Abstracts
We compared the biochemical characteristics of the beans of a naturally decaffeinated Arabica coffee (AC1) discovered in 2004 with those of the widely grown Brazilian Arabica cultivar "Mundo Novo" (MN). Although we observed differences during fruit development, the contents of amino acids, organic acids, chlorogenic acids, soluble sugars and trigonelline were similar in the ripe fruits of AC1 and MN. AC1 beans accumulated theobromine, and caffeine was almost entirely absent. Tests on the supply of [2-14C] adenine and enzymatic analysis of theobromine synthase and caffeine synthase in the endosperm of AC1 confirmed that, as in the leaves, caffeine synthesis is blocked during the methylation of theobromine to caffeine. The quality of the final coffee beverage obtained from AC1 was similar to that of MN.
Coffea arabica; caffeine; decaffeination; beverage quality
Foram comparadas as características bioquímicas das sementes de um cafeeiro Arabica naturalmente descafeinado (AC1), descoberto em 2004, com aquelas da cultivar Mundo Novo (MN), amplamente cultivada no Brasil. Apesar de terem sido observadas diferenças durante o desenvolvimento das sementes, os conteúdos de aminoácidos, ácidos orgânicos, ácidos clorogênicos, açúcares solúveis e trigonelina foram similares nas sementes de frutos maduros de AC1 e MN. Sementes de AC1 acumularam teobromina, e a cafeína estava praticamente ausente. Experimentos com o fornecimento de [2-14C] adenina e análises enzimáticas de teobromina sintase e cafeína sintase nas sementes de AC1 confirmaram que, assim como em folhas, a síntese de cafeína é bloqueada na metilação de teobromina a cafeína. A qualidade final da bebida de AC1 foi similar a de MN.
Coffea arabica; cafeína; descafeinado; qualidade de bebida
BASIC AREAS
ARTICLE
Characterisation of AC1: a naturally decaffeinated coffee
Caracterização de AC1: um café naturalmente descafeinado
Luciana Benjamim BenattiI; Maria Bernadete SilvarollaII; Paulo MazzaferaI,* (* ) Corresponding author: pmazza@unicamp.br
IUNICAMP, Instituto de Biologia, Departamento de Biologia Vegetal, Caixa Postal 6109, 13083-970 Campinas (SP), Brasil
IIInstituto Agronômico (IAC), Centro Análise e Pesquisa Tecnológica do Agronegócio do Café "Alcides Carvalho", Av. Barão de Itapura, 1481, 13012-970 Campinas (SP), Brasil
ABSTRACT
We compared the biochemical characteristics of the beans of a naturally decaffeinated Arabica coffee (AC1) discovered in 2004 with those of the widely grown Brazilian Arabica cultivar "Mundo Novo" (MN). Although we observed differences during fruit development, the contents of amino acids, organic acids, chlorogenic acids, soluble sugars and trigonelline were similar in the ripe fruits of AC1 and MN. AC1 beans accumulated theobromine, and caffeine was almost entirely absent. Tests on the supply of [2-14C] adenine and enzymatic analysis of theobromine synthase and caffeine synthase in the endosperm of AC1 confirmed that, as in the leaves, caffeine synthesis is blocked during the methylation of theobromine to caffeine. The quality of the final coffee beverage obtained from AC1 was similar to that of MN.
Key words:Coffea arabica, caffeine, decaffeination, beverage quality.
RESUMO
Foram comparadas as características bioquímicas das sementes de um cafeeiro Arabica naturalmente descafeinado (AC1), descoberto em 2004, com aquelas da cultivar Mundo Novo (MN), amplamente cultivada no Brasil. Apesar de terem sido observadas diferenças durante o desenvolvimento das sementes, os conteúdos de aminoácidos, ácidos orgânicos, ácidos clorogênicos, açúcares solúveis e trigonelina foram similares nas sementes de frutos maduros de AC1 e MN. Sementes de AC1 acumularam teobromina, e a cafeína estava praticamente ausente. Experimentos com o fornecimento de [2-14C] adenina e análises enzimáticas de teobromina sintase e cafeína sintase nas sementes de AC1 confirmaram que, assim como em folhas, a síntese de cafeína é bloqueada na metilação de teobromina a cafeína. A qualidade final da bebida de AC1 foi similar a de MN.
Palavras-chave:Coffea arabica, cafeína, descafeinado, qualidade de bebida.
1. INTRODUCTION
Coffea arabica was originated in Ethiopia, and the product made from the infusion of its roasted beans spread worldwide as a refreshing drink due mostly to the alkaloid caffeine. Two Coffea species dominate the world market, C. arabica, which is known as Arabica coffee and represents approximately 70-75% of the world market, and C. canephora, or Robusta, representing nearly 25-30% of the market (Dias et al., 2007; see also Statistics of Coffee Trade at http://www.ico.org/trade_statistics.asp).
For people sensitive to caffeine, drinking coffee may cause some unwanted effects, including palpitations, gastrointestinal disturbances, anxiety, tremors, increased blood pressure and insomnia (Crozier et al., 2012). Due in large part to these symptoms, the decaffeinated coffee market has grown significantly since the establishment of the first decaffeination patents (Mazzafera et al., 2009). Currently, the only commercially available decaffeinated beans are those that have been artificially treated using chemical processes. The drawback of chemical decaffeination methods is that, along with the removal of caffeine, there are also losses of or changes to important chemical compounds that contribute to the flavour and aroma of the beverage (Farah et al., 2006a,b; Toci et al., 2006; Abrahão et al., 2008). In an attempt to meet the demands of customers sensitive to caffeine while maintaining the original quality of a coffee product, studies have focused on the selection and breeding of coffee trees that produce beans with a low caffeine content, which would therefore not require chemical processing methods (Mazzafera et al., 2009).
In 1987, the Agronomic Institute of Campinas established a breeding program to reduce the caffeine content in Arabica coffee beans (Mazzafera and Carvalho, 1992; Mazzafera et al., 1997; Silvarolla et al., 1999; 2000). As part of this program, analysis was performed on the alkaloids from C. arabica accessions from Ethiopia and maintained in the germplasm collection of the IAC (Silvarolla et al., 2000; 2004). Among more than 3,000 trees representing 300 accessions, three plants were discovered whose beans had very low concentrations of caffeine, and these plants were named AC1, AC2 and AC3 (Silvarolla et al., 2004). The AC1 plant was the most suitable candidate for the genetic transferral of the "no caffeine" trait to cultivars with high productivity (M.B. Silvarolla, unpublished data). The measured caffeine content of AC1 beans was 0.76 mg g-1. Theobromine, the immediate precursor of caffeine but which is also involved in caffeine catabolism, was found to accumulate in the leaves of this plant (Silvarolla et al., 2004; Mazzafera et al., 2009). When AC1 leaves were incubated with [2-14C] caffeine or [2-14C] adenine (the latter also a precursor of caffeine), caffeine was degraded normally, but the biosynthesis of caffeine (1,3,7-trimethylxanthine) from the methylation of theobromine (3,7-dimethylxanthine) was blocked. The activity of the methyltransferase responsible for this conversion, caffeine synthase, was found to be reduced in AC1 leaves. Unfortunately, the three naturally decaffeinated plants had low productivity, a typical characteristic of wild plants that hinders large-scale planting and commercialisation.
The AC1 plant has been used in breeding programs to transfer the "no caffeine" trait to commercial cultivars of C. arabica, which have high productivity. However, the available biochemical characterisations of AC1 are preliminary because they were limited to the biosynthesis of caffeine in the leaves (Silvarolla et al., 2004). More recently, the caffeine synthase gene was analysed in AC1 fruits (Maluf et al., 2009). This analysis revealed some complexities of the regulation of caffeine biosynthesis, suggesting that this pathway is subject to the transcriptional control of caffeine synthase. Thus, the present study aimed to characterise the biochemistry of AC1 beans, a knowledge that is missing for this naturally decaffeinated plant.
2. MATERIAL AND METHODS
Plant material and sampling
We compared the AC1 (Silvarolla et al., 2004) with the "Mundo Novo" cultivar of Coffea Arabica (MN), which is commercially cultivated in Brazil and has a caffeine content between 1% and 1.2% in the beans. Both cultivars were grown in the same experimental plot at the Fazenda Santa Elisa of the Agronomic Institute of Campinas, located in Campinas, São Paulo, Brazil. The samples used in the assays were collected during 2008-2009.
For the analysis of methylxanthines in different organs of the AC1 and MN plants, we collected root fragments, the first three leaf pairs (the first up to 1 cm, the second up to 5 cm and the third up to 8 cm) and internode samples (the first, third and fifth internodes). The roots were collected by digging around the plants and harvesting the secondary and tertiary roots at a depth of 30 cm. Prior to their analysis, the roots were washed with running water.
Flowers and fruits were also collected for analyses. Fruits were collected at eight different phenological stages, from fruit at a very young stage ("pinhead" stage) to fruit that were fully developed and mature ("cherry" stage). At each stage, the fruits were cut in half; the pericarp, perisperm and endosperm were separated using a scalpel; and the fresh and dried weights of the respective tissues were determined. During this procedure, the tissues were kept on ice and further lyophilised in liquid nitrogen. The dry weight was determined using the lyophilised material. The contents of methylxanthine alkaloids, trigonelline, soluble sugars, amino acids, organic acids, chlorogenic acids and free phenols were determined from extracts obtained from the endosperm. The qualitative analysis of amino acid content was only performed for the last fruit samples collected.
Green fruits (with a liquid endosperm occupying the entire locule) were collected from AC1 and MN plants for activity analysis of theobromine synthase (the methylation of 7-methylxanthine to 3,7-dimethylxanthine, E.C. 2.1.1.159) and caffeine synthase (the methylation of 3,7-dimethylxanthine to 1,3,7-trimethylxanthine, E.C.2.1.1.160). Immediately after fruit collection, the endosperms were separated and placed in liquid nitrogen and maintained at -80 ºC until further analysis. Green fruits at the same stage of development were used in the radioactive tracer experiments with [2-14C] adenine.
Radiochemicals
[3H] S-adenosyl-methionine (specific activity=14.9 Ci mmol-1) was obtained from Perkin-Elmer, Inc., USA, and [2-14C] adenine (283 mCi mmol-1) and [2-14C] caffeine (51.2 mCi mmol-1) were obtained from GE Healthcare, UK.
Analysis
The lyophilised tissues were macerated with a mortar and pestle, and the extractions were performed using 100 mg of tissue in 5 mL of methanol (70%). The extraction mixture was maintained in a water bath at 50 ºC for 1 h with occasional stirring. After centrifugation, the extracts were recovered and stored at -40 ºC. These extracts were used for all of the biochemical analyses, except for the analysis of organic acids, which utilised 100 mg of tissue in 5 mL of 4 mM H2SO4 containing 5 mM dithiothreitol. The samples were stirred on ice for 1 h, and the extracts were centrifuged at 14,000 rpm for 10 min and stored in a freezer at -40 ºC until they were used for HPLC analysis.
The analysis of caffeine, theobromine, trigonelline and chlorogenic acids was performed using a Shimadzu HPLC system operating with a diode detector and a C18 reverse phase column (4.6 mm x 250 mm, 5 µm particles, ACE). The mobile phases used were 0.5% acetic acid (A) and methanol (B), and the gradient used was as follows: 0-5 min at 0 to 5% B, 5-30 min at 50 to 70% B, 30-32 min at 70 to 100% B, 32-34 min at 100 to 0% B and 34-44 min maintained at 0% B. The flow was 0.8 mL min-1. Caffeine, theobromine and trigonelline were detected at 272 nm, and chlorogenic acids were detected at 313 nm. The injection volume was 10 µL of sample extract. The concentrations were calculated using calibration curves obtained from pure standards (Sigma). Chlorogenic acids were identified from absorption spectra obtained from the diode detector between 190 nm and 350 nm, and the concentration was calculated relative to a calibration curve obtained from the 5-caffeoylquinic acid isomer (Sigma), which is the most abundant chlorogenic acid isomer in coffee (Farah and Donangelo, 2006).
Glucose, fructose and sucrose were analysed using the Shimadzu HPLC system (with peak tubing) and operating with electrochemical detector at 400 mV, a Dionex CarboPac PA1 (4 mm x 50 mm) pre-column and a Dionex CarboPac PA1 column (4 mm x 250 mm). The mobile phase was 40 mM NaOH and a 15 min run was used for each sample. The flow rate was 1.2 mL min-1. Sugar concentrations were calculated using calibration curves derived from pure standards (Sigma).
The qualitative analysis of amino acids was performed using the Shimadzu HPLC system equipped with a manual Rheodyne injector and a fluorescence detector operating at 250 nm (excitation) and 480 nm (emission). The amino acids were separated in a C18 reverse phase column (4 mm x 250 mm, 5 µm, Supelco LC-18). The mobile phases used were as follows: (A) 50 mM sodium acetate, 50 mM Na2HPO4, pH 7.25 adjusted with HCl, containing 0.2% tetrahydrofuran and 0.2% methanol and (B) a mixture containing 65% methanol and 35% water (Jarret et al., 1986). The following gradient was used: 0-21 min at 25 to 46% B, 21-26 min at 46 to 48% B, 26-35 min at 48 to 60% B, 35-45 min at 60 to 70% B, 45-49 min at 70 to 100% B, 49-64 min maintained at 100% B and 64-65 min at 100 to 25% B. The flow rate was 0.8 mL min-1. Derivatization of the samples was carried out with o-phthalaldehyde (Jarret et al., 1986). The amino acid concentrations were calculated using a mixture of 18 amino acids (AAS-18, Sigma) plus glutamine and asparagine (Sigma) as the reference standard.
The analysis of organic acids was performed using the Shimadzu HPLC system equipped with a Rheodyne injector and a diode detector operating at 210 nm. The substances were separated in an Aminex HPX-87H column (Bio-Rad). Fifteen microlitres of sample was applied at a flow rate of 0.6 mL min-1. The mobile phase was 4 mM H2SO4, and the isocratic run lasted 30 min. Concentrations were calculated using pure reference standards of oxalic, malic, citric and succinic acids (Sigma).
From the ethanolic extracts, the total free amino acid content of these extracts was determined using the ninhydrin reagent (Cocking and Yemm, 1954).
Activity of caffeine synthase and theobromine synthase
The endosperms stored at - 80 ºC were macerated in liquid nitrogen using a mortar and pestle, and 1 g of tissue was combined with 5 mL of 200 mM Na2HPO4 buffer, 5 mM EDTA, 10 mM 2ß-mercaptoethanol, 1.5% ascorbic acid and 4% polyvinylpolypyrrolidone (Kato et al., 1996). After homogenisation, the extract was maintained on ice for 15 min with occasional agitation and then centrifuged for 15 min at 30,000 g (4 ºC). The supernatant was recovered, saturated to 80% with (NH4)2SO4 and then centrifuged for 15 min at 30,000 g (4 ºC). The recovered precipitate was dissolved in 200 mM Na2HPO4 buffer and desalted in a PD10-Sephadex G25 column (Amersham), using the 50 mM Na2HPO4 buffer for protein elution. The protein concentrations in the desalted extracts were determined using a "ready-to-use" reagent from Bio-Rad (Bradford, 1976). The substrates used were 7-methylxanthine for the determination of theobromine synthase activity and paraxanthine for caffeine synthase activity. In a 1.5 mL Eppendorf tube, the following were combined: 0.11 µCi of [3H]-S-adenosyl methionine, 100 µg of protein and 10 µL of substrate at 3.5 mM. The final volume was adjusted to 200 µL with 50 mM Na2HPO4 buffer. The reaction was incubated at 28 ºC for 30 min and stopped with 100 µL of 6 N HCl. One millilitre of chloroform was added to the reactions containing the paraxanthine substrate, and 1 mL of a chloroform:isopropyl alcohol (17:3, v/v) mixture was added to the reactions containing the 7-methylxanthine substrate (Mazzafera et al., 1994b). The Eppendorf tubes were vortexed for 30 s, and the organic fraction was collected and transferred to scintillation vials, and then dried using flowing air in a fume hood. Scintillation liquid (5 mL) was added to the dried contents, and the amount of radioactivity incorporated into the theobromine or caffeine was determined in a scintillation counter for the 14C isotope for 2 min.
Metabolism of [2-14C] adenine and[2-14C] caffeine
The addition of [2-14C] adenine and [2-14C] caffeine was performed in fruits as previously described (Mazzafera et al., 1994a). Briefly, 0.2 µCi of [2-14C] adenine or 0.2 µCi of [2-14C] caffeine was applied onto a small incision made at the fruit peduncle. The fruits were placed in a plastic box, with the stalk positioned upward. Once the [2-14C] adenine or [2-14C] caffeine solutions were absorbed, 5 x 10 µL of 100 mM Na2HPO4 buffer, pH 6, was applied at the incision area. A cotton ball soaked in water was placed at the centre of the box to prevent the fruit from drying out. The box was covered and maintained under white fluorescent light (150 µmol photons m-2 s-1). The fruits were removed after 24 h ([2-14C] adenine) or 48 h ([2-14C] caffeine), and the endosperms were separated with a scalpel and lyophilised. For the extraction procedure, the lyophilised material was ground in a mortar, transferred to a screw cap tube with 70% methanol (20 mg mL-1) and incubated in a 50 ºC water bath for 1 h. The extracts were centrifuged for 10 min at 12,000 g, and 500 µL was dried in a Speed-Vac (Savant). The dried samples were dissolved in 30 µL of water, constantly stirred for 1 h and subjected to thin layer chromatography on silica sheets GF254 (Merck). A 10 µL volume of an aqueous solution containing 5 µg of caffeine, 5 µg of theobromine and 5 µg of theophylline was applied on to the sample spots. The chromatography was developed with a chloroform:methanol mixture (9:1, v/v), and after drying, the spots were visualised under UV light (254 nm) and circled with a pencil. The reference values (Rf values) for caffeine, theobromine and theophylline were 0.51, 0.41 and 0.37, respectively. Spots visualized under UV light were scraped from the plate with a spatula and transferred to scintillation tubes, and 1 mL of methanol and 5 mL of scintillation liquid were added to each tube. Radioactivity was determined with a scintillation counter for the 14C isotope for 2 min.
Statistical analysis:
Three replicates were performed for all biochemical measurements. The activity of theobromine synthase and caffeine synthase was estimated from five replicates. Analysis of variance and post-hoc comparison of means (Tukey's test, p<0.05) were performed using the statistical analysis program SISVAR (Ferreira, 2000). For the analysis of the metabolism of [2-14C] adenine and [2-14C] caffeine, extracts from five replicates were used.
3. RESULTS
Fruit development
Although some differences were observed, the fruits of AC1 and MN showed similar patterns of development (Figure 1). However, the mass of AC1 fruit was always lower than those of MN, and the smaller size of these fruits was also observed visually.
Methylxanthines in different plant organs
An analysis of the caffeine content during AC1 and MN endosperm development showed that AC1 had a higher total (caffeine + theobromine) alkaloid content (Figure 2). However, the AC1 theobromine levels were always similar to the caffeine levels found in MN. The final concentration of caffeine in MN was 8.5±0.17 mg g-1. AC1 theobromine contents varied between 6.75 and 13.42 mg g-1, with the largest amounts present in the immature endosperm. The theobromine content in the AC1 mature endosperm was 6.48±0.48 mg g-1, and the caffeine content in the mature AC1 endosperm was 0.40±0.02 mg g-1.
Methylxanthines were not detected in the roots of either plant. In MN flowers, the caffeine content was 1.06±0.11 mg g-1, and in AC1 flowers, the caffeine content was 1.04±0.03 mg g-1. In both MN and AC1, the methylxanthine levels decreased with leaf maturity (Figure 2). Caffeine and theobromine were detected in MN leaves, but in AC1 leaves, only theobromine was detected.
In MN, theobromine was detected only in the first internode; in contrast, this methylxanthine was present in the first, third and fifth internodes of AC1 plants. Caffeine was present in the three internodes analysed in MN, but it was not detected in any of the AC1 internodes. It was also observed that the caffeine and theobromine contents decreased sharply from the first to the third internodes in both plants.
Sugars
In the young endosperm, which did not fully occupy the locule and had a milky appearance, there were higher levels of reducing sugars (glucose and fructose) compared with sucrose (Figure 3a, b). As the fruits ripened, the sucrose content increased, and the reducing sugar levels decreased. In ripe fruits, the sucrose content was 37.07±5.06 mg g-1 and 66.2±7.4 mg g-1 for AC1 and MN, respectively.
Organic acids
Oxalic, citric, malic and succinic acids were observed in MN and AC1 endosperms at different stages of fruit development (Figure 3c, d). In MN, citric acid was present at higher levels than the other acids at all development stages; this was not the case in AC1. Nevertheless, citric acid was the organic acid with the highest concentration in mature AC1 endosperms. Quantitative differences in the contents of oxalic, succinic and malic acid were observed in MN, and these acids were present at similar levels in AC1.
Amino acids
MN had a greater total free amino acid content in the mature endosperm (34.98±3.28 nmol mg-1) compared with AC1 (25.2±1.47 nmol mg-1). The profile of amino acids obtained from the endosperms of mature fruits showed that asparagine, glutamate, aspartate and alanine were the most abundant amino acids in both MN and AC1 (Table 1). In this order, MN and AC1, asparagine was present at 28.69 mol% and 33.61 mol%; glutamate at 27.38 mol% and 22.45 mol%; alanine at 14.76 mol% and 7.28 mol%; and aspartate at 9.50 mol% and 12.90 mol%. Glycine and threonine were detected at levels less than 0.1% in AC1, which was almost 10 times less than the levels in MN.
Phenolic compounds
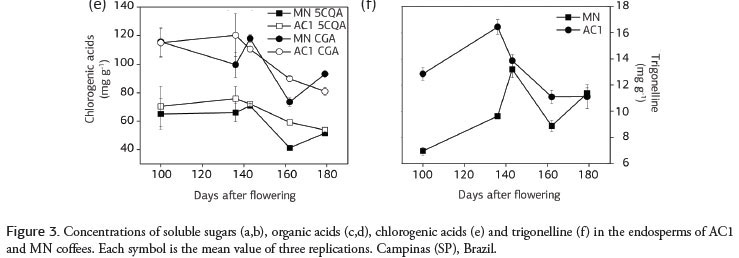
In the endosperms 5-caffeoylquinic acid (5CQA) was the predominant chlorogenic acid (CGA) in both MN and AC1 (Figure 3e), and the levels varied throughout fruit development. AC1 always had greater amounts of 5CQA, with the greatest difference observed approximately 162 days after flowering. When the beans were fully developed, the levels of 5CQA were similar between AC1 and MN. Seven other CGAs were detected based on the UV absorption spectra obtained in the HPLC detector diode, and the chromatographic profile of the CGAs (not shown) was very similar to that reported by Clifford et al. (2008). It was not possible to identify these acids due to a lack of standards, but based on quantifications made relative to 5CQA (Table 2), these other CGAs were present at higher levels in MN.
Trigonelline
The amount of trigonelline found in the endosperm of immature AC1 fruits was greater than that observed in MN (Figure 3f). However, this difference decreased as the fruits ripened. When the fruits were fully developed, the quantities found in the two plants were similar, approximately 11 mg g-1 in the endosperm.
Activity of theobromine and caffeine synthase
The activities of both theobromine synthase and caffeine synthase were higher in the endosperm of MN than in AC1 (Table 3). Theobromine synthase activity in AC1 was approximately half of that observed in MN, while the activity of caffeine synthase was 15 times lower in decaffeinated plants than in MN.
Metabolism of [14C] adenine and [2-14C] caffeine in fruits
Feeding [2-14C] adenine to AC1 fruits revealed that theobromine was the main methylxanthine labelled with radioactivity (Table 4); after 24 h of incubation, we detected 60 times more radioactivity in theobromine than in caffeine. Contrary to what was observed in the decaffeinated plants, radioactivity accumulated mainly in caffeine in the MN endosperm fed with [2-14C] adenine, although there was also a small amount of radioactivity present in theobromine. When [2-14C] caffeine was administered, radioactivity was detected in theophylline, which appears only in the catabolism of caffeine. Radioactivity was also detected in theobromine, which is both a precursor and a degradation product of caffeine.
4. DISCUSSION
Although mature AC1 fruits were smaller than mature MN fruits, the pattern of AC1 fruit development was similar to that of MN fruits. During coffee fruit development, the perisperm is substituted by the endosperm (Mendes, 1941; DeCastro and Marraccini, 2006), which pattern was similar in both MN and AC1. However, closer inspection of our data suggests that the AC1 fruits developed earlier than the MN fruits. We observed that the replacement of the perisperm by the endosperm occurred more rapidly in AC1. At 100 days after flowering (DAF), the fresh and dry weights of AC1 endosperms were considerably greater than those of MN endosperms. On the other hand, the ripe fruits of MN were larger, with greater values for both the fresh and dry weights. This difference may be considered a disadvantage for AC1 because fruit size is one of the desired physical attributes in quality coffees. However, necessarily this is not related with the beverage quality. For example, the varieties Mokka (Carvalho et al., 1991) and Laurina (Krug et al., 1954), which have small beans, produce coffee of excellent quality, and the small beans are accepted as intrinsic characteristics. Analysis of the beverage produced from the beans of AC1 indicated that the quality is good, having obtained the classification of "exotic" in the sensory characterisation (G.S. Giomo, IAC, personal communication). Because the size of the beans, AC1 demands special care for certain aspects of post-harvest processing, as well as roasting time and grinding when particle size and appearance of the final product is defined.
The analysis in the present study showed that no methylxanthines were detected in the roots of MN or AC1 what confirms findings previously reported for C. arabica seedlings (Zheng and Ashihara, 2004).
In both AC1 and MN, we observed a reduction in the leaf caffeine content with increased leaf age, which was previously observed in adult C. arabica plants (Hamidi and Wanner, 1964). We also found that younger internodes had greater amounts of methylxanthines in both MN and AC1, with a greater amount of caffeine in MN than in AC1. The endosperm of young MN fruits had higher caffeine content than the endosperm of ripe fruits; the same pattern was observed for theobromine in AC1 fruits. Both methylxanthines, caffeine and theobromine, exhibited the same distribution patterns when comparing young and old tissues or mature and immature endosperms in AC1 and MN. These patterns were previously observed for the caffeine content in leaves (Ashihara et al., 1996a,b), fruit (Koshiro et al., 2006) and branches (Zheng and Ashihara, 2004).
Interestingly, we observed that MN and AC1 flowers had similar quantities of caffeine. In C. Arabica flowers during the anthesis stage, caffeine is the most abundant purine alkaloid, with 0.58 mg g-1 in the petals and gynoecium and 1.36 mg g-1 in the stamens (Baumann, 2006).
The predominance of theobromine over caffeine was detected in all of the AC1 tissues analysed. Additionally, AC1 leaves incubated with [2-14C] adenine accumulated radioactivity in theobromine, not caffeine, similar to previous observations in leaves (Silvarolla et al., 2004). These findings provide evidence that the blockade of caffeine biosynthesis occurs at the step when theobromine is methylated to form caffeine.
AC1 beans have significantly lower amounts of caffeine than any wild or cultivated C. arabica tissues investigated thus far (Mazzafera et al., 2009). New hybrids developed in Madagascar, from crosses between C. arabica, C. canephora and C. eugenioides, had 0.37% caffeine and undetectable levels of theobromine; however, insufficient data regarding production were presented to support the commercial viability of these hybrids (Nagai et al., 2008).
Quantitative PCR analyses showed that the expression of genes coding for theobromine synthase (CTS2) and caffeine synthase (CCS1) were significantly reduced in the decaffeinated cultivar AC1 in comparison with a caffeinated coffee variety (Maluf et al., 2009). These results are not consistent with our results for the enzymatic activities, where caffeine synthase activity was almost absent and there was a partial reduction of theobromine synthase activity. A possible explanation for the reduced enzymatic conversion of 7-methylxanthine to theobromine is that caffeine synthase is a bi-functional enzyme, also mediating the biosynthesis of theobromine from 7-methylxanthine (Mizuno et al., 2003). Then, the reduction of theobromine synthase activity was in part because the lack of expression of caffeine synthase gene. The gene expression results are not supported also by the results of [2-14C] adenine feeding assays carried out with fruits (this work) and with leaves of AC1 (Silvarolla et al., 2004), which showed that the blockade in the caffeine synthesis occurs between theobromine and caffeine. The significant reduction of theobromine synthase expression in AC1 (Maluf et al., 2009) might be explained by the fact that the three methyltransferases of caffeine biosynthesis pathway in coffee share high sequence similarity (Mizuno et al., 2003; Kato and Mizuno, 2004; Yoneyama et al., 2006; Ashihara et al., 2008), what may have resulted in a lack of specificity in the primers used in the expression analysis in AC1 (Maluf et al., 2009).
The pattern of accumulation of soluble sugars in AC1 and MN was similar to that observed in C. arabica cv. Caturra (Rogers et al., 1999). We observed high glucose and fructose contents in the endosperm of young fruits, which decreased with ripening, while the reverse was observed for the sucrose content. Although the accumulation patterns were the same in AC1 and MN, quantitative differences were observed primarily for the sucrose content. In MN, the sucrose content in the mature endosperm reached 61 mg g-1, while in AC1, the levels were much lower (37 mg g-1). Although sucrose is an important compound that affects the quality of the final beverage product (Geromel et al., 2006; 2008), quantitative differences in the levels of this sugar are frequently observed between different C. arabica cultivars. Previous studies have shown that the sucrose content varies from 46 to 150 mg g-1 in mature beans (Clifford, 1985a; Rogers et al., 1999; Ky et al., 2001; Campa et al., 2004; Farah et al., 2006a; Murkovic and Derler, 2006). In addition to the intrinsic characteristics of each plant and/or cultivar, one possible explanation for the variation in sucrose levels in these studies is the exact stage of ripeness when the beans were collected. Although the red colour of the fruit may be indicative of maturation, varying intensities of red colouration may reflect biochemical differences, such as those observed in fruits collected from shaded plants (Geromel et al., 2008).
The main criticism of decaffeination methods is the removal of other substances that are important for the development of the product and the quality of the final beverage. Toci et al. (2006) reported that whole C. arabica beans had a sucrose concentration of 96.5 mg g-1 and that the concentration dropped to 38.5 mg g-1 after decaffeination with dichloromethane. Thus, the concentration of sucrose found in AC1 coffee beans requires further investigation. Although the sucrose concentration observed in the present study is within the concentration range found in different reports on coffee beans, it will be important to evaluate, over a period of several years, the sucrose content in AC1 plants grown in different coffee regions and subject to different environmental and cultivation influences.
The alkaloid trigonelline, which is largely degraded during the process of roasting coffee, gives rise to compounds that contribute to the aroma and flavour of the coffee beverage (Carvalho, 1962; Ashihara, 2006). Our results showed that in both studied genotypes, green fruits that had already developed the endosperm showed an accumulation of trigonelline similar to the levels found in mature beans. The levels of trigonelline observed in AC1 and MN are similar to the levels previously reported for different C. arabica cultivars (Ky et al., 2001; Campa et al., 2004; Farah et al., 2006a; Koshiro et al., 2006).
In both AC1 and MN plants, citric acid was the organic acid present at the highest concentration in the endosperm of ripe fruit, with the highest content observed in MN. Although the concentration of citric acid in MN remained relatively constant during development, the concentration of citric acid increased in maturing AC1 plants. The same pattern of increased citric acid content was also observed in two varieties of C. arabica (Caturra commercial and Caturra 2308); at full maturity, the citric acid content in the beans of these varieties was approximately 15 mg g-1 (Rogers et al., 1999), a value similar to that observed in AC1. Alcázar et al. (Alcázar et al., 2003) also reported that citric acid was the most abundant organic acid in a variety of C. arabica varieties. In the present study, the accumulation of malic acid was also found to vary during the development of the endosperm in MN and AC1. In the Caturra varieties studied by Rogers et al. (1999), malic acid was found to be the second most abundant organic acid in beans, with a content between 4 and 5 mg g-1. Similarly, Alcázar et al. (2003) reported a malic acid content of 4.14 mg/g. In the present study, the malic acid content in AC1 was similar to these previously reported values, and malic acid was the second most abundant acid in this tissue. In MN, we observed lower malic acid levels than the levels observed in other varieties of C. arabica. The endosperm of the Caturra variety exhibited intermediate oxalate levels compared to the levels observed in AC1 and MN (Rogers et al., 1999).
In both MN and in AC1, the three most abundant amino acids were asparagine, aspartate and glutamate, a finding that is consistent with previous work (Clifford, 1985a). Despite this qualitative similarity, MN had significantly greater total free amino acid content than AC1. These three amino acids have also been observed at high concentrations in other C. arabica plants. In unroasted beans of Brazilian C. arabica cv. Typica, the most abundant amino acids were glutamate, aspartate and GABA (Casal et al., 2005). The observed differences in the levels of amino acids may be due to post-harvest processing (Arnold and Ludwig, 1996; DeCastro and Marraccini, 2006).
Although the levels of 5CQA were similar in the tissues studied, MN had a higher total amount of CGAs than AC1. Clifford (Clifford, 1985b) listed the contents of CGAs in Arabica coffee reported by several authors, which varied between 40.7 and 84.0 mg g-1, and concluded that in part the variation was a consequence of the analytical method used. Samples of C. arabica from different sources had 5CQA as the major isomer, with the concentrations ranging from 3.44 to 56.1 mg g-1 and with an average concentration of 47.9 mg g-1. The total concentration of several other CGA isomers was 65.7 mg g-1, with values ranging from 55.2 to 75.5 mg g-1. Therefore, CGA concentrations differ greatly among different coffee varieties, and the results available in the literature depend on the analysis method used (Clifford, 1985b). In the present study, however, the differences between MN and AC1 were not quantitatively large, and the same CGAs were detected in both genotypes.
Assessments of CGA content during C. arabica fruit development indicated that the overall content increases with maturity, with a peak occurring four weeks before full maturity (Clifford, 1985b). Our analysis did not match this level of detail, but approximately 30 days before harvest, CGA levels were close to the maximum and decreased thereafter.
The maintenance of similar levels of CGAs in AC1 and MN is important because these compounds contribute to the quality of the final beverage (Farah and Donangelo, 2006). Larger quantities of caffeoylquinic and feruloylquinic acid isomers are associated with reduced coffee quality, while larger quantities of dicaffeoylquinic acid are related to improved beverage quality (Farah et al., 2006b). The artificial decaffeination processes, including water decaffeination (Farah et al., 2006b) and dichloromethane decaffeination (Farah et al., 2006b; Toci et al., 2006), affect the amount and proportion of CGAs in Arabica coffee. In this regard, the AC1 cultivar has the advantage of maintaining these compounds at levels similar to those found in commonly consumed and appreciated coffees.
5. CONCLUSION
In light of consumer interest in a caffeine-free product that maintains the characteristics of a good quality coffee, AC1 has great potential to satisfy this consumer demand. Despite some differences described in the present study, the beans of AC1 and MN have a similar chemical composition, and the latter is a commercially exploited variety that is widely consumed as a good quality coffee. Some of the observed differences may result not only from the genetic backgrounds of the plants but also from environmental and cultivation factors, as previously demonstrated by reports on the chemical composition of coffee beans (Clifford, 1985a). Another advantage of AC1 coffee pertains to the fact that decaffeination with dichloromethane can leave solvent residues in decaffeinated products (Cohen et al., 1980; Page and Charbonneau, 1984). Although the levels of these solvent residues are not considered harmful (Mazzafera and Carvalho, 1991), the consumer may prefer not to consume such a product.
ACKNOWLEDGMENTS
This work was supported by Financiadora de Estudos e Projetos (FINEP-Brazil), Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) and Consórcio Brasileiro de Pesquisa e Desenvolvimento do Café. L.B.B. is supported by a doctoral fellowship from CAPES, and P.M. is supported by a research fellowship from CNPq.
Received: Mar. 3, 2012
Accepted: May 23, 2012
- ABRAHÃO, S.A.; PEREIRA, R.G.F.A.; LIMA, A.R.; FERREIRA, E.B.; MALTA, M. Compostos bioativos em café integral e descafeinado e qualidade sensorial da bebida. Pesquisa Agropecuária Brasileira, v.43, p.1799-1804, 2008.
- ALCÁZAR, A.; FERNÁNDEZ-CÁCERES, P.L.; MATÍN, M.J.; PABLOS, F.; GONZÁLEZ, A.G. Ion chromatographic determination of some organic acids, chloride and phosphate in coffee and tea. Talanta, v.61, p.95-101, 2003.
- ARNOLD, U.; LUDWIG, E. Analysis of free amino acids in green coffee beans II. changes of the amino acid content in arabica coffees in connection with post-harvest model treatment. Zeitschrift für Lebensmitteluntersuchung und - Forschung A, v.203, p.379-384, 1996.
- ASHIHARA, H. Metabolism of alkaloids in coffee plants. Brazilian Journal of Plant Physiology, v.18, p.1-8, 2006
- ASHIHARA, H.; MONTEIRO, A.M.; GILLIES, F.M.; CROZIER, A. Biosynthesis of caffeine in leaves of coffee. Plant Physiology, v.111, p.747-753, 1996a.
- ASHIHARA, H.; MONTEIRO, A.M.; MORITZ, T.; GILLIES, F.M.; CROZIER, A. Catabolism of caffeine and related purine alkaloids in leaves of Coffea arabica L. Planta, v.198, p.334-339, 1996b.
- ASHIHARA, H.; SANO, H.; CROZIER, A. Caffeine and related purine alkaloids: Biosynthesis, catabolism, function and genetic engineering. Phytochemistry, v.69, p.833-1076, 2008.
- BAUMANN, T.W. Some thoughts on the physiology of caffeine in coffee - and a glimpse of metabolic profiling. Brazilian Journal of Plant Physiology, v,18, p.243-251, 2006.
- BRADFORD, M.M. Rapid and sensitive method for quantitation of microgram quantities of protein utilizing principle of protein-dye binding. Analytical Biochemistry, v.72, p.248-254, 1976.
- CAMPA, C.; BALLESTER, J.F.; DOULBEAU, S.; DUSSERT, S.; HAMON, S.; NOIROT, M. Trigonelline and sucrose diversity in wild Coffea species. Food Chemistry, v.88, p.39-43, 2004.
- CARVALHO, A. Variability of the niacin content in coffee. Nature, v.194, p.1096, 1962.
- CARVALHO, A.; MEDINA, H.P.; FILHO, FAZUOLI, L.C.; GUERREIRO FILHO, O.; LIMA, M.M.A. Aspectos genéticos do cafeeiro. Revista Brasileira de Genética, v.14, p.135-183, 1991.
- CASAL, S.; MENDES, E.; OLIVEIRA, M.B.P.P.; FERREIRA, M.A. Roast effects on coffee amino acid enantiomers. Food Chemistry, v.89, p.333-340, 2005.
- CLIFFORD, M.N. Chemical and physical aspects of green coffee and coffee products. In: CLIFFORD, M.N.; WILSON, K.C. (Ed.). Coffee: Botany, Biochemistry and Production of Beans and Beverage. Westport, Connecticut: AVI Publishing, 1985a. p.305-374.
- CLIFFORD, M.N. Chlorogenic acids. In: CLARKE, R.J.; MACRAE, R. (Ed.). Coffee Chemistry. London, New York: Elsevier Applied Science, 1985b. Vol 1, p. 153-202.
- CLIFFORD, M.N.; KIRKPATRICK, J.; KUHNERT, N.; ROOZENDAAL, H.; SALGADO, P.R. LC-MSn analysis of the cis isomers of chlorogenic acids. Food Chemistry, v.106, p.379-385, 2008.
- COCKING, E.C.; YEMM, E.W. Estimation of amino acids by ninhydrin. Analyst, v.80, p.209-213, 1954.
- COHEN, J.M.; DAWSON, R.; KOKETSU, M. Technical report: extent-of-exposure survey of methylene chloride. Washington, DC: U.S.D.o.H.a.H. Services, 1980. (DHHS-NIOSH, Publ. No. 80-131)
- CROZIER, T.W.M.; STALMACH, A.; LEAN, M.E.J.; CROZIER, A. Espresso coffees, caffeine and chlorogenic acid intake: potential health implications. Food & Function, v.3, p.30-33, 2012.
- DECASTRO, R.D.; MARRACCINI, P. Cytology, biochemistry and molecular changes during coffee fruit development. Brazilian Journal of Plant Physiology, v.18, p.175-199, 2006.
- DIAS, P.C.; ARAUJO, W.L.; MORAES, G.A.B.K.; BARROS, R.S.; DAMATTA, F.M. Morphological and physiological responses of two coffee progenies to soil water availability. Journal of Plant Physiology, v.164, p.1639-1647, 2007.
- FARAH, A.; DONANGELO, C.M. Phenolic compounds in coffee. Brazilian Journal of Plant Physiology, v.18, p.23-36, 2006.
- FARAH, A.; MONTEIRO, M.C.; CALADO, V.; FRANCA, A.S.; TRUGO, L.C. Correlation between cup quality and chemical attributes of Brazilian coffee. Food Chemistry, v.98, p.373-380, 2006a.
- FARAH, A.; PAULIS, T.D.; MOREIRA, D.P.; TRUGO, L.C.; MARTIN, P.R. Chlorogenic acids and lactones in regular and water-decaffeinated Arabica coffees. Journal of Agricultural and Food Chemistry, v.54, p.374-381, 2006b.
- FERREIRA, D.F. Sistema de análises de variância para dados balanceados. Lavras: UFLA, 2000. (SISVAR 4. 1. pacote computacional)
- GEROMEL, C.; FERREIRA, L.P.; BONATELLI, M.L.; BOTTCHER, A.; POT, D.; PEREIRA, L.F.P.; LEROY, T.; VIEIRA, L.G.E.; MAZZAFERA, P. Sucrose metabolism during fruit development of Coffea racemosa Annals of Applied Biology, v.152, p.179-187, 2008.
- GEROMEL, C.; FERREIRA, L.P.; CAVALARI, A.A.; PEREIRA, L.F.P.; GUERREIRO, S.M.C.; VIEIRA, L.G.E.; LEROY, T.; POT, D.; MAZZAFERA, P.; MARRACCINI, P. Biochemical and genomic analysis of sucrose metabolism during coffee (Coffea arabica) fruit development. Journal of Experimental Botany, v.57, p.3243-3258, 2006.
- GEROMEL, C.; FERREIRA, L.P.; DAVRIEUX, F.; GUYOT, B.; RIBEYRE, F.; BRÍGIDA DOS SANTOS SCHOLZ, M.; PROTASIO PEREIRA, L.F.; VAAST, P.; POT, D.; LEROY, T.; FILHO, A.A.; ESTEVES VIEIRA, L.G.; MAZZAFERA, P.; MARRACCINI, P. Effects of shade on the development and sugar metabolism of coffee (Coffea arabica L.) fruits. Plant Physiology and Biochemistry, v.46, p.569-579, 2008.
- HAMIDI, A.; WANNER, H. The distribution pattern of chlorogenic acid and caffeine in Coffea arabica Planta, v.61, p.90-96, 1964.
- JARRET, H.W.; COOSKY, K.D.; ELLIS, B.; ANDERSON, J.M. The separation of o-phtalaldehyde derivatives of amino acids by reversed-phase chromatography on octylsilica column. Analytical Biochemistry, v.153, p.189-198, 1986.
- KATO, M.; KANEHARA, T.; SHIMIZU, H.; SUZUKI, T.; GILLIES, F.M.; CROZIER, A.; ASHIHARA, H. Caffeine biosynthesis in young leaves of Cammelia sinensis: In vitro studies on N-methyltransferase activity involved in the conversion of xanthosine to caffeine. Physiologia Plantarum, v.98, p.629-636, 1996.
- KATO, M.; MIZUNO, K. Caffeine synthase and related methyltransferases in plants. Frontiers in Bioscience, v.9, p.1833-1842, 2004.
- KOSHIRO, Y.; ZHENG, X.-Q.; WANG, M.-L.; NAGAI, C.; ASHIHARA, H. Changes in content and biosynthetic activity of caffeine and trigonelline during growth and ripening of Coffea arabica and Coffea canephora fruits. Plant Science, v.171, p.242-250, 2006.
- KRUG, C.A.; CARVALHO, A.; ANTUNES FILHO, H. Genética de Coffea XXI. Hereditariedade dos característicos de Coffea arabica L. var. Laurina (Smeathman) DC. Bragantia, v.13, p.247-255, 1954.
- KY, C.L.; LOUARN, J.; DUSSERT, S.; GUYOT, B.; HAMON, S.; NOIROT, M. Caffeine, trigonelline, chlorogenic acid and sucrose diversity in wild Coffea arabica L. and C. canephora P. accessions. Food Chemistry, v.75, p.223-230, 2001.
- MALUF, M.P.; SILVA, C.C.; OLIVEIRA, M.D.A.; TAVARES, A.G.; SILVAROLLA, M.B.; GUERREIRO FILHO, O. Altered expression of the caffeine synthase gene in a naturally caffeine-free mutant of Coffea arabica Genetics and Molecular Biology, v.32, p.802-810, 2009.
- MAZZAFERA, P.; BAUMANN, T.W.; SHIMIZU, M.M.; SILVAROLLA, M.B. Decaf and the steeplechase towards Decaffito-the coffee from caffeine-free Arabica plants. Tropical Plant Biology, v.2, p.63-76, 2009.
- MAZZAFERA, P.; CARVALHO, A. A cafeína do café. Campinas: Instituto Agronômico, 1991. p.1-22. (Documentos IAC, 25)
- MAZZAFERA, P.; CARVALHO, A. Breeding for low seed caffeine content of coffee (Coffea L.) by interspecific hybridization. Euphytica, v.59, p.55-60, 1992.
- MAZZAFERA, P.; CROZIER, A.; SANDBERG, G. Studies on the metabolic control of caffeine turnover in developing endosperms and leaves of Coffea arabica and Coffea dewevrei Journal of Agricultural and Food Chemistry, v.42, p.1423-1427, 1994a.
- MAZZAFERA, P.; WINGSLE, G.; OLSSON, O.; SANDBERG, G. S-adenosyl-L-methionine:theobromine 1-N-methyltransferase, an enzyme catalyzing the synthesis of caffeine in coffee. Phytochemistry, v.37, p.1577-1584, 1994b.
- MAZZAFERA, P.; SILVAROLLA, M.B.; LIMA, M.M.A.; MEDINA FILHO, H.P. Caffeine content of diploid coffee species. Ciência e Cultura, v.49, p.216-218, 1997.
- MENDES, A.J.T. Cytological observations in Coffea VI. Embryo and endosperm development in Coffea arabica L. American Journal of Botany, v.28, p.784-789, 1941.
- MIZUNO, K.; OKUDA, A.; KATO, M.; YONEYAMA, N.; TANAKA, H.; ASHIHARA, H.; FUJIMURA, T. Isolation of a new dual-functional caffeine synthase gene encoding an enzyme for the conversion of 7-methylxanthine to caffeine from coffee (Coffea arabica L.). FEBS Letters, v.534, p.75-81, 2003.
- MURKOVIC, M.; DERLER, K. Analysis of amino acids and carbohydrates in green coffee. Journal of Biochemical and Biophysical Methods, v.69, p.25-32, 2006.
- NAGAI, C.; RAKOTOMALALA, J.J.; KATAHIRA, R.; LI, Y.; YAMAGATA, K.; ASHIHARA, H. Production of a new low-caffeine hybrid coffee and the biochemical mechanism of low caffeine accumulation. Euphytica, v.164, p.133-142, 2008.
- PAGE, B.D.; CHARBONNEAU, C.F. Headspace gas chromatographic determination of methylene chloride in decaffeinated tea and coffe, with electrolytic conductivity detection. Journal of the Association of Official Analytical Chemists, v.67, p.757-761, 1984.
- ROGERS, W.J.; MICHAUX, S.; BASTIN, M.; BUCHELI, P. Changes to the content of sugars, sugar alcohols, myo-inositol, carboxylic acids and inorganic anions in developing grains from different varieties of robusta (Coffea canephora) and arabica (C. arabica) coffees. Plant Science, v.149, p.115-123, 1999.
- SILVAROLLA, M.B.; MAZZAFERA, P.; FAZUOLI, L.C. A naturally decaffeinated arabica coffee. Nature, v.429, p.826-826, 2004.
- SILVAROLLA, M.B.; MAZZAFERA, P.; LIMA, M.M.A. Caffeine content of Ethiopian Coffea arabica beans. Genetics and Molecular Biology, v.23, p.213-215, 2000.
- SILVAROLLA, M.B.; MAZZAFERA, P.; LIMA, M.M.A.; MEDINA FILHO, H.P.; FAZUOLI, L.C. Ploidy level and caffeine content in leaves of Coffea Scientia Agricola, v.56, p.661-663, 1999.
- TOCI, A.; FARAH, A.; TRUGO, L.C. Efeito do processo de descafeinação com diclorometano sobre a composição química dos cafés arábica e robusta antes e após a torração. Química Nova, v.29, p.965-971, 2006.
- YONEYAMA, N.; MORIMOTO, H.; YE, C.-X.; ASHIHARA, H.; MIZUNO, K.; KATO, M. Substrate specificity of N-methyltransferase involved in purine alkaloids synthesis is dependent upon one amino acid residue of the enzyme. Molecular Genetics and Genomics, v.275, p.125-135, 2006.
- ZHENG, X.-Q.; ASHIHARA, H. Distribution, biosynthesis and function of purine and pyridine alkaloids in Coffea arabica seedlings. Plant Science, v.166, p.807-813, 2004.
Publication Dates
-
Publication in this collection
06 Sept 2012 -
Date of issue
2012
History
-
Received
03 Mar 2012 -
Accepted
23 May 2012