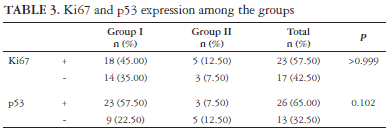Abstracts
CONTEXT: Gastrointestinal stromal tumor (GIST) is the most common mesenchymal tumor. Cellular proliferation and apoptosis is gaining importance for predicting prognosis in several cancers. OBJECTIVE: To investigate the Ki67 and p53 immunostaining in GISTs. METHODS: Specimens from 40 patients with GIST were assessed for immunohistochemical expression of Ki67 and p53. The tumors were divided according the risk of recurrence in two groups: I with high or intermediate risk and; II with low or very low risk. RESULTS: Among the 40 patients, 21 were men, the mean age was 56 years, 16 occurred in the small intestine and 13 in the stomach, 5 in the retroperitonium, 4 in the colon or rectum and 2 in the mesenterium. Thirty two tumors were from group I and 8 from group II. Half of the patients developed recurrence, being 90% of the group I (P = 0.114). The tumor Ki67 labelling index ranged from 0.02 to 0.35 (mean level 0.12). This index was marginally higher in the group I patients with recurrence (P = 0.09) compared to the patients of the same group without recurrence. p53 staining was expressed in 65% of the GISTs. A higher frequency of p53 and Ki67 had been found in the group I tumors when compared to the other group (P = 0.022; OR = 8.00 - IC 95%: 1.32-48.65). CONCLUSION: The most common site was the small intestine and 80% had a malignant potential justifying the high recurrence observed. No significant correlation was found between p53 and overall outcome of the patients. In group I patients, the evaluation Ki67LI may be a marker of prognosis. The positivity of both markers is higher among the patients with worst prognosis than in the others.
Gastrointestinal stromal tumors; Ki-67 antigen; Tumor suppressor protein p53
CONTEXTO: Os tumores estromais gastrointestinais (GIST) são os tumores mesenquimais mais frequentes. A proliferação intestinal e a apoptose são cada vez mais importantes na avaliação do prognóstico de diversos cânceres. OBJETIVO: Avaliar a imunoexpressão de Ki67 e p53 em GIST. MÉTODOS: Foram estudados a expressão de Ki67 e p53 por imunoistoquimica em tumores de 40 pacientes com GIST. Os tumores foram divididos segundo o risco de recurrência em 2 grupos: I com risco alto ou intermediário e II com risco baixo ou muito baixo. RESULTADOS: Entre os 40 pacientes, 21 eram do sexo masculino, a idade média foi de 56 anos, 16 ocorreram no intestino delgado, 13 no estômago, 5 no retroperitônio, 4 no cólon e reto, e 2 no mesentério. Trinta e dois tumores foram classificados no grupo I e 8 no grupo II. Metade dos pacientes desenvolveu recurrência, sendo 90% de cólon (P = 0,114). O índice de proliferação tumoral Ki67 variou entre 0,02 e 0,35 (média = 0,12). Este índice foi marginalmente superior nos tumores do grupo I com recurrência (P = 0,09), quando comparado aos do mesmo grupo sem recurrência. A expressão do p53 foi observada em 65% dos GISTs. Nos tumores do grupo I foi observada com maior frequência, expressão de p53 e Ki67 (P = 0,022; OR = 8.00 - IC 95%: 1,32-48,65). CONCLUSÃO: A localização mais comum foi no intestino delgado, 80% tinham potencial maligno, justificando a alta recurrência encontrada. Não se observou correlação significante entre p53 e evolução dos pacientes. Nos pacientes do grupo I, a avaliação do KI67LI pode ser um marcador de prognóstico. A positividade dos dois marcadores é maior entre os pacientes de pior prognóstico.
Tumores do estroma gastrointestinal; Antígeno Ki-67; Proteína supressora de tumor p53
ARTIGO ORIGINAL ORIGINAL ARTICLE
Ki67 and p53 in gastrointestinal stromal tumors - GIST
Ki67 and p53 em tumores estromais gastrointestinais - GIST
Lúcio Roberto de Oliveira das NevesI; Celina Tizuko Fujiyama OshimaII; Ricardo Artigiani-NetoII; Gianni YanaguibashiIII; Laércio Gomes LourençoIV; Nora Manoukian ForonesI
IMedicine Department, Gastroenterology Division
IIPathology Department
IIIStatistics Department
IVSurgery Department, "Universidade Federal de São Paulo", São Paulo, SP, Brazil
Correspondence Correspondence: Dr. Nora Manoukian Forones Disciplina de Gastroenterologia, Universidade Federal de São Paulo Rua Botucatu, 740 - 2º andar 04023-900 - São Paulo, SP, Brazil Email: nora@gastro.epm.br
ABSTRACT
CONTEXT: Gastrointestinal stromal tumor (GIST) is the most common mesenchymal tumor. Cellular proliferation and apoptosis is gaining importance for predicting prognosis in several cancers.
OBJECTIVE: To investigate the Ki67 and p53 immunostaining in GISTs.
METHODS: Specimens from 40 patients with GIST were assessed for immunohistochemical expression of Ki67 and p53. The tumors were divided according the risk of recurrence in two groups: I with high or intermediate risk and; II with low or very low risk.
RESULTS: Among the 40 patients, 21 were men, the mean age was 56 years, 16 occurred in the small intestine and 13 in the stomach, 5 in the retroperitonium, 4 in the colon or rectum and 2 in the mesenterium. Thirty two tumors were from group I and 8 from group II. Half of the patients developed recurrence, being 90% of the group I (P = 0.114). The tumor Ki67 labelling index ranged from 0.02 to 0.35 (mean level 0.12). This index was marginally higher in the group I patients with recurrence (P = 0.09) compared to the patients of the same group without recurrence. p53 staining was expressed in 65% of the GISTs. A higher frequency of p53 and Ki67 had been found in the group I tumors when compared to the other group (P = 0.022; OR = 8.00 - IC 95%: 1.32-48.65).
CONCLUSION: The most common site was the small intestine and 80% had a malignant potential justifying the high recurrence observed. No significant correlation was found between p53 and overall outcome of the patients. In group I patients, the evaluation Ki67LI may be a marker of prognosis. The positivity of both markers is higher among the patients with worst prognosis than in the others.
Headings: Gastrointestinal stromal tumors. Ki-67 antigen. Tumor suppressor protein p53.
RESUMO
CONTEXTO: Os tumores estromais gastrointestinais (GIST) são os tumores mesenquimais mais frequentes. A proliferação intestinal e a apoptose são cada vez mais importantes na avaliação do prognóstico de diversos cânceres.
OBJETIVO: Avaliar a imunoexpressão de Ki67 e p53 em GIST.
MÉTODOS: Foram estudados a expressão de Ki67 e p53 por imunoistoquimica em tumores de 40 pacientes com GIST. Os tumores foram divididos segundo o risco de recurrência em 2 grupos: I com risco alto ou intermediário e II com risco baixo ou muito baixo.
RESULTADOS: Entre os 40 pacientes, 21 eram do sexo masculino, a idade média foi de 56 anos, 16 ocorreram no intestino delgado, 13 no estômago, 5 no retroperitônio, 4 no cólon e reto, e 2 no mesentério. Trinta e dois tumores foram classificados no grupo I e 8 no grupo II. Metade dos pacientes desenvolveu recurrência, sendo 90% de cólon (P = 0,114). O índice de proliferação tumoral Ki67 variou entre 0,02 e 0,35 (média = 0,12). Este índice foi marginalmente superior nos tumores do grupo I com recurrência (P = 0,09), quando comparado aos do mesmo grupo sem recurrência. A expressão do p53 foi observada em 65% dos GISTs. Nos tumores do grupo I foi observada com maior frequência, expressão de p53 e Ki67 (P = 0,022; OR = 8.00 - IC 95%: 1,32-48,65).
CONCLUSÃO: A localização mais comum foi no intestino delgado, 80% tinham potencial maligno, justificando a alta recurrência encontrada. Não se observou correlação significante entre p53 e evolução dos pacientes. Nos pacientes do grupo I, a avaliação do KI67LI pode ser um marcador de prognóstico. A positividade dos dois marcadores é maior entre os pacientes de pior prognóstico.
Descritores: Tumores do estroma gastrointestinal. Antígeno Ki-67. Proteína supressora de tumor p53.
INTRODUCTION
The gastrointestinal stromal tumor (GIST) is a mesenchimal tumor of the gastrointestinal tract. GISTs are thought to originate from the intestinal cell of Cajal and function as intestinal pacemaker cells that regulate intestinal motility(1, 8, 15, 29, 36). Historically GIST was misclassified as leiomyomas or leyomiossarcomas. Subsequently, it has been determined that GISTs has particular immunophenotypical markers. The most notable one was is KIT (CD117), a tyrosine kinase inhibitor that is present in almost 95% of this tumors(15, 29, 33). KIT is a transmembrane protein that stimulate the cell proliferation and inhibit the apoptosis. PDGFRa (platelet-derived growth factor-∞) is also present in 5% of GIST(7, 16, 17, 25).
These tumors are rare (10 to 20 persons/1 million habitants)(5, 28). In the United States up to 5,000 new cases are diagnosed per year(5, 20).. There is no information about the incidence of this disease, in Brazil.
GIST can occur along the gastrointestinal tract, most commonly in stomach (70%)(10, 28, 29) and small intestine (20%-25%)(10, 28, 31). The clinical presentation depends on the site of the tumor and almost 20% of them may be asymptomatic. The diagnosis may be done by endoscopic exams, computed tomography or magnetic resonance of the abdomen(18, 22). Percutaneous biopsy can not be recommended in resectable tumors because the biopsy can precipitate tumor rupture, bleeding or dissemination. Histologically, GIST is characterized as spindle cell or infrequently as epithelyoid type. Immunohistochemically GISTs are immunopositive for CD117 (c-KIT) in 95%(7, 28). Some tumors are also positive for CD34 (70%-30%), desmine (<5%) and S-100 protein (10%)(7, 28).
The treatment of choice is surgery(9, 10), even in the patients with local resectable metastases(9). Recurrent or metastatic disease has been treated with imatinib (Gleevec), a target molecular therapy(5, 10, 16, 21). Imatinib is a potent inhibitor of KIT signaling tyrosine kinase inhibitor(5, 6, 10, 20, 29). Before the use of imatinib, the median survival of the patients with resectable disease was 66 months(5, 9) and 9 to 12 months in irresectable disease(3, 4, 9, 11, 12, 14, 38).
Almost 50% of the patients develop recurrence after complete resection of GIST and 5 year survival is usually about 50%. Tumor size and mitotic index are the common prognostic factors used. Tumors with more than 10 cm(9, 15) or with more than five mitosis(2, 28, 29) per 50 high power field (HPF) had worst prognosis. Mostly in this group of patients, the use of imatinib, a c-KIT inhibitor as an adjuvant treatment has been studied(5, 6, 7, 10, 20, 29).
New drugs and new markers of prognosis has been investigated to better know the strategy of treatment and to identify variables that affect survival. Cell proliferating and apoptosis markers cells, largely studied in others kind of gastrointestinal cancer, are being investigated in GIST(35, 37).
Ki67 is a nuclear proliferation associated antigen expressed in the growth and synthesis phases of the cell cycle (G1, S, G2 and M) but not in the resting phase, G0. This antigen provides information about the proportion of active cells in the cell cycle(15, 29, 32, 35, 37) .
The Tp53 tumor suppressor gene is mapped on chromosome 17p and plays a role in regulating the cell cycle, DNA repair and synthesis, and programmed cell death. Mutation of p53 leads to disruption of these pathways and resultant selective growth advantage of tumor cells. Immunohistochemical staining is positive for wild-type p53 as well as for mutant-type p53; however, most positive cells represent mutant p53, since the half-life of the wild-type p53 protein is very short(1).
The aim of this study was to evaluate Ki67 and p53 in GIST and its possible involvement in prognosis.
METHODS
Archival pretreatment pathological specimens were obtained from patients with GIST. The histologic diagnosis of all tumors was confirmed by two pathologists according to the characteristics described by MIETTINEN et al.(13, 29). All the tumors stained positively for KIT. The study was approved by a local human ethical committee.
Patient data include age, sex, local and size of the tumor. The biological potential(2, 10, 28, 29) was determined by the tumor size and the mitotic index (Table 1). Tumor size was recorded as the largest diameter in any dimension of the primary tumor and was stratified as <5 cm, 5 to 10 cm or >10 cm. Mitotic activity has been evaluated in 50 HPF for each tumor. The patients were classified regarding to the biological potential in two groups: group I (intermediate or high grade) or group II (very low or low grade). Each patient was classified at the end of the study as alive (without disease or with recurrence) or dead (by the disease progression or by another cause).
Imunohistochemistry
Expression of p53 and Ki67 were determined by immunohistochemistry, using avidin-biotin-peroxidase.
Sections of 3 µm mounted on 3-aminopropylotrimetoxy-silane (Sigma) coated slides were dewaxed in xylene, taken through ethanol to water to rehydrate. The sections were microwaved in 10 mM sodium citrate buffer (pH 6,0) at 10 min intervals for a total of 20 min. Endogenous peroxidase activity was blocked by incubating the sections in a solution of 3% hydrogen peroxide for 20 minutes at room temperature. After washing in PBS the sections were incubated with the primary monoclonal mouse anti-human human Ki67 (1:50), monoclonal mouse anti-human p53, clone DO7 (Dako) (1:100), overnight at 4º C. The sections were washed with PBS and incubated with a biotinylated secondary antibody for 30 minutes, followed by incubation with streptoavidine-biotin-peroxidase complex (DAKO) for further 30 minutes, at room temperature. Staining was carried out using a solution 3-3'diaminobenzidine (DAB-Sigma), containing 1% hydrogen peroxide and lightly counterstained with Harris hematoxylin.
Sections known to express high levels of p53 and Ki67 were included as positive controls, while negative control slides omitted the primary antibody. The sections were judged positive for p53 when 5% or more of the nucleus of tumor cells were stained as described by MEARA et al.(24) and NAKAMURA et al.(30). Ki67 labelling index was determined by observing 1000 nuclei in areas of the section with the highest labelling rates, and was considered positive when 10% or more of tumor cells were stained. These cut-off have been used to correlate with prognosis(34).
Statistical analysis
The significance of associations was determined by the chi-square test or the Fisher's exact test. The t Student test was done to compare the levels of the parameters studied between the two groups. Odds ratio and 95% confidence intervals were calculated to describe the association among the expected biological behavior and the proteins studied. P value <0.05 was considered statistically significant.
RESULTS
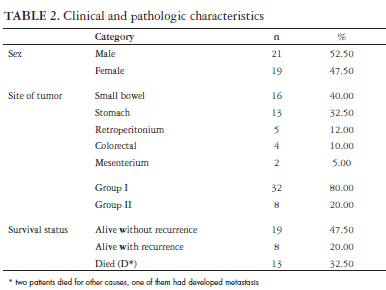
Among the 40 patients studied, 21 were men with an overall mean age of 56 years (range 22-84 years). The median follow up of the patients was 23 months (DP = 33.85 months). Eleven patients died of recurrent disease and two by others diseases, being one with recurrence of GIST. Twenty seven were alive, eight with recurrent disease (Table 2). Tumors were located in the small bowel in 16 cases (40%), in the stomach in 13 (32.50%), in the retroperitonium in 5 (12.50%), in the colon or rectum in 4 (10%) and in the mesenterium in 2 (5%) (Table 2). Pathological assessment of the resected tumors showed a median tumor size of 11.67 cm (DP = 8.62 cm). The mitotic counts were 5/50 HPF in 21 tumors (10 without mitosis), between 5-10/50 HPF in 3 and >10/HPF in 2. According to the NIH Consensus Conference criteria, of the 40 tumors, 3 patients were in the very low risk class, 5 in the low risk class, 10 in the intermediate risk class and 22 in the high risk class. Thirty two (80%) cases were classified as group I and eight (20%) as group II (Table 2). There was no difference among the ages between the two groups of patients.
p53 was positive in 3 (7.50%) of the cases of the group II and 23 (57.50%) of the cases with higher risk (group I, P = 0.102). Ki67 expression range from 0.02 to 0.35 (mean ± DP = 12.34 ± 9.53). Eighteen cases (45%) were positive for Ki67 in group I and 5 (12.50%) in group II (P >0.999) (Table 3). The mean Ki67 labelling index was 0.12 (ranged from 0.02 to 0.35) being marginally higher in group I patients with recurrence or death from the disease (P = 0.09). Nine point fifty seven percent of the cells staining positive in median.
During the follow up period, 75% of the patients of the group II and 43.75% of the group I were free of tumor (P = 0.235).
Although there were no correlation among the antigens studied and the overall outcome of the patients (P = 0.507 for p53 and P = 0.749 for Ki67) (Table 4), some interesting things were find in the group I patients. We found a significantly higher frequency of p53 and Ki67 in the group I tumors when compared to the other group (P = 0.022; OR = 8.00 - IC 95%: 1.32 - 48.65). The frequency of p53 positive imunoexpression were also higher in Ki67 positive tumors in the group I tumors (P = 0.048; OR = 12.00 - IC 95% [1.18-121.58]). The Ki67LI also had a marginal tendency to be higher in patients who developed recurrence or died from the disease (P = 0.09) (Table 5) but this difference was not observed in relation to p53 (P = 1).
DISCUSSION
The mean age of the patients was 56 years, which is similar to the results of others(28) who reported that GISTs can occur in people of all ages, mostly between 55 and 65 years, with a median age of 60 years old. This tumor occurs in both sexes equally(19, 26, 27) or slightly higher in men as observed in this study(8, 23, 32). The primary site of tumors noted arise in the small bowel (40%) or in the stomach (32%) in contrast to others which this tumor commonly arise mostly in the stomach (60%-70%) and in the small intestine in a smaller frequency (26%-35%) (29). Eighteen tumors had more than 10 cm in maximal diameter that is alone one of the main prognostic factor(9, 15). Almost half of the tumors had also more than 5 mitoses/per HPF that is also a factor of worst prognosis(2, 28, 29). The literature articles related that up to 20% of GISTs tumors had a high malignant potential(10, 28), however in this study this potential arise to 80%. This fact occurred because the group of patients enrolled was in treatment by the Oncology Division. Patients with lower risk GIST are frequently followed by surgeons and gastroenterologists in Brazil, being the patients followed by oncologists the GIST's patients with worst prognosis.
The expression of Ki67 was positive in 57.50% of the tumors. The expression of Ki67 changes widely from one study to another, probably by the variety of the cut-offs accepted by each author. We accepted the cut-off of 10% as described by TOQUET et al.(34) and NAKAMURA et al.(30).These authors have defined the mean level as the cut-off of the GISTs tumors studied (4.92) and MEARA et al.(24) as 5%. In this research the median of the Ki67LI obtained was 9.57 and the mean value was 12.34. WONG et al.(37) described that almost 20% of the GISTs were positive for Ki67 while others related a higher positive tumors(15, 35, 36). The positivity of p53 was observed in 65% of the tumors and the literature show similar results that are close to 50%(1, 35, 36, 37). The variety of cut-offs accepted for p53 also was different from one study research to another.
During the follow up 75% of the patients with low or very low malignant potential tumors were alive without disease and 43,75% were free of recurrence, however this difference was not significant.
The expression of Ki67 or p53 had not been correlated with best or worst prognosis, however in group I tumors which developed recurrence or metastasis the mean level of Ki67LI was marginal higher than in the others. A 12 times higher positivity of p53 was detected in group I Ki67 positive tumors. Among the group I tumors, the immunoexpression of both p53 and Ki67 was significantly higher when compared to the group II.
GISTs tumors are rare, however up to 2/3 developed recurrence or metastasis during the follow up. This risk is higher in high potential malignant tumors and the study of tecidual markers as Ki67 may help to decide which patient will have worst prognosis.
Recebido em 3/7/2008.
Aprovado em 20/8/2008
Oncology Group, Gastroenterology Division and Pathology Department, "Universidade Federal de Sao Paulo"
- 1. Al-Bozom IA. p53 expression in gastrointestinal stromal tumors. Pathol Int. 2001;51:519-23.
- 2. Amin MB, Ma CK, Linden MD, Kubus JJ, Zarbo RJ. Prognostic value of proliferating cell nuclear antigen index in gastric stromal tumors. Correlation with mitotic count and clinical outcome. Am J Clin Pathol. 1993;100:428-32.
- 3. Balcerzak SP, Benedetti J, Weiss GR, Natale RB. A phase II trial of paclitaxel in patients with advanced soft tissue sarcomas. A Southwest Oncology Group study. Cancer. 1995;76:2248-52.
- 4. Blair SC, Zalupski MM, Baker LH. Ifosfamide and etoposide in the treatment of advanced soft tissue sarcomas. Am J Clin Oncol. 1994;17:480-4.
- 5. Blanke CD, Corless CL. State-of-the art therapy for gastrointestinal stromal tumors. Cancer Invest. 2005;23(3):274-80.
- 6. Blay JY, Le Cesne A, Verweij J, Scurr M, Seynaeve C, Bonvalot S, Hogendoorn P, Jimeno J, Evrard V, van Glabbeke M, Judson I. A phase II study of ET-743/trabectedin ('Yondelis') for patients with advanced gastrointestinal stromal tumours. Eur J Cancer. 2004;40:1327-31.
- 7. Blay JY, Bonvalot S, Casali P, Choi H, Debiec-Richter M, Dei Tos AP, Emile JF, Gronchi A, Hogendoorn PC, Joensuu H, Le Cesne A, McClure J, Maurel J, Nupponen N, Ray-Coquard I, Reichardt P, Sciot R, Stroobants S, van Glabbeke M, van Oosterom A, Demetri GD; GIST consensus meeting panelists. Consensus meeting for the management of gastrointestinal stromal tumors. Report of the GIST Consensus Conference of 20-21 March 2004, under the auspices of ESMO. Ann Oncol. 2005;16:566-78.
- 8. Bucher P, Taylor S, Villiger P, Morel P, Brundler MA. Are there any prognostic factors for small intestinal stromal tumors?. Am J Surg. 2004;187:761-6.
- 9. DeMatteo RP, Lewis JJ, Leung D, Mudan SS, Woodruff JM, Brennan MF. Two hundred gastrointestinal stromal tumors: recurrence patterns and prognostic factors for survival. Ann Surg. 2000;231:51-8.
- 10. Demetri, G.D. Identification and treatment of chemoresistant inoperate or metastatic GIST: experience with the selective tyrosine kinase inhibitor imatinib mesylate (STI571). Eur J Cancer. 2002;38(Suppl 5):s52-s9.
- 11. Edmonson JH, Marks RS, Buckner JC, Mahoney MR. Contrast of response to dacarbazine, mitomycin, doxorubicin, and cisplatin (DMAP) plus GM-CSF between patients with advanced malignant gastrointestinal stromal tumors and patients with other advanced leiomyosarcoma. Cancer Invest. 2002;20:605-12.
- 12. Elias A, Ryan L, Sulkes A, Collins J, Aisner J, Antman KH. Response to mesna, doxorubicin, ifosfamide, and dacarbazine in 108 patients with metastatic or unresectable sarcoma and no prior chemotherapy. J Clin Oncol.1989;7:1208-16.
- 13. Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, Miettinen M, O'Leary TJ, Remotti H, Rubin BP, Shmookler B, Sobin LH, Weiss SW. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Hum Pathol. 2002;33:459-65.
- 14. Frustaci S, Foladore S, Buonadonna A, De Paoli A, Crivellari D, Carbone A, Sorio R, Morassut S, Monfardini S. Epirubicin and ifosfamide in advanced soft tissue sarcomas. Ann Oncol. 1993;4:669-72.
- 15. Hasegawa T, Matsuno Y, Shimoda T, Hirohashi S. Gastrointestinal stromal tumor: consistent CD117 immunostaining for diagnosis, and prognostic classification based on tumor size and MIB-1 grade. Hum Pathol. 2002;33:669-76.
- 16. Heinrich MC, Corless CL, Demetri GD, Blanke CD, von Mehren M, Joensuu H, McGreevey LS, Chen CJ, Van den Abbeele AD, Druker BJ, Kiese B, Eisenberg B, Roberts PJ, Singer S, Fletcher CD, Silberman S, Dimitrijevic S, Fletcher JA. Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. J Clin Oncol. 2003;21:4342-9.
- 17. Heinrich MC, Corless CL, Duensing A, McGreevey L, Chen CJ, Joseph N, Singer S, Griffith DJ, Haley A, Town A, Demetri GD, Fletcher CD, Fletcher JA. PDGFRA activating mutations in gastrointestinal stromal tumors. Science. 2003;299:708-10.
- 18. Horton KM, Juluru K, Montogomery E, Fishman EK. Computed tomography imaging of gastrointestinal stromal tumors with pathology correlation. J Comput Assist Tomogr. 2004;28:811-7.
- 19. Hou YY, Tan YS, Sun MH, Wei YK, Xu JF, Lu SH, A-Ke-Su SJ, Zhou YN, Gao F, Zheng AH, Zhang TM, Hou WZ, Wang J, Du X, Zhu XZ. C-kit gene mutation in human gastrointestinal stromal tumors. World J Gastroenterol. 2004;10:1310-4.
- 20. House MG, Guo M, Efron DT, Lillemoe KD, Cameron JL, Syphard JE, Hooker CM, Abraham SC, Montgomery EA, Herman JG, Brock MV. Tumor suppressor gene hypermethylation as a predictor of gastric stromal tumor behavior. J Gastrointest Surg. 2003;7:1004-14.
- 21. Kim HC, Lee JM, Choi SH, Kim KW, Kim SH, Lee JY, Han JK, Choi BI. Imaging of gastrointestinal stromal tumors. J Comput Assist Tomogr. 2004;28:596-604.
- 22. Lau S, Tam KF, Kam CK, Lui CY, Siu CW, Lam HS, Mak KL. Imaging of gastrointestinal stromal tumour (GIST). Clin Radiol. 2004;59:487-98.
- 23. Lerma E, Oliva E, Tugués D, Prat J. Stromal tumours of the gastrointestinal tract: a clinicopathological and ploidy analysis of 33 cases. Virchows Arch. 1994;424:19-24.
- 24. Meara RS, Cangiarella J, Simsir A, Horton D, Eltoum I, Chhieng DC. Prediction of aggressiveness of gastrointestinal stroma tumors based on immunostainong with bcl-2. Ki67 and p53. Cytopathology 2007;18:283-9.
- 25. Medeiros F, Corless CL, Duensing A, Hornick JL, Oliveira AM, Heinrich MC, Fletcher JA, Fletcher CD. KIT-negative gastrointestinal stromal tumors: proof of concept and therapeutic implications. Am J Surg Pathol. 2004;28:889-94.
- 26. Miettinen M, Sarlomo-Rikala M, Lasota J. Gastrointestinal stromal tumors: recent advances in understanding of their biology. Hum Pathol. 1999;30:1213-20.
- 27. Miettinen M, Sarlomo-Rikala M, Sobin LH, Lasota J. Gastrointestinal stromal tumors and leiomyosarcomas in the colon: a clinicopathologic, immunohistochemical, and molecular genetic study of 44 cases. Am J Surg Pathol. 2000;24:1339-52.
- 28. Miettinen M, Lasota J. Gastrointestinal stromal tumors - definition, clinical, histological, immunohistochemical, and molecular genetic features and differential diagnosis. Virchows Arch. 2001;438:1-12.
- 29. Miettinen M, Majidi M, Lasota J. Pathology and diagnostic criteria of gastrointestinal stromal tumors (GISTs): a review. Eur J Cancer. 2002;38(Suppl 5):s39-s51.
- 30. Nakamura N, Yamamoto H, Yao T, Oda Y, Nishiyama K, Imamura M, Yamada T, Nawata H, Tsuneyoshi M. Prognostic significance of expressions of cell-cycle regulatory proteins in gastrointestinal stromal tumor and the relevance of the risk grade. Human Pathol. 2005;36:828-37.
- 31. Ranchod M, Kempson RL. Smooth muscle tumors of the gastrointestinal tract and retroperitoneum. Cancer. 1977;39:255-62.
- 32. Schneider-Stock R, Boltze C, Lasota J, Miettinen M, Peters B, Pross M, Roessner A, Günther T. High prognostic value of p16INK4 alterations in gastrointestinal stromal tumors. J Clin Oncol. 2003;21:1688-97.
- 33. Taniguchi M, Nishida T, Hirota S, Isozaki K, Ito T, Nomura T, Matsuda H, Kitamura Y. Effect of c-KIT mutation on prognosis of gastrointestinal stromal tumors. Cancer Res. 1999;59:4297-300.
- 34. Toquet C, Le Neel JC, Guillou L. Elevated (>or=10%) MIB-1 proliferative index correlates with poor outcome in gastric stromal tumours patients: a study of 35 cases. Dig Dis Sci. 2002;47:2247-53.
- 35. Wang X, Mori I, Tang W, Utsunomiya H, Nakamura M, Nakamura Y, Zhou G, Kakudo K. Gastrointestinal stromal tumors: clinicopathological study of Chinese cases. Pathol Int. 2001;51:701-6.
- 36. Wang X, Mori I, Tang W, Utsunomiya H, Nakamura M, Nakamura Y, Zhou G, Kennichi K. Helpful parameter for malignant potential of gastrointestinal stromal tumors (GIST). Jpn J Clin Oncol. 2002;32:347-51.
- 37. Wong NA, Young R, Malcomson RD, Nayar AG, Jamieson LA, Save VE, Carey FA, Brewster DH, Han C, Al-Nafussi A. Prognostic indicators for gastrointestinal stromal tumours: a clinicopathological and immunohistochemical study of 108 resected cases of the stomach. Histopathology. 2003;43:118-26.
- 38. Zalupski M, Metch B, Balcerzak S, Fletcher WS, Chapman R, Bonnet JD, Weiss GR, Ryan J, Benjamin RS, Baker LH. Phase III comparison of doxorubicin and dacarbazine given by bolus versus infusion in patients with soft-tissue sarcomas: a Southwest Oncology Group study. J Natl Cancer Inst. 1991;83:926-32.
Correspondence:
Publication Dates
-
Publication in this collection
25 June 2009 -
Date of issue
June 2009
History
-
Received
03 July 2008 -
Accepted
20 Aug 2008