Abstracts
OBJECTIVE: To study the plasma lipid profile and lipid peroxidation in overweight or obese children and adolescents receiving care at the pediatric endocrinology clinic in HOSPED/UFRN, a university hospital. METHODS: Three groups were studied: overweight (n = 15), obese (n = 30) and control (n = 21) children and adolescents. To evaluate plasma lipid profile, total cholesterol, LDL-cholesterol, HDL-cholesterol and triglyceride levels were measured. Lipid peroxidation was determined by measuring malondialdehyde concentration. Data were analyzed using Student's t test, Tukey test, ANOVA and Pearson's correlation. RESULTS: Altered levels of total and LDL-cholesterol were observed mainly in overweight or obese males. HDL-cholesterol was borderline in the overweight and obese groups of both sexes. Obese females had the highest levels of triglycerides. Increased plasma lipid peroxidation was observed mainly in obese males. CONCLUSION: In the present population, the greatest alterations in lipid profile were observed in obese and overweight males. Plasma lipid peroxidation was more evident in obese males and females.
Lipid profile; plasma lipid peroxidation; obesity
OBJETIVO: Identificar o perfil lipídico e a peroxidação de lipídeos no plasma de crianças e adolescentes com sobrepeso e obesidade atendidos no Ambulatório de Endocrinologia Pediátrica do HOSPED/UFRN. MÉTODOS: Foram constituídos grupos com crianças e adolescentes com sobrepeso (n = 15), obesidade (n = 30) e controle (n = 21). O perfil lipídico foi avaliado por meio do colesterol total, LDL-colesterol, HDL-colesterol e triglicerídeos. A peroxidação de lipídeos no plasma foi medida pelo marcador malonildialdeído (MDA). A análise estatística foi realizada através do teste t de Student, teste de Tukey, ANOVA e correlação de Pearson. RESULTADOS: As alterações de colesterol total e LDL-colesterol estavam mais presentes nos grupos sobrepeso e obesidade masculinos. O HDL-colesterol mostrou-se em condições limítrofes nos grupos sobrepeso e obesidade em ambos sexos. As maiores concentrações de triglicerídeos foram registradas no grupo obesidade feminino. Identificou-se elevada peroxidação de lipídeos no plasma no grupo obesidade, principalmente no sexo masculino. CONCLUSÕES: Na amostra estudada, maiores alterações do perfil lipídico foram observadas no sexo masculino nos grupos sobrepeso e obesidade, e a peroxidação de lipídeos estava mais evidente no grupo obesidade em ambos sexos.
Análise de lipídeos; peroxidação de lipídeos; obesidade
ORIGINAL ARTICLE
Plasma lipid profile and lipid peroxidation in overweight or obese children and adolescents
Severina C. V. C. LimaI; Ricardo F. ArraisII; Maria G. AlmeidaIII; Zélia M. SouzaIV; Lúcia F. C. PedrosaV
IMSc., Universidade Federal do Rio Grande do Norte (UFRN), Natal, RN, Brazil
IIMSc. Assistant professor, Department of Pediatrics, UFRN, Natal, RN, Brazil
IIIPhD. Associate professor, Department of Toxicology, UFRN, Natal, RN, Brazil
IVAssociate professor, Department of Clinical Analysis, UFRN, Natal, RN, Brazil
VPhD. Associate professor, Department of Nutrition, UFRN, Natal, RN, Brazil
Correspondence Correspondence to Severina Carla Vieira Cunha Lima Rua Epitácio Andrade, 1420/104 CEP 59022-405 - Natal, RN, Brazil Tel.: +55 (84) 213.0095/982.8490 E-mail: scarla@samnet.com.brFinancial support: Fundo de Pesquisa - Pró-reitoria de Pesquisa da UFRN/CNPq (process 300127/91-0)
ABSTRACT
OBJECTIVE: To study the plasma lipid profile and lipid peroxidation in overweight or obese children and adolescents receiving care at the pediatric endocrinology clinic in HOSPED/UFRN, a university hospital.
METHODS: Three groups were studied: overweight (n = 15), obese (n = 30) and control (n = 21) children and adolescents. To evaluate plasma lipid profile, total cholesterol, LDL-cholesterol, HDL-cholesterol and triglyceride levels were measured. Lipid peroxidation was determined by measuring malondialdehyde concentration. Data were analyzed using Student's t test, Tukey test, ANOVA and Pearson's correlation.
RESULTS: Altered levels of total and LDL-cholesterol were observed mainly in overweight or obese males. HDL-cholesterol was borderline in the overweight and obese groups of both sexes. Obese females had the highest levels of triglycerides. Increased plasma lipid peroxidation was observed mainly in obese males.
CONCLUSION: In the present population, the greatest alterations in lipid profile were observed in obese and overweight males. Plasma lipid peroxidation was more evident in obese males and females.
Key words: Lipid profile,plasma lipid peroxidation, obesity.
Introduction
Some evidence suggests that the prevalence of overweight and obesity has significantly increased in the pediatric population, warning against a worldwide epidemic. This is alarming, because metabolic changes and the consequences of obesity, formerly observed only in adults, are now observed in younger individuals.1
In Brazil, demographic, socioeconomic and epidemiological changes have allowed for a transition in nutritional patterns. By comparing the data obtained from the National Survey on Family Expenditure (ENDEF-1974/75) with the data yielded by the Living Standards Measurement Survey (LSMS), conducted in 1996/97, we note an increase from 4.1% to 13.9% in the prevalence of overweight and obesity in children and adolescents aged between 6 and 18 years in the southeast and northeast of Brazil.2
The potential risk factor for a child becoming obese is the frequency of obesity among family members, including genetic and environmental factors, such as eating habits, which determine energy intake and the family's lifestyle.3
Epidemiological studies have shown a higher incidence of cardiovascular diseases in populations with high levels of total cholesterol and of LDL-cholesterol, and with low levels of HDL-cholesterol.4,5 A study with obese children and adolescents, with a median age of 11 years and 10 months for boys and 10 years and 9 months for girls, showed abnormal HDL values in (68.75%) of the patients and a high incidence of elevated triglyceride (35%) and VLDL (37%) levels.6
There is a paucity of studies concerning the prevention of obesity and dyslipidemia, and on the markers of lipid peroxidation, especially in children and adolescents. However, when complications of obesity are already established, as in coronary artery diseases, positive correlations are observed between markers of lipid peroxidation (MDA), total cholesterol and triglycerides (TG), and negative ones are noted with HDL.7
LDL oxidation may be present in specific phospholipid targets on the particle surface (the so-called "minimally oxidized LDL") or cause lipid and protein oxidation inside the particle. Consequently, there is a buildup of by-products such as malondialdehyde (MDA), considered to be a marker of oxidation.8 Thus, oxidized LDL turns macrophages into foamy cells and also increases adhesion, activation and migration of monocytes, resulting in the formation of atheromatous plaques.9
The early detection of lipid peroxidation in obese children and adolescents may help to delay or prevent chronic complications. In this regard, the aim of the present study was to investigate the lipid profile and peroxidation in the plasma of overweight and obese children and adolescents.
Methods
A matched case-control study was carried out with both male and female children and adolescents between January 2001 and December 2002 at the Pediatric Endocrinology Outpatient Clinic of the Children's Hospital of Universidade Federal do Rio Grande do Norte (UFRN), in Natal, Brazil. We assessed a sample of 45 patients among those 63 who were treated at the outpatient clinic during the study period, of whom 15 were diagnosed with overweight and 30 with obesity. The sample size, including the overweight, obesity, and control groups (66) was calculated with a 95%CI and a 6% margin of error. Patients were selected according to the following inclusion criteria: children or adolescents aged between 6 and 16 years, diagnosis of overweight or obesity according to the body mass index (BMI). The exclusion criteria were: absence of genetic syndromes associated with obesity or with any chronic diseases. Of 63 patients, five were not included because they were younger than six years, four had a chronic disease either detected or reported at the time of consultation, two had Prader-Willi syndrome, and seven did not show up for the scheduled exams or tests. Therefore, the final sample consisted of 45 patients.
The control group included 21 children and adolescents among those selected by the clinical screening service of UFRN. These patients were clinically evaluated by a physician and a nutritionist (authors of the study). Of the 30 patients selected for the control group, four were excluded from the study because they revealed an abnormal lipid profile and five because they did not show up for the tests. The following inclusion criteria were used for the control group: healthy nutrition, considering BMI 50th percentile, plasma lipid profile with desirable values and absence of chronic diseases. When matching the groups, some patients in the control group were matched with more than one patient, provided that the gender, age (with a variation up to six months), and level of sexual maturation (nonpubertal and pubertal) were the same, according to Tanner classification.10
The study was approved by the Research and Ethics Committee of UFRN. Parents or guardians gave their written consent after they received detailed and specific information about the risks and benefits of the study.
The BMI was used for the anthropometric assessment, using the cutoff points established by Must et al.11 and recommended by the World Health Organization, which define overweight as the values between the 85th and 95th percentiles; and obesity as the values above the 95th percentile.
Lipid profile was assessed by way of total cholesterol, LDL-cholesterol, HDL-cholesterol and TG, according to Kwiterovich12 reference values, determined by the enzymatic colorimetric assay, using LABTEST reagents. LDL-cholesterol values were obtained through Friedewald formula.
The concentration of lipid oxidation products in the plasma (MDA) was determined by the product that resulted from the reaction between thiobarbituric acid (TBA) and the aldehydes produced during lipid oxidation. The results were expresses in hmols/mL of plasma.13
The data were analyzed by STATISTICA version 5, using Student's t tests for BMI, analysis of variance (ANOVA), Tukey test, and Pearson's correlation coefficient for total cholesterol, LDL, HDL, TG and MDA between groups. A p value < 0.05 was established as statistically significant.
Results
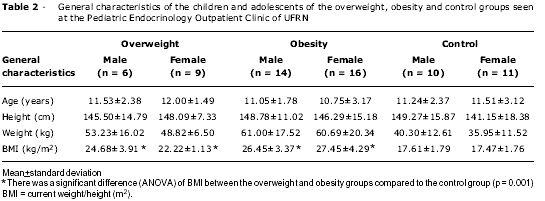
Of 66 children and adolescents, the mean ages in the overweight group were 11.53±2.38 years (range: 8.6 - 14.2) in males, and 12.00±1.49 years (range: 10.2 - 14.9) in females; the mean ages in the obesity group were 11.05±1.78 years (range: 7.6 - 13.5) in males and 10.75?3.17 years (range: 6.1 - 15.6) in females, and the mean ages in the control group were 11.24±2.37 years (range: 7.8 - 14.7) in males and 11.51±3.12 years (range: 6.0 - 15.1) in females.
With regard to the pubertal stage, fourteen (21.2%) boys were at stage 1, twelve (18.2%) at stage 2, and four (6%) at stage 3. As for the girls, ten (15.1%) were at stage 1, nine (13.7%) at stage 2, eight (12.1%) at stage 3, and nine (13.6%) at stage 4 (Table 1).
The general characteristics of the groups are shown in Table 2. BMI was statistically different (p = 0.001) between the overweight and obesity groups, compared to the control group.
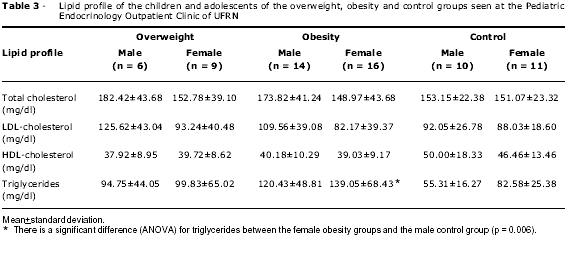
Table 3 shows the lipid profile of the patients. The male overweight and obesity groups had borderline levels of total cholesterol. Elevated LDL-cholesterol levels also considered to be borderline were detected in the male overweight group, but no statistically significant difference was observed. HDL-cholesterol levels in the male and female overweight and obesity groups are within the borderline reference values. Triglyceride levels in the female obesity group were high, with a statistically significant difference (p = 0.006) between the female obesity group and the male control group.
Figure 1 shows higher MDA levels in the male obesity group, differently from the male control group (p = 0.0002) and female control group (p = 0.0005); as well as in the female overweight group (p = 0.0156). The female obesity group showed statistical difference only in relation to the male group when compared to the control groups (p = 0.0243).
In the male obesity group, there was a positive correlation (r = 0.5194, p = 0.057) between BMI and total cholesterol and a negative correlation in the female group (r = -0.4280, p = 0.098) between BMI and HDL-cholesterol, but no statistical difference was observed. No significant correlations were found between lipid profile and MDA.
Discussion
The findings of the present study are consistent with a previous pilot study conducted with a subgroup at the same outpatient clinic, in which a prevalence of 76.9% of obese children and adolescents and a prevalence of 23.1% of overweight children and adolescents were observed.14
BMI did not show any difference between genders in the overweight and obesity groups, as also observed by Valverde et al.6 in children and adolescents.
The lipid profile of the overweight and obesity groups showed mean concentrations, with borderline or undesirable values, especially in male patients. Total cholesterol levels in the overweight and obesity groups were similar to the ones observed in the American population, whose cholesterol levels were 168 mg/dl in males and 173 mg/dl in females;4 which are higher than those obtained by Moura et al.15
There was a propensity for hypercholesterolemia in the overweight and obesity groups, especially in male patients, which might have been influenced by the frequency of pubertal girls (nine in the overweight group and nine in the obesity group). Hormonal changes, found at this stage, may act as a protective factor against changes in lipid profile. Before and after menarche, changes in lipid profile are sensitive to the influence of sex hormones, especially estrogen, which has a favorable effect on lipoproteins by increasing HDL-c and reducing LDL-c levels. In this context, females are at an advantage during adolescence and adulthood.16 Contrary findings were obtained from students in Campinas.17
The Framingham study revealed a continuous increase in the risk for coronary artery disease when cholesterol exceeded 180 mg%18. By analyzing the overweight and obesity groups, we found 12 children and adolescents at risk (four girls and eight boys).
LDL-cholesterol levels in our study were higher than those found by Moura et al.,15 which may be attributed to the high intake of saturated fat and to the low intake of dietary fiber. It should be highlighted that in the male overweight group, 50% of the sample showed undesirable levels of LDL-cholesterol; therefore, it is essential that health services identify children and adolescents at risk for overweight or any other evident risk factors for atherosclerosis.
HDL-cholesterol, which acts as protective factor against heart diseases, showed borderline values in the groups of patients, with lower levels than those found by Webber et al.4 and slightly higher levels than those observed by Moura and Sonati.19 In the overweight and obesity groups, we found 10 girls and 12 boys with HDL < 40 mg/dl. Gerber and Zielinsky analyzed the risk factors for atherosclerosis in childhood and found out that 31 of the children had an HDL < 40 mg/dl associated with total cholesterol >180 mg/dl20. Four (8.7%) patients in the overweight and obesity groups were at risk for atherosclerosis.
We observed a higher prevalence of undesirable TG levels in the male obesity group (35.7%) and in the female obesity group (50.0%), corroborating the findings of Valverde et al.,5 who investigated 74 obese children and adolescents and found abnormal HDL levels in 68.75% of the sample, in addition to elevated TG and VLDL levels.
Changes in MDA as a marker of lipid peroxidation, seen in overweight and obese children and adolescents, may be considered a potential risk factor for cardiovascular complications. Elevated lipid peroxidation has been detected in the plasma of patients with angina pectoris21 and coronary artery diseases.22
Plasma lipid peroxidation was an important marker of obesity, due to the high MDA levels observed, especially in male obese patients, who also showed borderline levels for total cholesterol. The analysis of range intervals of this variable indicates that the lower level observed in the obesity group (1.92±0.47) is higher than the mean value found in the control groups (1.34±0.51). High MDA levels were observed in the male overweight group, which revealed a higher rate of undesirable LDL-c levels (50%), although no statistically significant difference was found.
Our conclusion is that there was some difference as to lipid profile between genders, with elevated levels of total cholesterol and LDL-cholesterol in the male overweight and obesity groups, whereas high triglyceride levels predominated in the female obesity group. The high lipid peroxidation detected in the plasma of patients in the male and female obesity groups is an alert to the clinical management of obesity in pediatric patients, which should give special attention to the possible chronic complications of the disease.
Acknowledgments
Our thanks to nutritionist Ana Sueli de Andrade for her assistance. We also thank the children and adolescents who participated in the study.
References
Manuscript received Nov 07 2003,accepted for publication Oct 15 2003.
- 1. Styne DM. Childhood and adolescent obesity. Prevalence and significance. Pediatr Clin North Am. 2001;48:823-53.
- 2. Wang Y, Monteiro C, Popkin BM. Trends of obesity and underweight in older children and adolescents in the United States, Brazil, China, and Russia. Am J Clin Nutr. 2002;75:971-7.
- 3. Nguyen VT, Larson DE, Johnson RK, Goran MI. Fat intake and adiposity in children of lean and obese parents. Am J Clin Nutr. 1996;63:507-13.
- 4. Webber LS, Organian V, Luepker RV, Feldman HA, Stone EJ, Elder JP, et al. Cardiovascular risk factors among third grade children in four regions of the United States. Am J Epidemiol. 1995;141:428-39.
- 5. Freedman DS, Dietz WH, Srinivasan SR, Berenson GS. The Relation of overweight to cardiovascular risk factors among children and adolescents: The Bogalusa Heart Study. Pediatrics. 1999;103:1175-82.
- 6. Valverde MA, Vítolo MR, Patin RV, Escrivão MAMS, Oliveira FLCO, Ancona-Lopez F. Investigação de alterações no perfil lipídico de crianças e adolescentes obesos. Arch Latinoam Nutr. 1999;49:338-43.
- 7. Mahapatra S, Padhiary K, Mishra TK, Nayak N, Satpathy M. Study on body mass index, lipid profile and lipid peroxidation status in coronary artery disease. J Indian Med Assoc. 1998;96:39-41.
- 8. Navab M, Berliner JA, Watson AD. The Yin and Yang of oxidation in the development of the fatty streak. A review based on the 1994 George Lyman Duff Memorial Lecture. Arterioscler Thromb Vasc Biol. 1996;16:831-42.
- 9. Ross R. The pathogenesis of atherosclerosis: a perspective for the 1990s. Nature. 1993;362:801-09.
- 10. Tanner JM, Whitehouse RH. Clinical longitudinal standards for height, weight, height velocity, weight velocity, and stages of puberty. Arch Dis Child. 1976;62:57-62.
- 11. Must A, Dallal GE, Dietz WH. Reference data for obesity: 85th and 95th percentiles of body mass index (wt/ht2) - a correction. Am J Clin Nutr. 1991;53(4):839-46.
- 12. Kwiterovich POJR. Beyond Cholesterol: The Johns Hopkins Complete guide for Avoiding Heart Disease. Baltimore: The John Hopkins Press; 1989.
- 13. Yagi K. Lipid peroxides in biology and medicine. Washington: Acad Press; 1984.
- 14. Lima SCVC, Arrais RF. Estudo do perfil lipídico e hábitos alimentares das crianças portadoras de sobrepeso e obesidade atendidas no ambulatório de Endocrinologia Pediátrica da UFRN. Rev Saúde. 1999;13:51-5.
- 15. Moura EC, Nakamura SY, Romero VSV. Níveis de colesterol em escolares da primeira série de uma escola pública de Campinas, São Paulo, Brasil. Rev Nutr PUCCAMP. 1997;10:63-9.
- 16. Rabelo LM. Fatores de risco para doença aterosclerótica na adolescência. J Pediatr (Rio J). 2001;77:153-64.
- 17. Moura EC, Castro CM, Mellin AS, Figueiredo DB. Perfil lipídico em escolares de Campinas, SP, Brasil. Rev Saúde Pública. 2000;34:499-05.
- 18. Castelli WP. Epidemiology of coronary heart disease: The Framinghan Study. Am J Med. 1984;76(2A):4-12.
- 19. Moura EC, Sonati JG. Perfil lipídico de dietas e sua relação com os níveis de colesterolemia em escolares de uma escola pública de Campinas, São Paulo, Brasil. Rev Nutr PUCCAMP. 1998;11:69-5.
- 20. Gerber ZRS, Zielinsky P. Fatores de risco de aterosclerose na infância. Um estudo epidemiológico. Arq Bras Cardiol. 1997;69:231-6.
- 21. Mendis S, Sobotka PA, Leja FL, Euler DE. Breath pentane and plasma lipid peroxides in ischemic heart disease. Free Radic Biol Med. 1995;19:679-84.
- 22. Sakuma N, Hibino T, Sato T, Ohte N, Akita S, Tamai N, et al. Levels of thiobarbituric acid-reactive substance in plasma from coronary artery disease patients. Clin Biochem. 1997;30:505-07.
Publication Dates
-
Publication in this collection
22 June 2004 -
Date of issue
Feb 2004
History
-
Received
07 Nov 2002 -
Accepted
15 Oct 2003