Abstracts
Dysembryoplastic neuroepithelial tumor (DNT), described in 1988 and introduced in the WHO classification in 1993, affects predominantly children or young adults causing intractable complex partial seizures. Since it is benign and treated with surgical resection, its recognition is important. It has similarities with low-grade gliomas and gangliogliomas, which may recur and become malignant. OBJECTIVES: To investigate whether DNT was previously diagnosed as astrocytoma, oligodendroglioma, or ganglioglioma and to determine its frequency in a series of low-grade glial/glio-neuronal tumors. METHODS: Clinical, radiological, and histological aspects of 58 tumors operated from 1978 to 2008, classified as astrocytomas (32, including 8 pilocytic), oligodendrogliomas (12), gangliogliomas (7), and DNT (7), were reviewed. RESULTS: Four new DNT, one operated before 1993, previously classified as astrocytoma (3) and oligodendroglioma (1), were identified. One DNT diagnosed in 2002 was classified once more as angiocentric glioma. Therefore, 10 DNT (17.2%) were identified. CONCLUSIONS: Clinical-radiological and histopathological correlations have contributed to diagnose the DNT.
dysembryoplastic neuroepithelial tumor; low-grade gliomas; epilepsy
O tumor neuroepitelial disembrioplásico (DNT), descrito em 1988 e incorporado na classificação da OMS em 1993, acomete predominantemente crianças ou adultos jovens, causando crises convulsivas parciais complexas farmacorresistentes. Como é benigno e tratável com ressecção cirúrgica, seu reconhecimento é importante. Tem semelhanças com gliomas de baixo grau e gangliogliomas, que podem recidivar e malignizar. OBJETIVOS: Investigar se o DNT foi originalmente diagnosticado como astrocitoma, oligodendroglioma ou ganglioglioma e determinar sua frequência numa série de neoplasias gliais/glioneuronais de baixo grau. MÉTODOS: Foram revistos aspectos clínicos, radiológicos e histológicos de 58 neoplasias operadas entre 1978 e 2008, classificadas como astrocitomas (32, sendo 8 pilocíticas), oligodendrogliomas (12), gangliogliomas (7) e DNT (7). RESULTADOS: Foram identificados quatro novos DNT, um operado antes de 1993, originalmente diagnosticado como astrocitoma (3) e oligodendroglioma (1). Um DNT diagnosticado em 2002 foi reclassificado como glioma angiocêntrico. Portanto, 10 DNT (17,2%) foram identificados. CONCLUSÕES: Correlações clínico-radiológicas e histopatológicas contribuíram para o diagnóstico do DNT.
tumor neuroepitelial disembrioplásico; gliomas de baixo grau; epilepsia
ARTICLE
Dysembryoplastic neuroepithelial tumor originally diagnosed as astrocytoma and oligodendroglioma
Tumor neuroepitelial disembrioplásico diagnosticado originalmente como astrocitoma ou oligodendroglioma
Diego Cassol DozzaI; Flávio Freinkel RodriguesII; Leila ChimelliIII
INeurosurgeon; Master's degree at the Universidade Federal do Rio de Janeiro (UFRJ), Rio de Janeiro RJ, Brazil
IINeurosurgeon, Surgery Department, UFRJ, Rio de Janeiro RJ, Brazil
IIIDepartment of Pathology, School of Medicine, UFRJ; Division of Pathology, National Institute of Cancer, Rio de Janeiro RJ, Brazil
Correspondence Correspondence: Leila Chimelli Divisão de Patologia do INCA, DIPAT Rua Cordeiro da Graça 156 20220-400 Rio de Janeiro RJ - Brasil E-mail: lchimelli@inca.gov.br
ABSTRACT
Dysembryoplastic neuroepithelial tumor (DNT), described in 1988 and introduced in the WHO classification in 1993, affects predominantly children or young adults causing intractable complex partial seizures. Since it is benign and treated with surgical resection, its recognition is important. It has similarities with low-grade gliomas and gangliogliomas, which may recur and become malignant.
OBJECTIVES: To investigate whether DNT was previously diagnosed as astrocytoma, oligodendroglioma, or ganglioglioma and to determine its frequency in a series of low-grade glial/glio-neuronal tumors.
METHODS: Clinical, radiological, and histological aspects of 58 tumors operated from 1978 to 2008, classified as astrocytomas (32, including 8 pilocytic), oligodendrogliomas (12), gangliogliomas (7), and DNT (7), were reviewed.
RESULTS: Four new DNT, one operated before 1993, previously classified as astrocytoma (3) and oligodendroglioma (1), were identified. One DNT diagnosed in 2002 was classified once more as angiocentric glioma. Therefore, 10 DNT (17.2%) were identified.
CONCLUSIONS: Clinical-radiological and histopathological correlations have contributed to diagnose the DNT.
Key words: dysembryoplastic neuroepithelial tumor, low-grade gliomas, epilepsy.
RESUMO
O tumor neuroepitelial disembrioplásico (DNT), descrito em 1988 e incorporado na classificação da OMS em 1993, acomete predominantemente crianças ou adultos jovens, causando crises convulsivas parciais complexas farmacorresistentes. Como é benigno e tratável com ressecção cirúrgica, seu reconhecimento é importante. Tem semelhanças com gliomas de baixo grau e gangliogliomas, que podem recidivar e malignizar.
OBJETIVOS: Investigar se o DNT foi originalmente diagnosticado como astrocitoma, oligodendroglioma ou ganglioglioma e determinar sua frequência numa série de neoplasias gliais/glioneuronais de baixo grau.
MÉTODOS: Foram revistos aspectos clínicos, radiológicos e histológicos de 58 neoplasias operadas entre 1978 e 2008, classificadas como astrocitomas (32, sendo 8 pilocíticas), oligodendrogliomas (12), gangliogliomas (7) e DNT (7). RESULTADOS: Foram identificados quatro novos DNT, um operado antes de 1993, originalmente diagnosticado como astrocitoma (3) e oligodendroglioma (1). Um DNT diagnosticado em 2002 foi reclassificado como glioma angiocêntrico. Portanto, 10 DNT (17,2%) foram identificados.
CONCLUSÕES: Correlações clínico-radiológicas e histopatológicas contribuíram para o diagnóstico do DNT.
Palavras-Chave: tumor neuroepitelial disembrioplásico, gliomas de baixo grau, epilepsia.
The dysembryoplastic neuroepithelial tumor (DNT), which was described by Daumas-Duport et al., in 19881, and it was incorporated in the World Health Organization (WHO) classification in 19932, is a benign mixed glio-neuronal tumor usually supratentorial in the cerebral cortex, which occurs predominantly in children or young adults, who present intractable complex partial seizures before 20 years-old and absence of focal neurological deficit. Radiologically, cortical topography of the lesion, no mass effect or contrast enhancement, except in a small proportion of cases, iso or hypointensity in T1-weighted and hiperintensity in T2-weighted magnetic resonance imaging (MRI) are the main features3. Histologically, it typically shows a columnar pattern and multinodular architecture, which is often associated with cortical dysplasia. There are three histological subtypes: complex, simple, and non-specific forms, which may show similarities with low-grade diffuse gliomas3. As for the ganglioglioma, which is usually benign but may recur and become anaplastic, there are clinical, topographical and radiological similarities with the DNT, such as the frequent occurrence of epilepsy and the preference for the temporal lobe4.
Identification of DNT has therapeutic and prognostic implications, since surgical resection may provide good long-term seizure control in the majority of young patients, with epileptogenic glioneuronal tumors5 and aggressive therapy can be avoided, thus sparing these young patients of the deleterious long-term effects of radio- or chemotherapy3.
The aims of this study were to investigate whether the DNT had been previously diagnosed as low-grade astrocytoma, oligodendroglioma or ganglioglioma, due to the lack of correlations with clinical and radiological presentations or because it had been analyzed before its recognition by the WHO, in 1993. In addition, we intended to determine its frequency in a series of low-grade glial or glio-neuronal tumors.
METHODS
This is a retrospective study undertaken in two neurosurgical centers of the Federal University of Rio de Janeiro (UFRJ), Clementino Fraga Filho University Hospital (HUCFF) and the Institute of Neurology Deolindo Couto (INDC), from 1978 to 2008. At the INDC, this period was shorter because neurosurgical specimens were available from 1997 to 2007. We reviewed the medical records of all patients who underwent surgery for resection of low-grade astrocytomas, oligodendrogliomas, gangliogliomas, and DNT. Patients with incomplete data, such as the lack of histological blocks and radiological images, were excluded.
In a series of 908 brain tumors (704 in HUCFF and 204 in the INDC), there were 94 astrocytomas, 35 oligodendrogliomas, 18 gangliogliomas, and 7 DNT. After excluding those without all the required information, 58 tumors were selected, with the following histological diagnostics: 32 astrocytomas (8 were pilocytic), 12 oligodendrogliomas, 7 gangliogliomas, and 7 DNT.
The histological diagnoses were reviewed by a neuropathologist. If necessary, new paraffin sections stained with hematoxylin and eosin were prepared. Seven cases required immunohistochemical study to support the diagnosis. The following antibodies were used: epithelial membrane antigen (EMA - dilution 1:80, clone, p. 28-29, DAKO), synaptophysin (SF - 1:30 dilution, clone Snp88, BIOGENEX); neurofilament (NF-1: 100 dilution, clone RT97, NOVOCASTRA), glial fibrillary acidic protein (GFAP - dilution 1: 100, clone GA5, NOVOCASTRA), and Ki67 (dilution 1:50, clone Ki88, BIOGENEX), chromogranin (CGR - 1:50 dilution, clone LK2H10, NOVOCASTRA).
This study was approved by the Ethics Committee in Research of the INDC-UFRJ.
RESULTS
Four new DNT were identified. They were previously classified as astrocytoma (three cases) and oligodendroglioma (one case). Only one was operated before 1993. One case previously diagnosed as DNT in 2002 was re-classified as angiocentric glioma. Therefore, there were ten cases of DNT in this sample (17.2%), four of them re-classified after the revision, taking into account the clinical, radiological, and histological criteria.
Mean age at the onset of symptoms was 11.9 years-old (range - from 2 to 26 years-old), the average time of seizure disabilities to diagnosis was 94.1 months (range 0.4 to 360 months), the average age at the time of surgery was 26.1 years-old (range 13 to 45 years-old) and the average time of follow-up was 59.5 months (range 3 to 156 months). The topography was quite variable. Temporal, parietal, and frontal lobes were involved in three cases (30%), and the occipital lobe in one (10%).
Seizures were present in all cases, six (60%) with intractable epilepsy and four (40%) responded to the antiepileptic drug (AED). Simple and complex partial seizures occurred in four (40%) patients each, while primary generalized tonic-clonic seizures were seen in two (20%) cases.
Cerebral computed tomography (CT) scan was performed on eight patients, all showing hypodensity in the region of tumor. Calcification was present in three (37.5%), in one (12.5%) tumor there was contrast enhancement and two (25%) cases had mild mass effect in the surrounding brain. Skull deformation occurred in one (12.5%) case. The characteristics of patients with DNT are showed in the Table. MRI was performed in seven patients and the main characteristics were hypointensity in T1-weighted and hiperintensity in T2-weighted image (Fig 1), which was present in all cases. There was contrast enhancement in three (42.8%), two of them heterogeneous and one ring-shaped. Perilesional edema was observed in two (28.6%) cases and mass effect due to cyst was observed in one (14.3%) case (Fig 2).
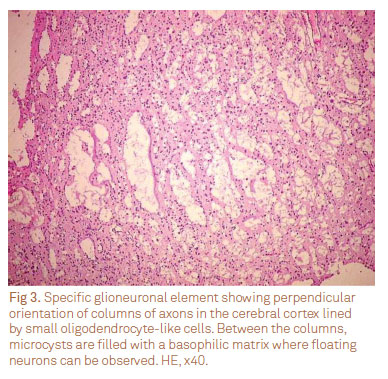
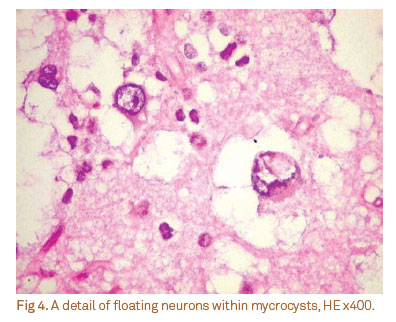
The histological subtypes were four (40%) complex, five (50%) simple, and one (10%) non-specific forms. Other histological features included calcification (three cases) and cortical dysplasia (one case). Figs 3 and 4 show the classical histological features of the DNT.
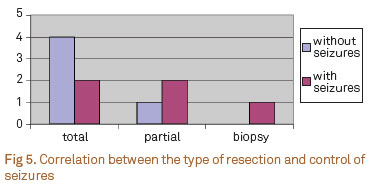
Six patients (60%) diagnosed with DNT had total resection of the tumor, four of which (66.6%) were free of seizures, one without medications and three taking only one medication. The two (33.3%) that remained with seizures, one was taking one AED and the other three AED. The control of seizures was obtained after partial resection in one out of three patients. The patient who underwent biopsy was not free of seizures. Therefore, seizures were controlled in five patients (50%) after surgery (Fig 5).
Considering the four cases that were re-classified as DNT, three had the complex histological features and one had the simple form. The radiological features of two were typical, but one had contrast enhancement and mass effect due to edema and another had calcification. As for the resection and seizure control, the two with typical radiological features had total resection and seizure control, while the other two (one partial resection, one biopsy) were not free of seizures.
DISCUSSION
It has been more than 20 years that Daumas-Duport et al.1 described and proposed the new tumor entity called dysembryoplastic neuroepithelial tumor. Despite the fact that nowadays, there is much more knowledge about the subject and the clinical and radiological criteria are well-established, diagnostic difficulties may remain due to the complexity of some histological and radiological presentations6.
We identified 10 DNT in this series of 58 low-grade gliomas/glioneuronal tumors, 4 of which were re-classified after revision of the clinical, radiological, and histological data. Their original diagnoses were astrocytoma in three cases and oligodendroglioma in one, only one operated before 1993. The re-classification of a tumor, which had been previously diagnosed as DNT as angiocentric glioma, was due to the fact that it was operated in 2002 and this entity, that may resemble a DNT, was recognized by the WHO, in 20077.
The frequency of DNT in our series (17.2%) cannot reliably compare with the literature, partly because of the small number of cases, but also because variable estimations have been obtained according to the type of recruitment of patients, including tumors that were operated on for pharmacoresistant epilepsy, low-grade tumors, pediatric series, etc. For instance, in a series of 38 patients with low-grade tumors with intractable epilepsy, DNT were found in 26.3%8; among 340 primary central nervous system (CNS) tumors in children up to 17 years of age, DNT occurred in 0.6% of the cases9, and out of 129 patients less than 19 years of age, 17 (13.2%) were DNT10. In addition, among all neuroepithelial tumors diagnosed in one Institution comprising 22 years11, the DNT was diagnosed in 1.2% of patients under age 20 and in 0.24% of patients over 20 years.
Seizures were present in all our cases and began early in life, as reported in most series9,10,12. This was an important criterion to re-classify the four DNT.
Imaging findings in our cases also corroborated those in the literature13-15. However, radiological features of two out of the four re-classified DNT were not so typical, one with contrast enhancement and mass effect caused by edema and another with calcification. Although these findings have been described in various series of DNT15-17, they may have accounted for the misdiagnosis of a low-grade diffuse glioma (astrocytoma or oligodendroglioma) in our cases. As for the histological presentations, all four re-classified tumors presented the simple (one case) or complex (three cases) forms, as classically described in the literature1,2,18. Even not justifying the previous diagnosis of low-grade diffuse glioma, it is worth mentioning that complex variants have been appointed to induce an occasional glioma misdiagnosis19, because they have a different clinical profile and a more variable histopathological and MRI appearance. In addition, other recently described variants (diffuse and associated with ganglioglioma)6, together with the nonspecific form3, may also induce misdiagnosis, if one is not aware of the classical clinical and radiological criteria. Another reason for not having diagnosed DNT in one of our cases is that the sample was probably not representative of the lesion, since the patient was submitted to a biopsy. It is well-known that larger samples are usually necessary to show the classical architecture of a DNT3.
Control of seizures was obtained in 50% of the patients, as opposed to 80 to 100%20-22, which is directly related to the extent of resection8,12,23. We could observe that the management of seizures was more effective after total resection of the lesion than with partial resection, which has also been applied for the four re-classified tumors, one of which underwent a biopsy, and as expected, did not improve clinically. Patients with complete resection had the best seizure control taking less antiepileptic drugs.
In conclusion, DNT are benign tumors with varying characteristics and require careful clinical and radiological assessment to assist the histopathologic diagnosis and avoid additional therapy. Complete resection of the lesion should be the main goal of surgery, while isolated stereotactic biopsy should be avoided, because, not only the diagnosis may not be confirmed, however more importantly, the seizure control cannot be reached.
References
1. Daumas-Duport C, Scheithauer BW, Chodkiewicz JP, Laws Jr ER, Vedrenne C. Dysembryoplastic neuroepithelial tumor: a surgically curable tumor of young patients with intractable partial seizures. Report of thirty-nine cases. Neurosurgery 1988:23:545-556.
2. Kleihues P, Burger PC, Scheithauer BW. The new WHO classification of brain tumors. Brain Pathol 1993:3:255-268.
3. Daumas-Duport C, Pietsch T, Hawkins C. Dysembryoplastic neuroepithelial tumor. In: Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (Eds). WHO classification of tumours of the central nervous system. 4th ed. Lyon, France: IARC Press; 2007: 99-102.
4. Becker AJ, Wiestler OD, Shankar SK, Figarella-Branger D, Blümcke I. Gangliocytoma/Ganglioglioma. In: Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (Eds). WHO classification of tumours of the central nervous system. 4th ed. Lyon, France: IARC Press; 2007:103-105.
5. Giulioni M, Galassi E, Zucchelli M, Volpi L. Seizure outcome of lesionectomy in glioneuronal tumors associated with epilepsy in children. J Neurosurg 2005;102(Suppl 3):288-293.
6. Thom M, Toma A, An S, et al. One hundred and one dysembryoplastic neuroepithelial tumors: an adult epilepsy series with immunohistochemical, molecular genetic, and clinical correlations and a review of the literature. J Neuropathol Exp Neurol 2011;70:859-878.
7. Burger PC, Jouvet A, Preusser M, Hans VH, Rosenblum MK, Lellouch-Tubiana A. Angiocentric glioma. In: Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (Eds). WHO classification of tumours of the central nervous system. 4th ed. Lyon, France: IARC Press; 2007: 92-93.
8. Ruban D, Byrne RW, Kanner A, et al. Chronic epilepsy associated with temporal tumors: long-term surgical outcome. Neurosurg Focus 2009;27:e6.
9. Rickert CH, Paulus W. Epidemiology of central nervous system tumors in childhood and adolescence based on the new WHO classification. Childs Nerv Syst 2001;17:503-511.
10. Prayson RA. Tumours arising in the setting of paediatric chronic epilepsy. Pathology 2010;42:426-431.
11. Rosemberg S, Vieira GS. Tumor Neuroepithelial Disembrioplásico. Um estudo epidemiológico de uma única instituição. Arq Neuropsiquiatr 1998;56:232-236.
12. Nolan MA, Sakuta R, Chuang N, et al. Dysembryoplastic neuroepithelial tumors in childhood: long-term outcome and prognostic features. Neurology 2004;62:2270-2276.
13. Stanescu Cosson R, Varlet P, Beuvon F, et al. Dysembryoplastic neuroepithelial tumors: CT, MR findings and imaging follow-up: a study of 53 cases. J Neuroradiol 2001;28:230-240.
14. Lee DY, Chung CK, Hwang YS, et al. Dysembryoplastic neuroepithelial tumor: radiological findings (including PET, SPECT, and MRS) and surgical strategy. J Neurooncol 2000;47:167-174.
15. Yu AH, Chen L, Li YJ, Zhang GJ, Li KC, Wang YP. Dysembryoplastic neuroepithelial tumors: magnetic resonance imaging and magnetic resonance spectroscopy evaluation. Chin Med J (Engl) 2009;20;122:2433-2437.
16. Ostertun B, Wolf HK, Campos MG, et al. Dysembryoplastic neuroepithelial tumors: MR and CT evaluation. AJNR Am J Neuroradiol 1996;17:419-430.
17. Fernandez C, Girard N, Paz Paredes A, Bouvier-Labit C, Lena G, Figarella-Branger D. The usefulness of MR imaging in the diagnosis of dysembryoplastic neuroepithelial tumor in children: a study of 14 cases. AJNR Am J Neuroradiol 2003;24:829-834.
18. Pasquier B, Peoc HM, Fabre-Bocquentin B, et al. Surgical pathology of drug-resistant partial epilepsy. A 10-year-experience with a series of 327 consecutive resections. Epileptic Disord 2002;4:99-119.
19. Campos AR, Clusmann H, von Lehe M, et al. Simple and complex dysembryoplastic neuroepithelial tumors (DNT) variants: clinical profile, MRI, and histopathology. Neuroradiology 2009;51:433-443.
20. Chan CH, Bittar RG, Davis GA, Kalnins RM, Fabinyi GC. Long-term seizure outcome following surgery for dysembryoplastic neuroepithelial tumor. J Neurosurg 2006;104:62-69.
21. Minkin K, Klein O, Mancini J, Lena G. Surgical strategies and seizure control in pediatric patients with dysembryoplastic neuroepithelial tumors: a single-institution experience. J Neurosurg Pediatrics 2008;1:206-210.
22. Spalice A, Ruggieri M, Grosso S, et al. Dysembryoplastic neuroepithelial tumors: a prospective clinicopathologic and outcome study of 13 children. Pediatr Neurol 2010;43:395-402.
23. Tian AG, Edwards MS, Williams NJ, Olson DM. Epilepsy surgery following brain tumor resection in children. J Neurosurg Pediatr 2011;7:229-234.
Received 16 September 2011
Received in final form 17 January 2012
Accepted 24 January 2012
Conflict of interest: There is no conflict of interest to declare.
- 1. Daumas-Duport C, Scheithauer BW, Chodkiewicz JP, Laws Jr ER, Vedrenne C. Dysembryoplastic neuroepithelial tumor: a surgically curable tumor of young patients with intractable partial seizures. Report of thirty-nine cases. Neurosurgery 1988:23:545-556.
- 2. Kleihues P, Burger PC, Scheithauer BW. The new WHO classification of brain tumors. Brain Pathol 1993:3:255-268.
- 3. Daumas-Duport C, Pietsch T, Hawkins C. Dysembryoplastic neuroepithelial tumor. In: Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (Eds). WHO classification of tumours of the central nervous system. 4th ed. Lyon, France: IARC Press; 2007: 99-102.
- 5. Giulioni M, Galassi E, Zucchelli M, Volpi L. Seizure outcome of lesionectomy in glioneuronal tumors associated with epilepsy in children. J Neurosurg 2005;102(Suppl 3):288-293.
- 6. Thom M, Toma A, An S, et al. One hundred and one dysembryoplastic neuroepithelial tumors: an adult epilepsy series with immunohistochemical, molecular genetic, and clinical correlations and a review of the literature. J Neuropathol Exp Neurol 2011;70:859-878.
- 7. Burger PC, Jouvet A, Preusser M, Hans VH, Rosenblum MK, Lellouch-Tubiana A. Angiocentric glioma. In: Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (Eds). WHO classification of tumours of the central nervous system. 4th ed. Lyon, France: IARC Press; 2007: 92-93.
- 8. Ruban D, Byrne RW, Kanner A, et al. Chronic epilepsy associated with temporal tumors: long-term surgical outcome. Neurosurg Focus 2009;27:e6.
- 9. Rickert CH, Paulus W. Epidemiology of central nervous system tumors in childhood and adolescence based on the new WHO classification. Childs Nerv Syst 2001;17:503-511.
- 10. Prayson RA. Tumours arising in the setting of paediatric chronic epilepsy. Pathology 2010;42:426-431.
- 11. Rosemberg S, Vieira GS. Tumor Neuroepithelial Disembrioplásico. Um estudo epidemiológico de uma única instituição. Arq Neuropsiquiatr 1998;56:232-236.
- 12. Nolan MA, Sakuta R, Chuang N, et al. Dysembryoplastic neuroepithelial tumors in childhood: long-term outcome and prognostic features. Neurology 2004;62:2270-2276.
- 13. Stanescu Cosson R, Varlet P, Beuvon F, et al. Dysembryoplastic neuroepithelial tumors: CT, MR findings and imaging follow-up: a study of 53 cases. J Neuroradiol 2001;28:230-240.
- 14. Lee DY, Chung CK, Hwang YS, et al. Dysembryoplastic neuroepithelial tumor: radiological findings (including PET, SPECT, and MRS) and surgical strategy. J Neurooncol 2000;47:167-174.
- 15. Yu AH, Chen L, Li YJ, Zhang GJ, Li KC, Wang YP. Dysembryoplastic neuroepithelial tumors: magnetic resonance imaging and magnetic resonance spectroscopy evaluation. Chin Med J (Engl) 2009;20;122:2433-2437.
- 16. Ostertun B, Wolf HK, Campos MG, et al. Dysembryoplastic neuroepithelial tumors: MR and CT evaluation. AJNR Am J Neuroradiol 1996;17:419-430.
- 17. Fernandez C, Girard N, Paz Paredes A, Bouvier-Labit C, Lena G, Figarella-Branger D. The usefulness of MR imaging in the diagnosis of dysembryoplastic neuroepithelial tumor in children: a study of 14 cases. AJNR Am J Neuroradiol 2003;24:829-834.
- 18. Pasquier B, Peoc HM, Fabre-Bocquentin B, et al. Surgical pathology of drug-resistant partial epilepsy. A 10-year-experience with a series of 327 consecutive resections. Epileptic Disord 2002;4:99-119.
- 19. Campos AR, Clusmann H, von Lehe M, et al. Simple and complex dysembryoplastic neuroepithelial tumors (DNT) variants: clinical profile, MRI, and histopathology. Neuroradiology 2009;51:433-443.
- 20. Chan CH, Bittar RG, Davis GA, Kalnins RM, Fabinyi GC. Long-term seizure outcome following surgery for dysembryoplastic neuroepithelial tumor. J Neurosurg 2006;104:62-69.
- 21. Minkin K, Klein O, Mancini J, Lena G. Surgical strategies and seizure control in pediatric patients with dysembryoplastic neuroepithelial tumors: a single-institution experience. J Neurosurg Pediatrics 2008;1:206-210.
- 22. Spalice A, Ruggieri M, Grosso S, et al. Dysembryoplastic neuroepithelial tumors: a prospective clinicopathologic and outcome study of 13 children. Pediatr Neurol 2010;43:395-402.
- 23. Tian AG, Edwards MS, Williams NJ, Olson DM. Epilepsy surgery following brain tumor resection in children. J Neurosurg Pediatr 2011;7:229-234.
Correspondence:
Publication Dates
-
Publication in this collection
13 Sept 2012 -
Date of issue
Sept 2012
History
-
Received
16 Sept 2011 -
Accepted
24 Jan 2012 -
Reviewed
17 Jan 2012