lens exchange in high myopia: long term follow up. Br J Ophthalmol. 2005;89(6):670-2.
-
6Ruíz-Mesa R, Carrasco-Sánchez D, Díaz-Alvarez SB, Ruíz-Mateos MA, Ferrer-Blasco T, Montés-Micó R. Refractive lens exchange with foldable toric intraocular lens. Am J Ophthalmol. 2009;147(6):990-6.
-
7Packer M, Fine IH, Hoffman RS. Refractive lens exchange with the array multifocal intraocular lens. J Cataract Refract Surg. 2002;28(3):421- 4. Comment in: J Cataract Refract Surg. 2002;28(3):379-80.
-
8Dick HB, Gross S, Tehrani M, Eisenamann D, Pfeiffer N. Refractive lens exchange with an array multifocal intraocular lens. J Refract Surg. 2002;18(5):509-18.
-
9Fernández-Vega L, Alfonso JF, Rodríguez P, Montés-Micó R. Clear lens extraction with multifocal apodized diffractive intraocular lens implantation. Ophthalmology. 2007;114(8):1491-8.
-
10Kohnen T, Allen D, Boureau C, Dublineau P, Hartmann C, Mehdorn E, et al. European multicenter study of the AcrySof ReSTOR apodized diffractive intraocular lens. Ophthalmology. 2006;113(4):578-84.
-
11Alfonso JF, Fernández-Vega L, Baamonde MB, Montés-Micó R. Prospective visual evaluation of apodized diffractive intraocular lenses. J Cataract Refract Surg. 2007; 33(7):1235-43.
-
12Fernández-Vega L, Alfonso JF, Montés-Micó R, Amhaz H. Visual acuity tolerance to residual refractive errors in patients with an apodized diffractive intraocular lens. J Cataract Refract Surg. 2008;34(2):199-204.
-
13Alfonso JF, Fernández-Vega L, Valcárcel B, Ferrer-Blasco T Montés-Micó R. Outcomes and patient satisfaction after presbyopia bilateral lens exchange with the ReSTOR intraocular lens in emmetropes. J Refract Surg. 2010;26(12):927-33.
-
14Blaylock JF, Si Z, Vickers C. Visual and refractive status at different focal distances after implantation of the ReSTOR multifocal intraocular lens. J Cataract Refract Surg. 2006;32(9):1464-73.
-
15Montés-Micó R, Ferrer-Blasco T, Cerviño A. Analysis of the possible benefits of aspherical intraocular lenses: Review of the literature. J Cataract Refract Surg. 2009; 35(1):172-81. Comment in: J Cataract Refract Surg. 2009;35(6):962-3; author reply 963-4.
-
16Alfonso JF, Fernández-Vega L, Señaris A, Montés-Micó R. Prospective study of the Acri.LISA bifocal intraocular lens. J Cataract Refract Surg. 2007;33(11):1930-5.
-
17Alfonso JF, Fernández-Vega L, Valcárcel B, Montés-Micó R. Visual performance after AcrySof ReSTOR aspheric intraocular lens implantation. J Optom. 2008;1:30-5.
-
18Alió JL, Elkady B, Ortiz D, Bernabeu G. Clinical outcomes and intraocular optical quality of a diffractive multifocal intraocular lens with asymmetrical light distribution. J Cataract Refract Surg. 2008;34(6):942-8.
-
19Alfonso JF, Puchades C, Fernández-Vega L, Valcárcel B, Montés-Micó R, Ferrer-Blasco T. Visual acuity comparison of 2 models of bifocal aspheric intraocular lenses. J Cataract Refract Surg. 2009; 35(4):672-6.
-
20Alfonso JF, Fernández-Vega, F, Amhaz H, Montés-Micó R, Valcárcel B, Ferrer-Blasco T. Visual function after implantation of an aspheric bifocal intraocular lens. J Cataract Refract Surg. 2009;35(5):885-92.
-
21Rodríguez-Galietero A, Montés-Micó R, Muñoz G, Albarrán Diego C. Comparison of contrast sensitivity and color discrimination after clear and yellow intraocular lenses implantation J Cataract Refract Surg. 2005;31(9):1736-40.
-
22Rodríguez-Galietero A, Montés-Micó R, Muñoz G, Albarrán-Diego C. Blue-light filtering intraocular lens patients with diabetes contrast sensitivity and chromatic discrimination. J Catarac Refract Surg. 2005;31(11):2088-92.
-
23Barraquer C, Cavelier C, Mejia LF. Incidence of retina detachment following clear-lens extraction in myopic patients. Retrospective analysis. Arch Ophthalmol. 1994;112(3): 336 -9. Comment in: Arch Ophthalmol. 1994;112(3):321-3.
-
24Lyle WA, Jin GJ. Phacoemulsification with intraocular lens implantation in high myopia. J Cataract Refract Surg. 1996;22(2):238-42.
-
25Colin J, Robinet A, Cochener B. Retinal detachment after clear lens extraction for high myopia: seven-year follow-up. Ophthalmology. 1999;106(12):2281-4; discussion 2285.
-
26Fritch CD. Risk of retinal detachment in myopic eyes after intraocular lens implantation: a 7-year study. J Cataract Refract Surg. 1998;24(10):1357-60.
-
27Sanders DR, Retzlaff JA, Kraff MC, Gimbel HV, Raanan MG. Comparison of the SRK/T formula and other theoretical and regression formulas J Cataract Refract Surg. 1990; 16(3)341- 6.
-
28Hoffer KJ. Clinical results using the Holladay 2 intraocular lens power formula. J Cataract Refract Surg. 2000;26(8):1233-7.
-
29Einighammer J, Oltrup T, Bende T, Jean B. The individual virtual eye: a computer model for advanced intraocular lens calculation. J Optom. 2009;2:70-82.
Correspondence address:
Publication Dates
-
Publication in this collection
02 Aug 2012 -
Date of issue
June 2012
History
-
Received
11 Jan 2012 -
Accepted
15 May 2012