Abstracts
OBJECTIVE: To evaluate clinical and histological features of duodenal ulcer in children and adolescents. METHODS: Forty-three children with duodenal ulcer were prospectively and consecutively evaluated in a 6-year period (7.2 patients per year). Evaluation included clinical questionnaire focused on dyspeptic symptoms, physical examination, and digestive endoscopy with gastric biopsies for histological examination and Helicobacter pylori detection. RESULTS: Diagnostic age ranged from 4 years and 8 months to 17 years and 4 months (mean age: 12 years and 4 months). Abdominal pain was the main symptom (39/43, 90.7%), which was epigastric in 31/39, periumbilical in 7/39, and nocturnal in 27/39. Other symptoms were loss of appetite (32/43, 74.4%), vomiting (30/43, 69.8%), postprandial fullness (23/43, 53.5%), weight loss (22/43, 51.2%), and abdominal tenderness (19/43, 44.2%). Upper gastrointestinal bleeding occurred in 19/43 (44.2%), whereas anemia occurred in (21/43, 48.8%). Helicobacter pylori infection was detected in 41/43 (95.3%). All infected patients presented acute chronic gastritis in antrum, with lymphomononuclear infiltrate predominance in 92% of them. Eradication of the bacterium occurred in 68.3%. Ulcer healing occurred in all eradicated patients and in 89% of non-eradicated patients. CONCLUSION: Duodenal ulcer was associated with chronic gastritis due to Helicobacter pylori in the majority of patients. Many complications occurred, especially upper digestive bleeding.
Peptic ulcer; dyspepsia; Helicobacter pylori; children
OBJETIVO: Avaliar as características clínicas e histológicas de crianças e adolescentes com úlcera duodenal. MÉTODOS: Foram avaliadas prospectiva e consecutivamente 43 pacientes com úlcera duodenal ao longo de 6 anos (7,2 pacientes por ano). A avaliação consistiu de questionário clínico de sintomas dispépticos, exame físico e endoscopia digestiva com biópsias gástricas para exame histológico e detecção do Helicobacter pylori. RESULTADOS: A idade no diagnóstico variou de 4 anos e 8 meses a 17 anos e 4 meses (mediana = 12 anos e 4 meses). O sintoma mais freqüente foi dor abdominal (39/43, 90,7%): no epigástrio em 31/39, periumbilical em 7/39 e noturna em 27/39. Outros sintomas freqüentes foram: diminuição do apetite (32/43, 74,4%), vômitos (30/43, 69,8%), plenitude pós-prandial (23/43, 53,5%), perda de peso (22/43, 51,2%) e sensibilidade abdominal (19/43, 44,2%). Hemorragia digestiva alta ocorreu em 19/43 pacientes (44,2%), e anemia em 21/43 (48,8%). H. pylori foi detectado em 41/43 (95,3%), e todos esses pacientes apresentaram gastrite crônica ativa de antro, 92% deles com predomínio do infiltrado linfomononuclear. A erradicação da bactéria ocorreu em 68,3% dos pacientes infectados, mas houve cicatrização da lesão em 100% dos pacientes erradicados e em 89% dos não-erradicados. CONCLUSÃO: Úlcera duodenal foi associada à gastrite crônica por H. pylori na grande maioria dos pacientes. Houve grande freqüência de complicações, especialmente hemorragia digestiva alta.
Úlcera péptica; dispepsia; gastrite; Helicobacter pylori
ORIGINAL ARTICLE
Clinical and histological features of duodenal ulcer in children and adolescents
Elisabete KawakamiI; Rodrigo S. MachadoII; Jacqueline A. FonsecaII; Francy R. S. PatrícioIII
IAssociate professor, School of Medicine, Universidade Federal de São Paulo (UNIFESP), São Paulo, SP, Brazil
IIM.Sc.; Doctoral student, School of Medicine, Universidade Federal de São Paulo (UNIFESP), São Paulo, SP, Brazil
IIIAssociate professor, Department of Pathological Anatomy, School of Medicine, Universidade Federal de São Paulo (UNIFESP), São Paulo, SP, Brazil
Correspondence Correspondence Elisabete Kawakami Rua Loefgreen, 1596 CEP 04040-032 - São Paulo, SP, Brazil Phone/Fax: +55 (11) 5573.8998 E-mail: elkawakami.dped@epm.br
ABSTRACT
OBJECTIVE: To evaluate clinical and histological features of duodenal ulcer in children and adolescents.
METHODS: Forty-three children with duodenal ulcer were prospectively and consecutively evaluated in a 6-year period (7.2 patients per year). Evaluation included clinical questionnaire focused on dyspeptic symptoms, physical examination, and digestive endoscopy with gastric biopsies for histological examination and Helicobacter pylori detection.
RESULTS: Diagnostic age ranged from 4 years and 8 months to 17 years and 4 months (mean age: 12 years and 4 months). Abdominal pain was the main symptom (39/43, 90.7%), which was epigastric in 31/39, periumbilical in 7/39, and nocturnal in 27/39. Other symptoms were loss of appetite (32/43, 74.4%), vomiting (30/43, 69.8%), postprandial fullness (23/43, 53.5%), weight loss (22/43, 51.2%), and abdominal tenderness (19/43, 44.2%). Upper gastrointestinal bleeding occurred in 19/43 (44.2%), whereas anemia occurred in (21/43, 48.8%). Helicobacter pylori infection was detected in 41/43 (95.3%). All infected patients presented acute chronic gastritis in antrum, with lymphomononuclear infiltrate predominance in 92% of them. Eradication of the bacterium occurred in 68.3%. Ulcer healing occurred in all eradicated patients and in 89% of non-eradicated patients.
CONCLUSION: Duodenal ulcer was associated with chronic gastritis due to Helicobacter pylori in the majority of patients. Many complications occurred, especially upper digestive bleeding.
Peptic ulcer, dyspepsia, Helicobacter pylori, children.
Chronic gastritis associated with Helicobacter pylori infection is often observed in children with primary duodenal ulcer, and there is common agreement that the bacterium can be eradicated by the marked reduction in the incidence of ulcer relapses.1 Colonization of the gastric mucosa by Helicobacter pylori, a microaerophilic, spiral-shaped Gram-negative bacillus, is currently uncommon among children who live in industrialized countries, compared to those who live in developing countries, with a prevalence up to the tenth year of life of 5 to 10% and up to 80%, respectively.2 The major risk factors for acquiring the infection are: living in a developing country, being a child with infected parents, and low family income.3 Most patients are asymptomatic, and approximately 15% develop peptic ulcer and 1%, gastric cancer.4 After gastritis is established, the bacterium often persists throughout the patient's life, and spontaneous eradication is rare. There is no definitive conclusion about the influence of bacterial virulence factors, host response or environmental conditions on the course of this disease.
The actual prevalence of duodenal ulcer among pediatric patients remains unclear. Four to six new cases/year are reported in large centers, but gastric ulcer is seldom observed. In developing countries, dissimilar prevalence rates for duodenal ulcer exist due to ethnic or regional differences.5 The clinical symptoms of duodenal ulcer often are atypical, but adolescents have symptoms similar to those observed in adults.6
The aim of the present study is to assess clinical and histological aspects of duodenal ulcer associated with Helicobacter pylori infection in children and adolescents.
Methods
A prospective study was carried out with 34 (79.1%) patients with duodenal ulcer and nine (20.9%) patients with erosive duodenitis (whose clinical course is the same as that of duodenal ulcer) treated at the Division of Digestive Endoscopy, Discipline of Pediatric Gastroenterology of UNIFESP/EPM in a consecutive period of six years (mean of 7.2 cases/year). Patients who had been taking nonsteroidal anti-inflammatory drugs or antibiotics for at least one month before endoscopic examination and those with other digestive or extradigestive diseases were not included in the study.
Clinical assessment
A clinical questionnaire containing dyspeptic signs and symptoms was applied. Patients themselves answered the questions, but their answers were confirmed by their parents, whenever necessary. When the answers were not clear or when the answers given by patients and parents differed, the information was left out of the analysis. The signs and symptoms concerned the ones observed before endoscopic examination, including abdominal pain (site: epigastric, periumbilical, diffuse, other site; frequency; type of pain: burning, colicky, sharp, feeling of hunger, undefined; intensity: mild, moderate, intense; night pain; relationship with food intake: relief, deterioration or no relation), vomiting, nausea, weight loss, loss of appetite, family history, abdominal sensitivity, postprandial fullness, upper gastrointestinal bleeding, and anemia. Pain intensity was related to interference with patient's usual activities. Night pain was defined as the kind of pain that awakens the patient at night. Family history of peptic ulcer (first or second-degree relative) was only considered when peptic ulcer was confirmed by endoscopic examination. The nutritional status was assessed using the body mass index.7
Helicobacter pylori detection
Four endoscopic biopsies were obtained from the antrum within 2 cm of the pylorus for rapid urease test and histological examination (HE and modified Giemsa). The endoscope and the biopsy forceps were disinfected after each procedure by means of intense manual cleaning with an enzyme solution, immersion in a 2% glutaraldehyde solution for 20 minutes and rinsing under running water.
The rapid urease test was prepared on the day of endoscopic examination with an aqueous solution containing urea at 10%, not commercially available, and kept at room temperature.8 For histological analysis, the specimens were placed on a filter paper, positioned and fixed in 10% formaldehyde solution. The spiral-shaped bacterium stained with modified Giemsa and hematoxylin-eosin was detected in the gastric mucosal epithelial layer or on the surface of epithelial cells. Inflammation of the gastric mucosa was assessed according to the modified Sydney system.9 Patients were considered to be infected when the histological exam or rapid urease test yielded positive results. H. pylori eradication after antimicrobial therapy was checked using the same tests on gastric biopsies obtained endoscopically two to three months after the end of treatment.
Treatment for H. pylori infection
Treatments for the eradication of H. pylori were used based on the protocol available at the time of diagnosis. During this period, new treatments replaced those whose eradication rate was not according to expectations. Double or triple therapy containing colloidal bismuth subcitrate (7-8 mg/kg/day in four doses) or proton pump inhibitor was used in association with one or two of the following antibiotics: amoxicillin (50 mg/kg/day in three doses); clarithromycin (15 mg/kg/day in two doses); tetracycline (50 mg/kg/day in three doses for children older than 10 years); nitroimidazole derivatives, tinidazole (10-20 mg/kg/day in two doses) or metronidazole (20 mg/kg/day in two doses); furazolidone (7 mg/kg/day in two doses) for one to two weeks. In cases of therapeutic failure, new antibiotic therapies were used, replacing those which more frequently resulted in bacterial resistance (clarithromycin and nitroimidazole derivatives) or the length of treatment was extended from one to two weeks. Three patients used colloidal bismuth subcitrate for four to eight weeks combined with amoxicillin and nitroimidazole derivative. H2 receptor antagonists (ranitidine 4-6 mg/kg/day, up to 150 mg in two doses, for six to eight weeks) or proton pump inhibitors (omeprazole 0.7 mg/kg/day up to 20 mg, for four weeks) followed antimicrobial therapy.
Statistical analysis
Quantitative variables were described as median and variance, whereas qualitative variables were described as sample proportion.10
The study protocol was approved by the Medical Ethics Committee of UNIFESP- EPM.
Results
Age at the onset of symptoms in 43 patients ranged from three years to 16 years and four months (median = 10 years and nine months), whereas age at diagnosis ranged from four years and eight months to 17 years and four months (median = 12 years and four months). The diagnosis was established in 72.1% (31/43) of the cases after the age of 10 years, and the male/female ratio was 1.65:1.
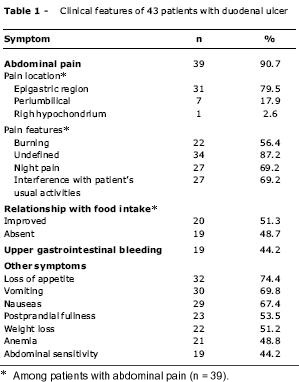
Clinical features are shown in Table 1. Ulcer complications were observed in 48.8% of patients (21/43): upper gastrointestinal bleeding affected 44.2% (19/43) and gastroduodenal obstruction due to pyloric stenosis or duodenal stenosis affected 4.7% (2/43) of the patients. In most of the patients (16/19, 84.2%), gastric bleeding was accompanied by abdominal pain, but after three days the pain disappeared; 11 patients had acute hemorrhage and eight had previously complained of hemorrhage. In these eight patients, the major complaint at diagnosis was abdominal pain in six and vomiting in two. Out of 19 patients with gastric bleeding, 16 had anemia (hemoglobin levels between 4.5 and 9.9 mg/dl), 11 needed blood transfusion, but no patient required endoscopic hemostasis. Peristaltic contractions in the epigastric region associated with fullness and vomiting were the major complaint of a seven-year-old with pyloric stenosis. With regard to the nutritional status at the time of diagnosis, 76.7% of the patients were well-nourished (15th - 85th percentiles), 11.6% showed weight deficit (percentile < 15) and 11.6% were overweight (percentile > 85). Three of five patients with weight deficit had ulcer complications.
The time between symptom onset and diagnosis was longer in younger patients: in 10 patients with symptom onset before the age of six years it was of 1 year and 4 months to 10 years and 6 months (median = 5 years); in 12 patients with symptom onset between 6 and 10 years it was of 1 month to 5 years and 10 months (median = 2 years and 8 months) and in 21 patients with symptom onset after the age of 10 years it was of 2 days to 2 years and 7months (median = 1 year). The diagnosis was established earlier when gastric bleeding was present. The time interval between symptom onset and the endoscopic examination in 12 patients with gastric bleeding as major complaint ranged from 2 days to 2 months (median = 6 days) whereas in 26 patients with abdominal pain, it ranged from 9 months to 10 years (median = 2 years and 6 months).
Family history of peptic ulcer disease was observed in 24/42 (57.1%) patients, but one patient was excluded for having been adopted. Duodenal ulcer was observed in seven first-degree relatives and in three second-degree relatives, gastric ulcer was detected in six first-degree relatives and in four second-degree relatives, and was undefined in four second-degree patients.
Helicobacter pylori
The bacterium was found in 41/43 patients (95.3%), and the rapid urease test and histological examination were positive in 85.4% (35/41). Histological examination alone was positive in five patients, compared to one patient in the rapid urease test. This patient had had a positive histological examination before the treatment and had not been previously treated with eradication therapy.
Histology
All 43 patients had active chronic gastritis. The intensity of the neutrophil infiltrate was mild in 43%, moderate in 35% and intense in 22%. The histological analysis of the gastric mucosa showed lymphoid follicles in 28% of the patients, being associated with nodular lesions in the gastric antrum in 31% of the cases.
Treatment for H. pylori infection. Helicobacter pylori was eradicated after antimicrobial therapy in 68.3% of the patients (28/41), with different therapeutic regimens. In 64.3% (18/28) the bacterium was eradicated after the first antimicrobial treatment, in 14.3% (4/28) after the second treatment and in 21.4% (6/28) after the third treatment. When the bacterium was not eliminated, antibiotics that had not been previously used or those which did not result in bacterial resistance (nitroimidazole derivatives) were chosen. Healing of the ulcer occurred in 100% of the patients in whom the bacterium was successfully eradicated and in 89% of those in whom it was not
Discussion
The annual frequency of duodenal ulcer (7.2 new cases per year) was slightly higher than that described at other referral centers (four to six cases/year), but similar to that obtained in another center in Brazil, which reported 27 patients in four years, 6.75 patients a year.11 Recently, a Russian study has surprisingly reported 31 adolescents in one year, all of them with H. pylori-positive duodenal ulcer.12 Roma et al., who reviewed the results of endoscopic examinations of 2,550 children/nine years with gastrointestinal symptoms, reported a 2% prevalence of peptic ulcer, of which 81% was duodenal.13
Duodenal ulcer occurs at any age, but its frequency increases after the tenth year of life, as observed in this study.1 This age-related increase in incidence may be related to the epidemiology of Helicobacter pylori infection, whose prevalence increases with age in both industrialized and developing countries.2 Late diagnosis occurred mainly in younger children, as pointed out by other authors.14
Abdominal pain was the most frequent symptom, corroborating the findings of other studies, and it affected the epigastric region in most of the patients.11,12,14 Typical characteristics of peptic ulcer disease, such as improvement with the intake of some foods, albeit less frequent among children, do occur and should be investigated.15 Abdominal pain should be distinguished from functional abdominal pain, much more prevalent among children, which may interfere with usual activities, and the differential diagnosis with organic disease based only on clinical criteria may be a difficult task. Clinical data with a better discriminatory power to differentiate between symptomatic children with and without ulcer are: pain in the epigastric region, relationship of pain to food intake, vomiting, night pain (patient is awakened), bitter taste in the mouth, retrosternal burning, upper gastrointestinal bleeding and family history.12,16 Night pain, despite sensitive, is not so specific, and if the high prevalence of functional abdominal pain is taken into account, its positive predictive value is low.16,17
Family history of peptic ulcer has been reported in 26 to 68% of the cases, and is more frequent, the closer the consanguineous relationship between family members.14,15 In the present study, we considered relatives up to the second degree of consanguinity or affinity, and because of that, our results might have been underestimated as only cases with endoscopic diagnosis were registered. The occurrence of family cases may result from the dissemination of Helicobacter pylori in the household, since the history of peptic ulcer disease among family members is more frequent in infected patients. These observations may be explained by the intrafamilial transmission of H. pylori, from a common source or from person-to-person comtact.18
Complications of duodenal ulcer include upper gastrointestinal bleeding, perforation and obstruction.15,19 Its relative frequency is higher among children, probably due to late diagnosis.19 Diagnosis may be further delayed when the major complaint is abdominal pain, compared with hematemesis, as observed in this study. In patients in whom abdominal pain is the major clinical symptom, difficulties related to the characterization of pain and to the high prevalence of functional abdominal pain may delay the diagnosis. In our study, hemorrhage was the most frequently reported ulcer complication, while in the literature, the frequency of upper gastrointestinal bleeding ranges from 24 to 80%.1,20 Hematemesis as an isolated symptom without abdominal pain occurred in 15.8% (3/19) of the patients, but it may be present in up to 25% of the cases.21 Only one patient had pyloric obstruction, whereas a study conducted in southern India in 198322 reported 8/12 patients with pyloric stenosis out of 29 patients with duodenal ulcer in 18 years, comparatively to much lower rates reported by other authors.
In the present study, chronic gastritis associated with Helicobacter pylori infection was observed in most patients, similarly to what occurred in other studies conducted in different countries, in which this infection was detected in up to 100% of patients with duodenal ulcer, although some studies report rates as low as 27%.1,12,18,20,23 This difference may be attributed to the focal distribution of the bacterium and to the lower number of biopsy specimens collected. Histological examination and the rapid urease test exhibit good diagnostic accuracy, as observed in our setting, with a sensitivity and specificity for the histological analysis of 92 and 100% and of 100 and 84.2% for the rapid urease test.8 The patient in whom only the urease test was positive had had a positive histological examination before, in addition, the biopsy revealed neutrophil infiltrate, a finding that indicates high specificity for Helicobacter pylori infection, as the bacterium may have a focal distribution.24
The presence of duodenal ulcer in infected patients is mainly related to the occurrence of antral gastritis,1,20,24 as observed in the present study. There has been predominance of polymorphonuclear infiltrate in adult patients,25 whereas in children, inflammatory infiltrate is usually more chronic, with a significantly larger number of lymphoplasmacytic cells than polymorphonuclear ones.21 However, histological findings in infected children are not consistent, and seemingly depend on socioeconomic conditions.26
The two uninfected patients had neutrophil infiltrate, which indicates high specificity for Helicobacter pylori infection, and perhaps, the bacterium has not been detected because of the focal distribution of the infection.24 The use of a noninvasive test, as the 13C urea breath test, could complement the diagnosis, but unfortunately this test is not available for routine use in our setting.27
The antimicrobial treatments used showed a low eradication rate (68.3%). There is no therapeutic regime that is 100% efficient; therefore, it is necessary to combine antimicrobials taking into consideration regional factors, such as bacterial resistance and level of adherence to onerous medical treatments, with multiple drugs and side effects. So, there is no therapeutic regimen universally validated for children.28,29 Throughout the years, after combining several antibiotics with antisecretory drugs or bismuth salts, we obtained the best results with triple therapy containing amoxicillin, clarithromycin and proton pump inhibitor with an eradication rate of 73 and 50% in 10 and seven days of treatment, respectively,30 and more recently, 88.9% in a preliminary intention-to-treat assessment of 18 patients (unpublished data) with furazolidone, clarithromycin and omeprazole for seven days.
In conclusion, duodenal ulcer was associated with chronic gastritis and Helicobacter pylori infection in most children and adolescents. Complications were highly frequent, including especially upper gastrointestinal bleeding, and late diagnosis occurred especially in younger children.
References
7. Center for Diseases Control/National Center for Health Statistics. 2000 United States Growth Charts [site na internet]. Disponível: http://www.cdc.gov/growthcharts. Acessado: 10 de janeiro de 2003.
Manuscript received Jan 09 2004, accepted for publication Mar 31 2004.
- 1. Drumm B, Rhoads JM, Stringer DA, Sherman PA, Ellis LE, Durie PR. Peptic ulcer disease in children: etiology, clinical findings and clinical course. Pediatrics. 1988;82:410-4.
- 2. Megraud F, Brassens-Rabbe MP, Denis F, Belbouri A, Hoa DQ. Seroepidemiology of Campylobacter pylori infection in various populations. J Clin Microbiol. 1989;27:1870-3.
- 3. Malaty HM, Graham DY. Importance of childhood socioeconomic status on the current prevalence of Helicobacter pylori infection. Gut. 1994;35:742-5.
- 4. Hansson LE, Nyren O, Hsing AW, Bergstrom R, Josefsson S, Chow WH, et al. The risk of stomach cancer in patients with gastric or duodenal ulcer disease. N Engl J Med. 1996;335:242-9.
- 5. Tovey F. Peptic ulcer in India and Bangladesh. Gut. 1979;20:329-47.
- 6. Chelimsky G, Czinn S. Peptic ulcer disease in children. Pediatr Rev. 2001;22:349-55.
- 8. Ogata SK, Kawakami E, Patricio FR, Pedroso MZ, Santos AM. Evaluation of invasive and non-invasive methods for the diagnosis of Helicobacter pylori infection in symptomatic children and adolescents. Sao Paulo Med J. 2001;119:67-71.
- 9. Dixon MF, Genta RM, Yardley JH, Correa P. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol. 1996;20:1161-81.
- 10. Altmann DG. Practical Statistics for Medical Research. 1st ed. London: Chapman & Hall; 1991.
- 11. Ferreira CT, Pires ALG, Soares MG, Silva GL, Lima JP, Silveira TR. Ulcera péptica na criança. Rev AMRIGS. 1989;33:296-9.
- 12. Nijevitch AA, Sataev VU, Vakhitov VA, Loguinovskaya VV, Kotsenko TM. Childhood peptic ulcer in the Ural area of Russia: clinical status and Helicobacter pylori-associated immune response. J Pediatr Gastroenterol Nutr. 2001;33:558-64.
- 13. Roma E, Kafritsa Y, Panayiotou J, Liakou R, Constantopoulos A. Is peptic ulcer a common cause of upper gastrointestinal symptoms? Eur J Pediatr. 2001;160:497-500.
- 14. Murphy MS, Eastham EJ, Jimenez M, Nelson R, Jackson RH. Duodenal ulceration: review of 110 cases. Arch Dis Child. 1987;62:554-8.
- 15. Deckelbaum RJ, Roy CC, Lussier-Lazaroff F, Morin CL. Peptic ulcer disease: a clinical study in 73 children. CMAJ. 1974;111:225-8.
- 16. Tomomasa T, Hsu JY, Shigeta M, Itoh K, Ohyama H, Terashima N, et al. Statistical analysis of symptoms and signs in pediatric patient with peptic ulcer. J Pediatr. Gastroenterol Nutr. 1986;5:711-5.
- 17. Olafsdottir E, Ellertsen B, Berstad A, Fluge G. Personality profiles and heart rate variability (vagal tone) in children with recurrent abdominal pain. Acta Paediatr. 2001;90:638-42.
- 18. Drumm B, Perez-Perez GI, Blaser MJ, Sherman PM. Intrafamilial clustering of Helicobacter pylori infection. N Engl J Med. 1990;322:359-63.
- 19. Raffensperger JC, Condon JB, Greengard J. Complications of gastric and duodenal ulcers in infancy and childhood. Surg Gynecol Obstet. 1966;123:1269-74.
- 20. Israel DM, Hassal E. Treatment and long-term follow-up of Helicobacter pylori associated duodenal ulcer disease in children. J Pediatr. 1993;123:53-8.
- 21. Hassal E, Dimmick JE. Unique features of Helicobacter pylori disease in children. Dig Dis Sci. 1991;36:417-23.
- 22. Gahukamble DB, Fenn AS, Devadatta J, Mammen KE. Duodenal ulcer in South Indian children. Am J Surg. 1983;145:322-4.
- 23. Elitsur Y, Lawrence Z. Non-Helicobacter pylori related duodenal ulcer disease in children. Helicobacter. 2001;6:239-43.
- 24. Warren JR. Gastric pathology associated with Helicobacter pylori Gastroenterol Clin North Am. 2000;29:705-51.
- 25. Wyatt JI, Dixon MF. Chronic gastritis a pathogenic approach. J Pathol. 1988;154:113-24.
- 26. Bedoya A, Garay J, Sanzon F, Bravo LE, Bravo JC, Correa H, et al. Histopathology of gastritis in Helicobacter pylori-infected children from populations at high and low gastric cancer risk. Hum Pathol. 2003;34:206-13.
- 27. Kawakami E, Machado RS, Reber M, Patricio FR. 13 C-urea breath test with infrared spectroscopy for diagnosing Helicobacter pylori infection in children and adolescents. J Pediatr Gastroenterol Nutr. 2002;35:39-43.
- 28. Roma-Giannikou E, Shcherbakov PL. Helicobacter pylori infection in pediatrics. Helicobacter. 2002;7:50-5.
- 29. Hassal E. Approach to Helicobacter pylori infection in children. In: Hunt RH, Tytgat GNJ, editors. Helicobacter pylori: basic mechanisms to clinical cure. 1st ed. London: Kluwer Abcademic Publishers; 2000. p. 575-86.
- 30. Kawakami E, Ogata SK, Portorreal AC, Magni AM, Pardo ML, Patricio FR. Triple therapy with clarithromycin, amoxicillin and omeprazole for Helicobacter pylori eradication in children and adolescents. Arq Gastroenterol. 2001;38:203-6.
Correspondence
Publication Dates
-
Publication in this collection
07 Oct 2004 -
Date of issue
Aug 2004
History
-
Accepted
31 Mar 2004 -
Received
09 Jan 2004