Abstracts
The trail making test (TMT) pertains to a family of tests that tap the ability to alternate between cognitive categories. However, the value of the TMT as a localizing instrument remains elusive. Here we report the results of a functional magnetic resonance imaging (fMRI) study of a verbal adaptation of the TMT (vTMT). The vTMT takes advantage of the set-shifting properties of the TMT and, at the same time, minimizes the visuospatial and visuomotor components of the written TMT. Whole brain BOLD fMRI was performed during the alternating execution of vTMTA and vTMTB in seven normal adults with more than 12 years of formal education. Brain activation related to the set-shifting component of vTMTB was investigated by comparing performance on vTMTB with vTMTA, a simple counting task. There was a marked asymmetry of activation in favor of the left hemisphere, most notably in dorsolateral prefrontal cortex (BA 6 lateral, 44 and 46) and supplementary motor area/cingulate sulcus (BA 6 medial and 32). The intraparietal sulcus (BA 7 and 39) was bilaterally activated. These findings are in line with clinico-anatomic and functional neuroimaging data that point to a critical role of the dorsolateral and medial prefrontal cortices as well as the intraparietal sulci in the regulation of cognitive flexibility, intention, and the covert execution of saccades/anti-saccades. Many commonly used neuropsychological paradigms, such as the Stroop, Wisconsin Card Sorting, and go - no go tasks, share some patterns of cerebral activation with the TMT.
trail making; functional MRI; executive function; prefrontal cortex; intraparietal sulcus; cognitive switching
O Teste de trilhas (TT) pertence a uma família de testes que aferem a capacidade de alternar entre categorias cognitivas. Não obstante, o valor de localização do TT permanece obscuro. Neste estudo, relatamos a análise de um estudo de uma adaptação verbal do TT (TTv) com ressonância magnética funcional (RMf). O TTv realça a propriedade de mudança de categorias do TT convencional (escrito) ao mesmo tempo em que minimiza seus componentes visuoespaciais e visuomotores. O cérebro inteiro foi estudado pela técnica BOLD durante a execução alternante do TTvA e TTvB em sete adultos normais com mais de 12 anos de escolaridade. A ativação cerebral relacionada ao componente de alternância cognitiva da Parte B foi investigada comparando-se o desempenho no TTvB e no TTvA. Observamos acentuada assimetria de ativação a favor do hemisfério esquerdo, especificamente no córtex frontal dorsolateral (áreas 6 lateral, 44 e 46) e área motora suplementar/sulco do cíngulo (área 6 medial e 32). O sulco intraparietal (áreas 7 e 39) foi ativado bilateralmente. Esses achados estão de acordo com dados clínico-anatômicos e de imagem funcional que apontam para o papel crítico do córtex prefrontal dorsolateral e medial, bem como do sulco intraparietal, na regulação da flexibilidade cognitiva, dos mecanismos de intenção e execução de saques e anti-saques oculares. Muitos paradigmas neuropsicológicos de amplo emprego na prática clínica, como os testes de Stroop e de Wisconsin, e tarefas do tipo vai / não vai, compartilham padrões de ativação cerebral com o TT.
teste de trilhas; ressonância magnética funcional; córtex prefrontal; sulco intraparietal; função executiva; alternância cognitiva
THE CEREBRAL CORRELATES OF SET-SHIFTING
An fMRI study of the trail making test
Jorge Moll1 1 Study conducted at the Barra D'Or Hospital, RJ Rio de Janeiro, Brazil: LABSRede D'Or, Grupo de Neuroimagem e Neurologia do Comportamento (GNNC), RJ; 2 Hospital Universitário Gaffrée e Guinle e Instituto Philippe Pihel. , Ricardo de Oliveira-Souza1 1 Study conducted at the Barra D'Or Hospital, RJ Rio de Janeiro, Brazil: LABSRede D'Or, Grupo de Neuroimagem e Neurologia do Comportamento (GNNC), RJ; 2 Hospital Universitário Gaffrée e Guinle e Instituto Philippe Pihel. ,2 1 Study conducted at the Barra D'Or Hospital, RJ Rio de Janeiro, Brazil: LABSRede D'Or, Grupo de Neuroimagem e Neurologia do Comportamento (GNNC), RJ; 2 Hospital Universitário Gaffrée e Guinle e Instituto Philippe Pihel. , Fernanda Tovar Moll1 1 Study conducted at the Barra D'Or Hospital, RJ Rio de Janeiro, Brazil: LABSRede D'Or, Grupo de Neuroimagem e Neurologia do Comportamento (GNNC), RJ; 2 Hospital Universitário Gaffrée e Guinle e Instituto Philippe Pihel. ,
Ivanei Edson Bramati1 1 Study conducted at the Barra D'Or Hospital, RJ Rio de Janeiro, Brazil: LABSRede D'Or, Grupo de Neuroimagem e Neurologia do Comportamento (GNNC), RJ; 2 Hospital Universitário Gaffrée e Guinle e Instituto Philippe Pihel. , Pedro Angelo Andreiuolo1 1 Study conducted at the Barra D'Or Hospital, RJ Rio de Janeiro, Brazil: LABSRede D'Or, Grupo de Neuroimagem e Neurologia do Comportamento (GNNC), RJ; 2 Hospital Universitário Gaffrée e Guinle e Instituto Philippe Pihel.
ABSTRACT - The trail making test (TMT) pertains to a family of tests that tap the ability to alternate between cognitive categories. However, the value of the TMT as a localizing instrument remains elusive. Here we report the results of a functional magnetic resonance imaging (fMRI) study of a verbal adaptation of the TMT (vTMT). The vTMT takes advantage of the set-shifting properties of the TMT and, at the same time, minimizes the visuospatial and visuomotor components of the written TMT. Whole brain BOLD fMRI was performed during the alternating execution of vTMTA and vTMTB in seven normal adults with more than 12 years of formal education. Brain activation related to the set-shifting component of vTMTB was investigated by comparing performance on vTMTB with vTMTA, a simple counting task. There was a marked asymmetry of activation in favor of the left hemisphere, most notably in dorsolateral prefrontal cortex (BA 6 lateral, 44 and 46) and supplementary motor area/cingulate sulcus (BA 6 medial and 32). The intraparietal sulcus (BA 7 and 39) was bilaterally activated. These findings are in line with clinico-anatomic and functional neuroimaging data that point to a critical role of the dorsolateral and medial prefrontal cortices as well as the intraparietal sulci in the regulation of cognitive flexibility, intention, and the covert execution of saccades/anti-saccades. Many commonly used neuropsychological paradigms, such as the Stroop, Wisconsin Card Sorting, and go - no go tasks, share some patterns of cerebral activation with the TMT.
KEY WORDS: trail making, functional MRI, executive function, prefrontal cortex, intraparietal sulcus, cognitive switching.
Correlatos cerebrais da flexibilidade cognitiva: RM funcional do teste das trilhas
RESUMO - O Teste de trilhas (TT) pertence a uma família de testes que aferem a capacidade de alternar entre categorias cognitivas. Não obstante, o valor de localização do TT permanece obscuro. Neste estudo, relatamos a análise de um estudo de uma adaptação verbal do TT (TTv) com ressonância magnética funcional (RMf). O TTv realça a propriedade de mudança de categorias do TT convencional (escrito) ao mesmo tempo em que minimiza seus componentes visuoespaciais e visuomotores. O cérebro inteiro foi estudado pela técnica BOLD durante a execução alternante do TTvA e TTvB em sete adultos normais com mais de 12 anos de escolaridade. A ativação cerebral relacionada ao componente de alternância cognitiva da Parte B foi investigada comparando-se o desempenho no TTvB e no TTvA. Observamos acentuada assimetria de ativação a favor do hemisfério esquerdo, especificamente no córtex frontal dorsolateral (áreas 6 lateral, 44 e 46) e área motora suplementar/sulco do cíngulo (área 6 medial e 32). O sulco intraparietal (áreas 7 e 39) foi ativado bilateralmente. Esses achados estão de acordo com dados clínico-anatômicos e de imagem funcional que apontam para o papel crítico do córtex prefrontal dorsolateral e medial, bem como do sulco intraparietal, na regulação da flexibilidade cognitiva, dos mecanismos de intenção e execução de saques e anti-saques oculares. Muitos paradigmas neuropsicológicos de amplo emprego na prática clínica, como os testes de Stroop e de Wisconsin, e tarefas do tipo vai / não vai, compartilham padrões de ativação cerebral com o TT.
PALAVRAS-CHAVE: teste de trilhas , ressonância magnética funcional, córtex prefrontal, sulco intraparietal, função executiva, alternância cognitiva.
The ability to switch between cognitive categories, or "sets", is a classical indicator of normal neurobehavioral functioning1. The trail marking test (TMT) is widely employed as a diagnostic tool for eliciting shifts between cognitive sets2. It consists of two parts in which subjects must connect 25 circles containing numbers (Part A), or numbers and letters (Part B) arrayed pseudorandomly on a letter-size sheet of paper3. In Part A (TMTA) the circles are numbered from 1 to 25, whereas in Part B (TMTB) they contain numbers from 1 to 13 and letters from A to M (the Portuguese alphabet does not include the letter K), which must be connected in alternating progression, from 1-A to M-13. Total score is the time in seconds spent to complete each part. In view of the low localizing and lateralizing value of the TMT4,5, the cerebral areas that are critical for its execution have been difficult to define on the basis of clinico-anatomic data alone6. A verbal adaptation of the TMT (vTMT) has been increasingly employed in normal individuals as well as in mixed clinical samples7,8. The vTMT is more suited for probing the ability to switch between cognitive sets because it reduces to a minimum the visuospatial and visuomotor factors of the written format9.
In the present study we used functional magnetic resonance imaging (fMRI) to investigate the cerebral correlates of the vTMT, while attempting to preserve the basic cognitive structure of vTMT as a neuropsychologic instrument.
METHOD
Neuropsychological paradigm
Seven right-handed normal volunteers (three men) with ages ranging from 19 to 43 years (mean = 24±9 years) participated in the study after providing written informed consent. Subjects had more than 12 years of formal education and a mean Edinburgh Inventory score10 of 82 ± 3011. Before scanning, subjects were administered the written version of the TMT and rehearsed the verbal adaptation of the test. To match the scanning procedure as closely as possible, a few trials of 25 seconds each were administered during the training session. Subjects were asked to start counting covertly at 1 (vTMTA) and to alternate between numbers and letters (vTMTB) as quickly as possible when they heard the commands "count" and "alternate", respectively. To prevent unsolvable anagram effects, subjects were told to start over if they slipped off the track. During scanning, the procedure was repeated. Besides, they were asked to keep their eyes shut and to avoid making articulatory movements. After the scanning session, subjects rated the attentional effort allocated to each part of the test on 100 mm visual analogue scales (VAS) and reported how far they had progressed, on average, on each part of the test12. Total scores for vTMTA and vTMTB were, respectively, the highest number and letter reached in their best trials. For the purposes of data analysis, the numerical order of that letter in the alphabet was doubled and used as the final vTMTB score.
Statistical analysis of cognitive ratings
We obtained six cognitive measures from each participant, namely, TMT (A and B), vTMT (A and B), and the VAS for attentional effort for each part of vTMT. Performance indexes on Parts A and B of each task were compared with Student's t-test for paired samples, two-tailed11. To know whether any possible differences between vTMT A and B would survive the confounding effects of attentional effort and the explicit visuospatial/visuomotor factors of the written format, we compared vTMT A and B performances with analysis of covariance entering TMT A and B, and the VAS scores as covariates. Means differences were assessed post hoc with Scheffé's test. A 0.05 level of significance, two-tailed, was set for all statistical tests.
fMRI procedures
Functional data covering the whole brain were acquired on a Siemens Vision 1.5T scanner. One hundred functional volumes were collected during the alternation of vTMTA and vTMTB in a blocked design, each block lasting 25 seconds. There were 10 blocks for part A and ten blocks for part B of the test. Sequence parameters were: gradient-echo EPI, TE = 66 ms, TR = 5 s, flip angle = 90°, slice thickness 5.0 mm, inter-slice gap 0.25 mm, FOV = 250 mm, 128x128 matrix. Data were analyzed using BrainVoyager v. 3.9 (Brain Innovation, Germany). Functional data were motion corrected in three-dimensional space, and spatial (4mm FWHM) and temporal (3-32Hz) smoothing were performed in the frequency domain. Intersubject analysis was performed using the general linear model approach. Activation maps were created using cross-correlation analysis, transformed into the Talairach space, and coregistered to the anatomic MRI. P-values < 0.05, corrected for multiple comparisons, with a 3D cluster threshold of 200 voxels in volumetric interpolated data were considered significant13. Lateralization indexes for regions of interest (ROI) were computed following an adaptation of Oldfield's formula for handedness10, which took into account the number of voxels (NOV) per ROI:
RESULTS
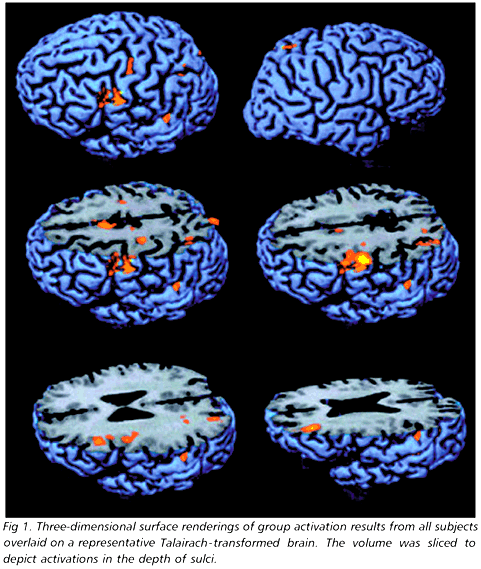
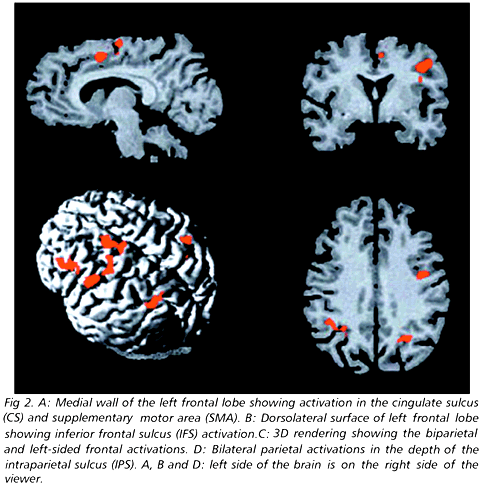
Both TMTB and vTMTB were lengthier than their A counterparts. Also, vTMTB demanded more attentional effort than vTMTA (Table 1). Nevertheless, vTMTB differed from vTMTA even after TMT and VAS ratings were statistically controlled [F(1,5) = 53.2, p < 0.002]. Areas of increased activation during performance of vTMTB as compared to vTMTA are shown in Table 2 and in Figs 1 and 2. The left hemisphere was more activated than the right, with an overall lateralization index of -0.63. Activations peaked in the lower third of dorsolateral prefrontal cortex, premotor cortex, left medial frontal cortex and bilaterally in the intraparietal sulcus (IPS). The frontal lateralization index was -1.00, with all activations falling in the left hemisphere. The parietal lateralization index was 0.37. A closer look at the coronal and axial cuts on Figure 2 shows that most activations fall within cortical sulci.
DISCUSSION
The TMT pertains to a family of neuropsychological tests which tap the ability to inhibit prepotent responses in favor of less habitual, yet more adaptive, ones. Other members of the family include the go-no go and the Stroop tasks14. The TMT differs from both, critically, because it requires verbal thinking to follow a predetermined conceptual route, which is given by the ordered series of numbers (part A), or numbers and letters (part B). Notwithstanding this fact, the similarities among these tasks may outweigh the differences. Behaviorally, all seem to share a common ground of cognitive interference, response conflict, and cognitive set shifting. Moreover, functional neuroimaging studies have shown that the areas activated by both the Stroop and the go-no go tasks overlap to a great extent, most notably in the middle dorsolateral and medial prefrontal cortex as well as in the posterior parietal cortex15,16. This pattern is remarkably similar to the one we found for the TMT.
The anterior hemispheric asymmetry seen in our subjects concurs with evidence implicating the left hemisphere in symbolic tasks. It may also reflect some structural peculiarities of vTMT proper. In support of this view, the conceptual handling of numbers was associated with activation of the left inferior frontal gyrus and sulcus, premotor cortex, and inferior parietal lobule17. The left inferior prefrontal cortex was likewise found to be more active in letter than in semantic fluency tasks18. The left prefrontal sectors activated in our study largely overlap with the inferior prefrontal area of other investigations which implicate the inferior frontal gyri and sulci in the mediation of cognitive switching. Thus, while the right prefrontal cortex seems to be critical for response suppression in go-no go tasks, the left is more directly involved in biasing responses towards novel principles at play15,16,19,20. The role of the inferior dorsolateral prefrontal cortex in providing flexibility to behavior is strengthened by the observation that cognitive inflexibility, or "perseveration", is a core symptom of damage to these areas21. The left premotor cortex activation seen in our subjects may be related to the task switching demands of vTMTB. This area was also activated in a study of the neural correlates of tool-use which required rapid manual shifts of tool-related actions22. The left premotor cortex was activated regradless of which hand was employed in the task, or whether real or imagined tool-use actions were performed. These observations provide additional evidence that the anterior half of the left hemisphere is critically involved in tasks that depend on rapid action and cognitive shifts.
An alternative, or complementary, explanation for the inferior dorsolateral activation involving working memory mechanisms might be raised. That this was not the case, however, is suggested by the fact that both vTMT A and B required comparable instructions (i.e. "count" and "alternate"), thus equalizing the working memory load across blocks. This conclusion is supported by the absence of activation in BA 9, which is related to the holding of instructions in working memory during task execution23.
The contribution of the medial frontal cortex to normal TMTB performance had already been suggested by case reports of patients with damage to this region24 or to its subcortical connections25,26. These individuals easily lose track of the alternating response pattern. The supracingular cluster of activation in our subjects was located in an area slightly posterior to the plane of the anterior commissure, corresponding to the supplementary motor area27. This area exerts a facilitatory influence on movement preparation and initiation or, more simply, on "intention"28. The fact that bilateral damage to the medial frontal lobe leads to loss of spontaneity and behavioral slowness29, while unilateral damage gives rise to the clinical syndrome of "motor neglect"30 lends support to the hypothesis that the frontomedial activation observed in the present study reflects the call into play of processes which lie at the intention-action interface. The absence of anterior cingulate activation is in agreement with recent studies showing that cognitive interference is primarily mediated by the cortex of the left inferior frontal sulcus, while the anterior cingulate is more directly related to response preparation28,31. Since our study protocol reduced overt motor responses to a minimum, it is not surprising that the anterior cingulate was not significantly activated in our subjects.
The cortex within and around the IPS is a complex somatosensory and visual convergence zone that has been functionally related to object and spatial working memory, visual search, tool-use praxis, reaching, and reallocation of attentional resources22,32. However, only a few studies have addressed the meaning of IPS activation in paradigms employing cognitive switching31. A pattern of bilateral IPS activation similar to the one found in the present investigation has been observed in association with the generation of anti-saccades, covert visual orienting and suppression of oculomotor responses33. Although we did not monitor the eye movements of our subjects, the possibility that bilateral IPS activation during vTMTB performance is related to the covert interplay of saccades and anti-saccades while the individual mentally tracks his progression along the conceptual letter-number trail deserves further experimental testing34.
The observation that activations tended to fall within cortical sulci adds to the evidence in favor of the "fundal cognition hypothesis"35. According to this hypothesis, the neural computations underlying complex cognitive operations take place preferentially within cortical sulci, in contrast to the gyral surfaces. The fundal cognition hypothesis is in keeping with the complexity of the operations involved in trail making.
Studies as the present one may provide new insights into the brain circuits underlying the execution of common neuropsychological tests by normal individuals and shed light on the mechanisms of impairment and recovery of function in patients with brain disease. They may also improve neuropsychological test design by refining cognitive dimensions of interest on the basis of brain activation patterns.
Acknowledgements - The authors are indebted to Prof. Omar da Rosa Santos, Hospital Universitário Gaffrée e Guinle.
Received 12 April 2002. Accepted 24 June 2002.
Dr. Jorge Moll - Avenida Ayrton Senna 2541 - 22793-010 Rio de Janeiro, RJ - Brasil. E-mail: jorgemoll@neuroimage.com
- 1. Freeman T, Gathercole CE. Perseveration-the clinical symptoms-in chronic schizophrenia and organic dementia. Bri J Psychiatry 1966;112:27-32.
- 2. Butler M, Retzlaff P, Vanderploeg R. Neuropsychological test usage. Professional Psychology: Research and Practice 1991;22:510-512.
- 3. Reitan RM. Validity of the Trail Making Test as an indicator of organic brain damage. Perceptual and Motor Skills 1958;8:271-276.
- 4. Lezak MD. Neuropsychological assessment. 3.Ed. New York: Oxford Univ Press, 1995:381-384.
- 5. Reitan RM, Tarshes EL. Differential effects of lateralized brain lesions on the Trail Making Test. J Nervous Mental Dis 1959;129:257-262.
- 6. Spreen O, Strauss E. A Compendium of neuropsychological tests: administration, norms, and commentary. 2.Ed. New York: Oxford Univ Press, 1998.
- 7. Abraham E, Axelrod BN, Ricker JH. Application of the Oral Trail Making Test to a mixed clinical sample. Archi Clini Neuropsychol 1996;11:697-701.
- 8. Oliveira-Souza R, Moll J, Passman LJ, et al. Trail making and cognitive set-shifting. Arq Neuropsiquiatr 2000;58:826-829.
- 9. Crowe SF. The differential contribution of mental tracking, cognitive flexibility, visual search, and motor speed to performance on Parts A and B of the Trail Making Test. J Clin Psychol 1998;54:585-591.
- 10. Oldfield RC. The assessment and analysis of handedness: The Edinburgh Inventory. Neuropsychologia 1971;9:97-113.
- 11. Kachigan SK. Statistical analysis. an interdisciplinary introduction to univariate & multivariate methods. New York: Radius Press, 1986:589.
- 12. McCormack HM, Horne DJ, Sheather S. Clinical applications of visual analogue scales: a critical review. Psychol Med 1988;18:1007-1019.
- 13. Forman SD, Cohen JD, Fitzgerald M, Eddy WF, Mintun MA, Noll DC. Improved assessment of significant activation in functional magnetic resonance imaging (fMRI): use of a cluster size threshold. Magnetic Resonance Med 1995;33:636-647.
- 14. Weintraub S. Neuropsychological assessment of mental state. In Mesulan MM (Ed.) Principles of behavioral and cognitive neurology. 2.Ed. New York: Oxford Univ Press, 2000:121-173.
- 15. Garavan H, Ross TJ, Stein EA. Right hemispheric dominance for inhibitory control: an event-related functional MRI study. Proc Natl Acad Sci USA 1999;96:8301-8306.
- 16. Kawashima R, Satoh K, Itoh H, et al. Functional anatomy of GO/NO-GO discrimination and response selection: a PET study in man. Brain Res 1996;728:79-89.
- 17. Rickard TC, Romero SG, Basso G, Wharton C, Flitman S, Grafman J. The calculating brain: an fMRI study. Neuropsychologia 2000;38:325-335.
- 18. Paulesu E, Goldacre B, Scifo P, et al. Functional heterogeneity of left inferior frontal cortex as revealed by fMRI. NeuroReport 1997;8:2011-2016.
- 19. Dove A, Pollmann S, Schubert T, Wiggins CJ, von Cramon DY. Prefrontal cortex activation in task switching: an event-related fMRI study. Cogn Brain Res 2000;9:103-109.
- 20. Konishi S, Kawazu M, Uchida I, Kikyo H, Asakura I, Miyashita Y. Contribution of working memory to transient activation in human inferior prefrontal cortex during performance of the Wisconsin Card Sorting Test. Cer Cortex 1999;9:745-753.
- 21. Stuss DT, Levine B, Alexander MP, et al. Wisconsin Card Sorting Test performance in patients with focal frontal and posterior brain damage: effects of lesion location and test structure on separable cognitive processes. Neuropsychologia 2000;38:388-402.
- 22. Moll J, Oliveira-Souza R, Passman LJ, Cunha FC, Lima FS, Andreiuolo PA. Functional magnetic resonance imaging of pantomimes of tool use. Neurology 2000;54:1331-1336.
- 23. MacDonald AW, Cohen JD, Stenger VA, Carter CS. Dissociating the role of the dorsolateral prefrontal and anterior cingulate cortex in cognitive control. Science 2000;288:1835-1838.
- 24. Leimkuhler ME, Mesulam MM. Reversible go-no go deficits in a case frontal lobe tumor. Ann of Neurol 1985;18:617-619.
- 25. Sandson TA, Daffner KR, Carvalho PA, Mesulam MM. Frontal lobe dysfunction following infarction of the left-sided medial thalamus. Arch of Neurol 1991;48:1300-1303.
- 26. Jahanshahi M, Ardouin CMA, Brown RG, et al. The impact of deep brain stimulation on executive function in Parkinson's disease. Brain 2000;123;1142-1154.
- 27. Humberstone M, Sawle GV, Clare S, et al. Functional magnetic resonance imaging of single motor events reveals human presupplementary motor area. Ann Neurol 1997;42:632-637.
- 28. Heilman KM, Valenstein E, Watson RT. Intentional motor disorders. In Levin HS, Eisenberg HM, Benton AL (Eds.). Frontal lobe function and dysfunction. New York: Oxford Univ Press, 1991:199-213.
- 29. Fisher CM. Abulia minor vs agitated behavior. Clin Neurosurg 1984;31:9-31.
- 30. Laplane D, Degos JD. Motor neglect. J Neurol Neurosurg Psychiatry 1983;46:152-158.
- 31. Zysset S, Müller K, Lohmann G, von Cramon DY. Color-word matching Stroop task: separating interference and response conflict. Neuroimage 2001;13:29-36.
- 32. Belger A, Puce A, Krystal JH, Gore JC, Goldman-Rakic P, McCarthy G. Dissociation of mnemonic and perceptual processes during spatial and nonspatial working memory using fMRI. Hum Brain Mapp 1998;6:14-32.
- 33. Connolly JD, Goodale MA, Desouza JFX, Menon RS, Vilis T. A comparison of frontoparietal fMRI activation during anti-saccades and anti-pointing. J Neurophysiol 2000;84:1645-1655.
- 34. Knauff M, Kassubek J, Mulack T, Greenlee MW. Cortical activation evoked by visual mental imagery as measured by fMRI. NeuroReport 2000;11:3957-3962.
- 35. Markowitsch HJ, Tulving E. Cognitive processes and cerebral cortical fundi: Findings from positron-emission tomography studies. Proc Natl Acad Sci USA 1994;91:10507-10511.
Publication Dates
-
Publication in this collection
14 Jan 2003 -
Date of issue
Dec 2002
History
-
Accepted
24 June 2002 -
Received
12 Apr 2002