Abstracts
OBJECTIVE: Meningococcal disease continues to be a serious public health concern, being associated with high morbidity and mortality rates worldwide, particularly in Brazil. In addition to discussing recent changes in the global epidemiology of meningococcal disease, we also analyze the development and impact of new conjugate vaccines on the prevention of meningococcal disease, with emphasis on the different immunization strategies implemented with these vaccines. SOURCES OF DATA: MEDLINE databases were searched from 1996 to 2006, with emphasis on review articles, clinical trials and epidemiological studies. Information was also sought on the Centers for Disease Control and Prevention, Brazilian Ministry of Health and Centro de Vigilância Epidemiológica do Estado de São Paulo websites. SUMMARY OF THE FINDINGS: Five serogroups (A, B, C, W135 and Y) are responsible for virtually all cases of the disease worldwide, with marked regional and temporal differences. The new meningococcal serogroup C conjugate vaccines (MCC) offer unmistakable advantages over polysaccharide vaccines. MCC vaccines generate a more efficient and long-lasting antibody response, inducing immunologic memory and reduction of nasopharyngeal carriage. The immediate results of introducing these vaccines into immunization programs have been encouraging, with a dramatic reduction in the incidence of serogroup C disease, not only in vaccinated, but also in unvaccinated individuals (herd immunity). However, concerns have arisen regarding the long-term effectiveness of these vaccines, especially for infants vaccinated in the routine schedule. CONCLUSIONS: The reported waning of efficacy more than 1 year after routine infant immunization supports alternative schedules incorporating a booster dose of MCC vaccine given at 12-18 months of age, in order to maintain long-term protection. The recent licensure of the tetravalent meningococcal conjugate vaccine represents, at last, a real possibility of a broader protection against meningococcal disease, although the need to develop an effective vaccine against serogroup B remains.
Neisseria meningitidis; meningococcal disease; conjugate meningococcal vaccines
OBJETIVO: A doença meningocócica é, ainda hoje, um sério problema de saúde pública, estando associada a elevadas taxas de morbidade e letalidade no mundo e, em especial, no Brasil. Além de discutir as recentes mudanças na epidemiologia da doença meningocócica no mundo, analisamos o desenvolvimento e o impacto das novas vacinas conjugadas na prevenção da doença meningocócica, com ênfase nas diferentes estratégias de imunização utilizadas com essas vacinas. FONTE DOS DADOS: Foram pesquisadas as bases de dados MEDLINE no período de 1996 a 2006, com destaque para artigos de revisão, ensaios clínicos e epidemiológicos. Também foi realizada busca de informações nos portais do Centro de Controle de Doenças, Ministério da Saúde do Brasil e Centro de Vigilância Epidemiológica do Estado de São Paulo. SÍNTESE DOS DADOS: Cinco sorogrupos (A, B, C, W135 e Y) respondem por virtualmente todos os casos da doença no mundo, com marcantes diferenças regionais e temporais. As novas vacinas conjugadas contra o meningococo C apresentam inequívocas vantagens em relação às vacinas polissacarídicas. Induzem uma resposta de anticorpos mais eficiente e duradoura, propiciando memória imunológica e redução da incidência de portadores. Os resultados imediatos da introdução dessas vacinas nos programas de imunização foram animadores, com dramática redução da incidência de doença, inclusive em não-vacinados (imunidade de rebanho). Entretanto, recentemente constatou-se perda da eficácia após alguns anos da aplicação da vacina, especialmente entre os lactentes vacinados. CONCLUSÕES:A perda de efetividade das vacinas conjugadas contra o meningococo C, observada após alguns anos da imunização de lactentes jovens, justifica a mudança dos esquemas de vacinação, com a incorporação de uma dose de reforço entre 12 e 18 meses de idade para garantir uma proteção mais duradoura. O recente licenciamento da vacina quadrivalente meningocócica conjugada representa, enfim, a real possibilidade de uma proteção mais abrangente contra a doença meningocócica, restando ainda a necessidade de se desenvolver uma vacina eficaz contra o meningococo B.
Neisseria meningitidis; doença meningocócica; vacinas meningocócicas conjugadas
REVIEW ARTICLE
Meningococcal conjugate vaccines: efficacy and new combinations
Marco Aurélio Palazzi SáfadiI; Analíria Pimentel BarrosII
IProfessor assistente, Faculdade de Ciências Médicas, Santa Casa de São Paulo, São Paulo, SP, Brasil. Membro, Comissão Permanente de Assessoramento em Imunizações, Secretaria da Saúde do Estado de São Paulo, São Paulo, SP, Brasil. Coordenador, Serviço de Infectologia Pediátrica, Hospital São Luiz, São Paulo, SP, Brasil
IIMestre. Professora adjunto, Faculdade de Ciências Médicas, Universidade de Pernambuco, Recife, PE. Membro, Departamento de Infectologia Pediátrica, Sociedade Brasileira de Pediatria, Rio de Janeiro, RJ, Brasil. Presidente, Departamento de Infectologia, Sociedade de Pediatria de Pernambuco, Recife, PE, Brasil
Correspondence
ABSTRACT
OBJECTIVE: Meningococcal disease continues to be a serious public health concern, being associated with high morbidity and mortality rates worldwide, particularly in Brazil. In addition to discussing recent changes in the global epidemiology of meningococcal disease, we also analyze the development and impact of new conjugate vaccines on the prevention of meningococcal disease, with emphasis on the different immunization strategies implemented with these vaccines.
SOURCES OF DATA: MEDLINE databases were searched from 1996 to 2006, with emphasis on review articles, clinical trials and epidemiological studies. Information was also sought on the Centers for Disease Control and Prevention, Brazilian Ministry of Health and Centro de Vigilância Epidemiológica do Estado de São Paulo websites.
SUMMARY OF THE FINDINGS: Five serogroups (A, B, C, W135 and Y) are responsible for virtually all cases of the disease worldwide, with marked regional and temporal differences. The new meningococcal serogroup C conjugate vaccines (MCC) offer unmistakable advantages over polysaccharide vaccines. MCC vaccines generate a more efficient and long-lasting antibody response, inducing immunologic memory and reduction of nasopharyngeal carriage. The immediate results of introducing these vaccines into immunization programs have been encouraging, with a dramatic reduction in the incidence of serogroup C disease, not only in vaccinated, but also in unvaccinated individuals (herd immunity). However, concerns have arisen regarding the long-term effectiveness of these vaccines, especially for infants vaccinated in the routine schedule.
CONCLUSIONS: The reported waning of efficacy more than 1 year after routine infant immunization supports alternative schedules incorporating a booster dose of MCC vaccine given at 12-18 months of age, in order to maintain long-term protection. The recent licensure of the tetravalent meningococcal conjugate vaccine represents, at last, a real possibility of a broader protection against meningococcal disease, although the need to develop an effective vaccine against serogroup B remains.
Key words: Neisseria meningitidis, meningococcal disease, conjugate meningococcal vaccines.
Introduction
Few diseases have as much power to cause panic among the population as meningococcal disease, primarily because of its potential epidemic nature the rapid onset of illness and, in certain situations, its severity and high fatality rates. Worldwide, approximately 500,000 cases of invasive meningococcal disease occur annually, leaving around 60,000 patients with permanent sequelae and causing more than 50,000 deaths.1,2 In the light of this, the possibility of preventing this infection with vaccines takes on fundamental importance. Motivated by increasing incidence rates of serogroup C meningococcal disease, some countries have introduced the new meningococcal C conjugate vaccines into their routine immunization schedule, leading to a significant reduction in incidence during the period immediately following vaccination.3 However, after some years, reports of cases of meningococcal disease in vaccinated children raised doubts about the duration of vaccine effectiveness in infants vaccinated during the first months of life.4 In this article we present a summary of the epidemiologic trends of meningococcal disease worldwide and discuss the impact of the introduction of new meningococcal conjugate vaccines in several different countries and the prospects for future strategies for the prevention of meningococcal disease.
Global epidemiology of meningococcal disease
The causative agent of meningococcal disease, Neisseria meningitidis, is a gram-negative, aerobic, non-motile diplococcus belonging to the Neisseriaceae family. Meningococci can be classified into 13 different serogroups based on the antigenic composition of the polysaccharide capsule: A, B, C, D, H, I, K, L, W135, X, Y, Z, 29E. Serogroups A, B, C, Y and W135 are responsible for virtually all cases of the disease, infecting humans only. Meningococci can be further classified into serotypes and serosubtypes, according to the antigenic composition of their outer membrane proteins (OMP) PorB and PorA, respectively.5
Invasive infections by N. meningitidis results in a wide clinical spectrum that includes meningitis and meningococcemia, or both, with meningitis being the most common clinical presentation.6 Against this background, the term "meningococcal disease" is appropriate and has been adopted internationally.7 The distribution of patients by sex reveals a slight predominance of the disease among male patients. The disease also exhibits seasonality, with a larger number of cases during the winter.8
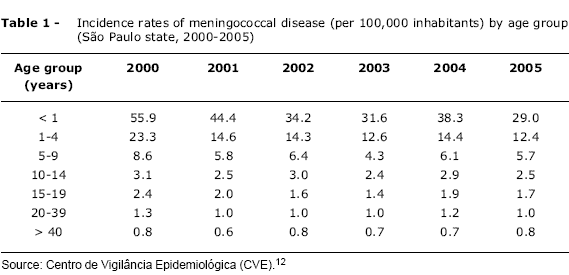
Meningococcal disease affects individuals of all age groups, but the highest incidence is in children under 5, and especially among infants aged 3 to 12 months.9,10 During epidemics, a shift in the age-distribution of meningococcal disease is observed, with increased numbers of cases among adolescents and young adults.11 The higher incidence of disease among infants, from 3 months of age, is related to a reduction in maternal antibody titers that had been passively acquired during pregnancy.5 From 12 months on, children develop naturally acquired immunity, with increased protective antibody titers and, consequently, reduced incidence rates. A second peak in incidence can be observed, in some populations, among adolescents and young adults, probably as a result of increased risk of transmission of the disease, particularly in college students residing in dormitories.9 It is important to point out that, in contrast with what is observed in the USA and certain European countries, in Brazil no increased incidence of the disease has been observed among adolescents and young adults12 (Table 1).
Meningococcal disease occurs all over the world, although there are marked geographical differences in incidence and in the distribution of the different serogroups that cause disease. Historically, serogroup A is associated with epidemic disease in developing countries, especially in Sub-Saharan Africa, which is known as the meningitis belt. The annual incidence of disease during these epidemics can be as high as 1,200 cases per 100,000 inhabitants. Outbreaks of meningococcal disease caused by the W135 serogroup have been recently reported among nomadic Muslims in Saudi Arabia and also in countries in the African belt.13
In developed regions such as the USA and in Europe, the disease is mostly endemic. In Europe, more than 95% of cases are attributed to serogroups B and C. A high proportion of cases attributed to serogroup B are seen in Norway, Germany, Denmark and Holland, while in Spain, Greece, Slovakia, the Czech Republic, Ireland and the United Kingdom, a proportional increase in cases attributed to the C serogroup were observed from the end of the 1990s onwards.14,15 In the USA, incidence rates of meningococcal disease have remained steady over recent years (approximately 1 case per 100,000 inhabitants), with serogroup B being the primary cause of endemic disease and serogroup C related to outbreaks among adolescents and young adults. Increases in the proportion of cases due to serogroup Y have been observed during the last decade, mainly among adults and the elderly.13,16 New Zealand has been affected by an epidemic situation caused by meningococcus B (strain B:4:P1,7-2.4) since 1991, with a total of 5,300 cases of meningococcal disease and 215 deaths reported between 1991 and 2004 with incidence rates of up to 14 cases per 100,000 inhabitants in 2003. During that period around 80% of cases were attributed to the epidemic strain.17 In 2004 New Zealand embarked upon a mass vaccination program for children and adolescents under 19 with a vaccine using outer-membrane vesicle (OMV) proteins developed specifically to combat the epidemic strain in a partnership between the New Zealand government, the Norwegian Institute of Public Health and the Chiron laboratory. Characteristics peculiar to epidemics caused by meningococcus B are insidious onset and prolonged duration, sometimes as long as 10 years.18
During the 1970s, Brazil suffered its largest recorded epidemic of meningococcal disease, with its epicenter in São Paulo and characterized by two overlapping epidemic waves, the first provoked by serogroup C meningococcus, starting in April 1971, and the second by serogroup A meningococcus, beginning in April 1974, without the incidence of cases related to serogroup C meningococcus returning to endemic values by then. The incidence rate, which had been 2.1 cases per 100 thousand inhabitants in 1970, reached a peak of 179 cases per 100,000 inhabitants in 1974. This epidemic provided the first major experience with polysaccharide A and C vaccines on a large scale, resulting in control of the epidemic from 1975 on.19 During the 1980s there was a period of reduced disease incidence (1 case/100,000 inhabitants), with serogroup B becoming more prevalent than C and practically no cases of serogroup A reported. From 1987 onwards there was an increase in the number of cases, with epidemics attributed to serogroup B in several locations around the country. This growth reached its peak in 1996, with 7,104 cases recorded (4.5 cases/100,000 inhabitants), to a great extent resulting from outbreaks in large cities such as São Paulo and Rio de Janeiro. Nevertheless, from 2002 onwards an increase in the proportion of cases attributed to serogroup C was recorded, demonstrating an increasing trend in the proportion of this serogroup in some regions of the country, such as, for example, the state of São Paulo, where it was responsible for 63% of identified cases of meningococcal disease in 2005, with serogroup B responding for 32% of cases and other serogroups for 5% (Figure 1). The case fatality rates of this disease in our country are unfortunately very high, around 18 to 20% in recent years.12,20
Development of conjugate vaccines
The polysaccharide vaccines currently available offer protection against serogroups A, C, W135 and Y. These vaccines, in common with other unconjugated polysaccharide vaccines, do not generate adequate immunoresponse in children under 2 years of age because of the lack of response to T-independent antigens at this age. Another important characteristic of these vaccines is that, even in patients over 2 years of age, the protection offered is of limited duration; they are unable to induce immune memory. Furthermore, they are capable of inducing hyporesponsiveness after subsequent doses. For these reasons these polysaccharide vaccines are not used as routine, but are indicated only for high risk groups or during outbreaks or epidemics.21,22
The conjugation of polysaccharides to protein carriers (non-toxic diphtheria mutant toxin [CRM197] or tetanus toxoid) alters the nature of the antipolysaccharide response to a T-dependent response. When B cells recognize the polysaccharide they process the conjugated carrier protein and present peptide epitopes to T-CD4+ cells. This antigenic complex induces the production of elevated antibody levels, including in young infants, higher antibody avidity and increases serum bactericidal activity. They also induce the formation of long-lasting memory B lymphocyte populations, providing an excellent amnestic response (booster effect) on re-exposure. Furthermore, these vaccines have the capacity to reduce nasopharyngeal colonization, reducing the number of carriers among those vaccinated and so transmission of the disease within the population (herd immunity).5,23-29
The first conjugate vaccines, developed during the 1980s, contained meningococcal A and C capsular oligosaccharides conjugated to a mutant diphtheria toxin - CRM197. Initial studies with these vaccines confirmed good immunogenicity, induction of immunologic memory and an acceptable safety profile.30-32 Nevertheless, the low prevalence of meningococcal disease caused by serogroup A in developed countries directed the development of conjugate meningococcal vaccines towards controlling disease caused by serogroup C. Therefore monovalent conjugated meningococcal vaccines were developed from meningococcal serogroup C isolates containing O-acetyl groups (Oac(+)) in their polysaccharide capsule, conjugated to the mutant diphtheria toxin (MCC-CRM197 - Meningitec - Wyeth Laboratories, and Menjugate - Chiron Laboratories). These vaccines proved themselves immunogenic in infants, toddlers, older children, adolescents and adults.24-29
Later, studies to characterize meningococcus antigens found that around 12% of meningococcal serogroup C isolates that cause disease do not have O-acetyl groups in their polysaccharide capsules (Oac(-)).33 This finding suggested the possibility that the immunoresponse, based fundamentally on group-specific antibodies,34 provoked by these vaccines using Oac(+) polysaccharides, could be ineffective against Oac(-) strains. A vaccine was then developed that utilized a de-O-acetylated capsular polysaccharide conjugated to a tetanus toxoid (MCC-TT- Neisvac-C - Laboratory Baxter). This Oac(-) vaccine elicits the production of antibodies aimed at epitopes present in meningococcal serogroup C isolates with and without O-acetyl groups, thus generating a wider-ranging response and higher serum bactericidal antibody titers (SBA).35-38
The routine immunization schedule currently licensed in Brazil for meningococcal vaccines conjugated to mutant diphtheria toxins (MCC-CRM197 - Meningitec and Menjugate) is three doses, from 2 months of age, with a minimum interval of 1 month between doses and, for the meningococcal vaccine conjugated to tetanus toxoid (MCC-TT - Neisvac-C), the schedule is two doses, from 2 months, with a minimum interval of 2 months between doses. For children aged 1 year and older, adolescents and adults, any of the vaccines should be given in a single dose.
As a result of the low incidence of meningococcal disease caused by serogroup C, randomized, controlled, phase III trials that assessed the efficacy of the vaccine in a given population were impractical. Serological correlates of protection against meningococcal serogroup C disease were therefore used to infer the effectiveness of these vaccines.5,38 The parameter accepted as a correlate for protection, i.e. the lowest antibody titer required to consider an individual protected is an SBA ≥ 4, using human complement, obtained from classic studies undertaken by Dr. Goldschneider with North-American recruits in the 1960s.39 As a response to shortages of human complement, laboratories replace it with baby rabbit complement. While SBA titers obtained using rabbit complement do not correspond exactly to SBA titers from human complement, it is accepted that titers of SBA ≥ 8 correlate to protection when complement obtained from baby rabbits is used.40,41 An additional method for confirming immunity is by detecting a fourfold or greater increase in antibody titers after vaccination. The presence of immunologic memory is a fundamental element of protection with these vaccines and it can be confirmed by a substantial antibody response after challenge with a dose of polysaccharide vaccine and by increases in antibody avidity index as time passes, indicating the presence of T-mediated response.5 Both pre- and post-licensing trials demonstrated excellent immunogenicity and the presence of immunologic memory associated with all three available conjugate vaccines.24-29,35-38
Phase I and II trials demonstrated that MCC vaccines had a reactogenicity profile that was comparable with that observed with other vaccines on the routine schedule. These were mainly characterized by local reactions like tenderness and erythema at the site of administration of the vaccine and systemic reactions like irritability, fever, headaches, vomiting and myalgia.25,27,28,35,36 After its inclusion on the United Kingdom immunization program, passive and active surveillance systems were set up to detect adverse events associated with MCC vaccines. Active surveillance was maintained on 2,800 vaccinated infants and four adverse events considered serious were observed (hypotonia, uncontrollable crying, agitation and maculopapular exanthema).42 After around 18 million doses had been administered alone or concomitantly with other vaccines, the passive surveillance system detected an incidence of 1 case of seizures for every 60,000 doses and 1 case of anaphylactic reactions for every 500,000 doses, with no associated deaths reported.43 In an observational study carried out in our country, we monitored for up to 30 days 151 children who were given meningococcal vaccine conjugated to mutant diphtheria toxin and it was possible to confirm that the vaccine was well tolerated, with few adverse events, characterized basically by fever (14%) and irritability (7%) among infants under 1 year and local reactions (25%) among those over 1 year. We did not record any adverse events considered serious during the study.44
In terms of possible interactions with other vaccines administered concomitantly or in combination, there is growing evidence to suggest that the response to meningococcal C conjugate vaccines may be reduced, whether conjugated to tetanus toxoid or to mutant diphtheria toxin, when administered at the same time as heptavalent pneumococcal conjugate vaccine or acellular combined vaccines.45-47
European experience of mass immunization with meningococcal C conjugate vaccine
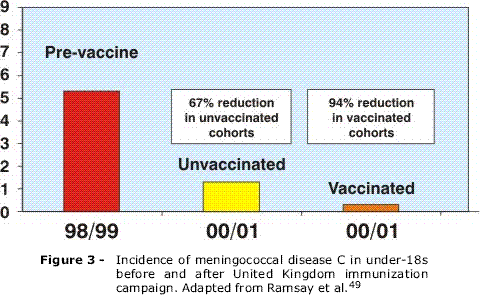
The first place to introduce the conjugate vaccine on a routine schedule was the United Kingdom, in 1999, vaccinating, in less than 1 year, more than 15,000,000 people under 17 years old.3 Initial results were encouraging with an 81% reduction in the incidence of meningococcal disease due to serogroup C for 1998-1999 compared with 2000-2001 (Figure 2). The estimated efficacy of the vaccine was up to 97% in adolescents and 92% in preschool children given a single dose of the vaccine and 91% in infants given three doses of the vaccine, at 2, 3 and 4 months. The number of deaths due to meningococcal disease attributed to serogroup C dropped from 67 in 1999 to five in 2001.48 A significant reduction in the incidence of meningococcal disease was observed, even in age groups that were not vaccinated, apparently demonstrating that conjugate vaccines protect not only individuals who are vaccinated, but also the rest of the population, as a result of the reduced number of people carrying bacteria in their nasopharynx49 (Figure 3). Later, other European countries, such as Spain and Ireland, also carried out mass immunization campaigns and introduced the vaccine on routine schedules at 2, 4 and 6 months of age. Holland and Belgium adopted an alternative strategy, introducing the vaccine in a single dose between 12 and 14 months of age on their routine vaccination schedules. The success achieved in the United Kingdom was also observed in these other countries, with significant reductions in the incidence of meningococcal serogroup C disease.38
The effect of mass vaccination on the numbers of carriers was assessed by a study in the United Kingdom involving 16,000 adolescents aged 15 to 17 years, which observed a reduction of 66% in rates of people carrying serogroup C meningococci in their nasopharynx, comparing periods before and after the introduction of conjugate vaccines.50 In the same study no replacement with other meningococcus serogroups was observed in the vaccinated population.
One concern that arose after the dramatic reduction in the incidence of meningococcal disease from serogroup C in countries that have adopted mass vaccination was the possibility that the introduction of conjugate vaccine would exercise immune pressure on the meningococci population, resulting in replacement with another serogroup, for example B, for which effective vaccines do not exist. To date surveillance data from the United Kingdom show that in parallel with the drop in incidence of meningococcal disease caused by serogroup C, no increases were observed in the absolute number of cases caused by serogroup B.51 Nevertheless, attention was aroused by a phenomenon recently observed in Spain, where, after the introduction of conjugate vaccine against serogroup C, an outbreak was observed caused by the ST11 meningococcus B clone, which had previously been associated predominantly with serogroup C, leading to a suspicion that capsular switching had taken place, associated with the mass immunization.52
Doubts about the real possibility of capsular switching and serogroup replacement emphasize the need for a rigorous surveillance system in areas that introduce conjugate meningococcal vaccines to their routine vaccination schedules.
Long term protection
It is important to point out that licensure was granted for these vaccines solely on the basis of safety and immunogenicity studies. The immunization program with meningococcal conjugate vaccines for serogroup C (MCC), implemented in the United Kingdom, was successful at controlling the incidence of meningococcal disease caused by serogroup C, as a result of high short-term effectiveness with the MCC vaccines. Nevertheless, the duration of the protection conferred by these vaccines remains uncertain.
Studies that have assessed the long-term immunogenicity of the conjugate vaccines have revealed SBA titers reduced to below levels correlated with protection few years after the immunization of these children.26,27,53,54 This waning in seroprotection was observed both in infants, who were given three doses of the vaccine during their first year of life, and in toddlers given just one dose of the vaccine. The studies also demonstrated that the fall in antibodies was observed with all of the conjugate vaccines, although it was less accentuated with the meningococcal vaccine conjugated to tetanus toxoid than with the meningococcal vaccine conjugated to mutant diphtheria.37,55 Nevertheless, in all of these studies excellent immunoresponse was demonstrated after a challenge dose of polysaccharide vaccine was given, clearly indicating immunologic memory. In relation to the memory immunoresponse to booster doses, it was also observed that the response was stronger in children who had been given just one dose of conjugate vaccine in the first year of life compared with children who had been given two or three doses of vaccine.55 It was initially believed that the presence of immunologic memory and herd immunity would be sufficient to guarantee the effectiveness of the vaccine, despite the fall in SBA titers. At this point we should emphasize that in meningococcal disease the agent can invade a few hours after it has colonized the nasopharynx, whereas the immunoresponse, even in individuals who have previously exhibited a response, requires days to take effect adequately. It is, therefore, easy to understand that immunologic memory alone is not sufficient to guarantee protection against invasive meningococcal disease.
In relation to herd immunity, there is no certainty on its duration; there are also concerns that, after the number of carriers in the population has dropped, there may be an increase in individual susceptibility to meningococcus C due to the absence of opportunities for natural exposure to the agent. It is therefore believed that the protection offered by the meningococcal C conjugate vaccine to populations subjected to mass immunization depends on a combination of persistence of bactericidal antibody titers, induction of herd immunity and presence of immunologic memory.
All of these considerations gained prominence after the publication of the first studies, starting in 2004, that confirmed fall in effectiveness more than 1 year after vaccination.53 For infants vaccinated at 2, 3 and 4 months, in the United Kingdom, effectiveness dropped from 93% in the first year to -81% (-7,430 to 71) more than 1 year after vaccination. There were 21 cases of meningococcal C disease in vaccinated children, more than 1 year after the end of the vaccination course, with 18 of these cases occurring during 3 years' follow-up; for those vaccinated between 1 and 2 years of age, effectiveness dropped from 92% in the first year to 61% (-327 to 94) 1 year after vaccination; for the remaining vaccinated age groups, protection 1 year after vaccination remained above 80% (Figure 4). A similar phenomenon was observed in Spain, with infants vaccinated at 2, 4 and 6 months of age.56,57 Therefore, as had been verified with the vaccine for Haemophilus influenzae b (Hib), the protection conferred by the MCC vaccines is directly related to the age at which they are given, i.e. those children given the vaccine aged 1 year or older develop stronger and longer lasting protection than those given routine vaccination during the first months of life.
An alternative strategy that has been shown to be more cost-effective was adopted in Holland, when, in 2002, a routine immunization program was implemented with a single dose of the meningococcal C- tetanus toxoid conjugate vaccine at 14 months of age. In addition, a catch-up campaign was conducted targeting nearly all children and adolescents aged 1 to 18 years with the same vaccine. The first results, published in 2006, demonstrated a dramatic reduction in the incidence of meningococcal disease, both in vaccinated as well as in unvaccinated age groups, with the highest reduction (99%) observed in the age groups that were vaccinated. To date, there have been no reports of meningococcal disease in previously vaccinated children in Holland, although the follow-up period was shorter than in Spain or the United Kingdom.58 These results suggest that the adoption of a routine immunization program using just one dose of meningococcal C conjugate vaccine given during the second year of life offers the possibility of excellent and, probably, long lasting protection against meningococcal disease. Since adolescents are generally the primary carriers of meningococcus C in the population, their inclusion in catch-up campaigns will also result in efficient herd immunity induction. It is also clear that this strategy is only possible in countries, like Holland, which do not have high incidence rates of meningococcal disease during the first year of life.
The results of these studies53,56,57 that demonstrate waning efficacy after some years, particularly among children routinely immunized during the first few months of age, given two or three doses of vaccine, indicate that there is a need to implement different routine immunization schedules from those currently in use. The incorporation of a booster dose after 1 year of age does appear to be unquestionably necessary to guarantee long-term protection for infants immunized during their first year. Portugal was the first European country to add a booster dose at 15 months of age to their routine schedule of primary immunization with two doses of meningococcal C conjugate vaccine, at 3 and 5 months of age. In February 2006, the United Kingdom announced changes to its vaccination schedule, altering the routine immunization regimen for meningococcal C conjugate vaccine to two doses, at 3 and 4 months, with a booster dose at 12 months of age using a combination serogroup C meningococcal-Haemophilus influenzae b conjugate vaccine (MenC-Hib), in addition to incorporating the heptavalent conjugate pneumococcal vaccine.59 In countries like Brazil, where the meningococcal C conjugate vaccine is not yet part of the Programa Nacional de Imunizações (Brazilian National Immunization Program) and therefore without possibility of inducing herd immunity through reduced numbers of carriers, the need for a booster dose for children vaccinated during their first 6 months of life to confer longer-lasting protection seems even more obvious. Among those aged over 1 year, vaccinated with a single dose, efficacy remains elevated, demonstrating that the protection offered by the MCC vaccines is age dependent, and children vaccinated at older ages exhibit more consistent and long-lasting responses, as already observed after vaccination with Hib conjugate vaccines.
Mathematical models attempting to predict the prevalence of serogroup C meningococcal disease in the United Kingdom after varying vaccination schedules indicated that the best cost-effectiveness would be obtained with a single dose at 12 to 15 months.60 This strategy does not, however, appear a possible option in countries with epidemiological conditions similar to those found in Brazil, since in our country a significant proportion of meningococcal disease cases occur among infants under 12 months of age.2,19,20 Considering that the memory response after a booster dose is more substantial in children given just one dose of vaccine during the first year of life compared with those given two or three doses,55 an interesting and cost-effective solution for countries like Brazil could perhaps be an immunization program with a regimen using just one dose for infants, followed by a second dose after 1 year of age.
New conjugate meningococcal vaccine presentations
In 2005, on the basis of short-term safety and immunogenicity data, the USA licensed a quadrivalent meningococcal vaccine containing 4 µg of capsular polysaccharides from serogroups A, C, W135 and Y conjugated to 48 µg of diphtheria toxoid protein (Menactra - Sanofi-Pasteur).61 Studies conducted with this vaccine demonstrated an acceptable safety profile, with no severe adverse events observed during pre-licensure studies. Furthermore, excellent immunogenicity was confirmed among adolescents 11 to 18 years old after one dose of the vaccine (with 99.8, 98.8, 99.5 and 98.6% of individuals reaching SBA titers > 1/128 for the serogroups A, C, Y and W135, respectively) and also among adults aged 18 to 55 years (with 99.8, 98.8, 97.0 and 97.1% of individuals reaching SBA titers > 1/128 for serogroups A, C, Y and W135, respectively).1,62 Nevertheless, the results of studies assessing the immunogenicity of the vaccine in infants were no better than modest, suggesting the need for alternative presentations, perhaps with the addition of adjuvants, in order that this quadrivalent conjugate meningococcal vaccine could be used for the prevention of meningococcal disease in infants.63 In May 2005, the Advisory Committee on Immunization Practices (ACIP) recommended the quadrivalent meningococcal conjugate vaccine for all adolescents aged 11 to 12 years in the USA, and also for adolescents who have not been vaccinated before high-school entry at 15 years.61 In October 2005, the Vaccine Adverse Event Reporting System (VAERS), coordinated by the Centers for Disease Control and Prevention (CDC) in the USA, notified the occurrence of five cases of Guillain-Barré syndrome (GBS) in adolescents aged 17 to 18 years, 14 to 31 days after receiving the quadrivalent meningococcal conjugate vaccine. At that point 2.5 million doses of the vaccine had been distributed in the USA. Based on previous data from the CDC and North-American healthcare agencies, the expected risk of GBS in the 11 to 19 year-old population is one to two cases per 100,000 inhabitants/year. It was, therefore, concluded that the GBS incidence rate identified 6 weeks after vaccination of the adolescents was comparable to the expected rate for the population, based on surveys from previous years. The pre-licensure trials, involving 7,000 adolescents, did not detect any cases of GBS among those vaccinated. In partnership with North-American private healthcare networks, the CDC began rapid monitoring of adverse events, covering 110,000 vaccinated adolescents, without identifying any cases of GBS among these adolescents. Another aspect of relevance is the fact that in the United Kingdom, between 1999 and 2005, 30 million doses of meningococcal C conjugate vaccines were given to children and adolescents younger than 18 years of age, with five cases of GBS reported amongst vaccinated people. This rate was below that expected for the population in question. This being so, the data available to date are insufficient to establish a link between the quadrivalent conjugate meningococcal vaccine and the occurrence of GBS.64
A conjugate vaccine that combined nine pneumococcus serotypes with meningococcal serogroup C was developed with the intention of easing its introduction into the already saturated infant immunization calendar. However, the first results published in 2005 demonstrated that the immunogenicity for meningococcal C was reduced, limiting its development.47
The major challenge in meningococcal disease prevention continues to be the development of vaccines that are immunogenic against serogroup B. The capsular polysaccharide of meningococcus B has an antigenic structure (acetylneuraminic a-2-8-N acid) similar to that found in embryonic neural tissues. This peculiar characteristic, in addition to making it impossible for polysaccharide vaccines containing serogroup B to be immunogenic, also results in a risk of autoimmune reactions to the use of these vaccines.65,66 As a result, no polysaccharide conjugate vaccines have been developed for meningococcus B that could be shown to be immunogenic and risk free. One attempt to overcome this problem was to develop vaccines that used non-capsular components of meningococcus B. Vaccines based on outer membrane proteins (OMV), developed in Cuba and Norway, were used with success to control outbreaks; however, the immunoresponse to these vaccines is specific to the serosubtypes of meningococcus B included in the vaccine, preventing the protection offered from covering other meningococcus B serosubtypes.67,68 One alternative, and promising strategy to develop a vaccine against serogroup B appears to be reverse vaccinology, through genome sequencing of the bacteria, in order to identify proteins that could be antigenic candidates.69
Final comments
Meningococcal disease is an important cause of morbidity and death worldwide, particularly among infants and young children. Its high case fatality rates, even in the face of early therapeutic intervention, emphasize the need for vaccines that can be used routinely for the prevention of this disease. Polysaccharide vaccines against serogroups A, C, W135 and Y, while immunogenic in adults, do not offer adequate protection when administered to children less than 2 years old, in addition to not offering lasting protection. The development of vaccines against serogroup C conjugated to protein carriers (non-toxic diphtheria mutant toxin [CRM197] or tetanus toxoid) has changed the nature of the immunoresponse, inducing production of elevated antibody levels, even among young infants, and also offering excellent response on re-exposure (immunologic memory). Furthermore, these vaccines have the capacity to reduce nasopharyngeal colonization by the bacteria, reducing the number of carriers among those vaccinated and transmission of the disease in the general population (herd immunity). These vaccines are highly effective, with a dramatic reduction in the incidence of meningococcal disease caused by serogroup C in countries that have introduced them in their mass immunization programs. However, recent data on waning efficacy among children immunized during their first year of life have raised doubts with relation to the duration of protection and have caused the regimens under which these vaccines are administered to be altered in order to confer longer-lasting protection, adding a booster dose after 1 year of age for children who have been routinely immunized with two or three doses during the first months of life. The possibility of reducing the number of doses necessary for the routine primary immunization of infants to two or even just one dose of the meningococcal C conjugate vaccine, adding a booster dose after 1 year, to guarantee longer-lasting protection, was an attractive possibility, for example to be introduced in the Programa Nacional de Imunizações, as it would offer a cost-effective strategy to reduce the elevated morbidity and mortality of meningococcal disease attributed to serogroup C in Brazil.
On the other hand, to date attempts to obtain an effective and broad-spectrum vaccine for meningococcus B have failed. While OMV vaccines have proven themselves effective for the control of outbreaks caused by strains homologous with the vaccine, they do not offer protection that covers the remaining meningococcus B serosubtypes, in addition to only offering modest immunogenicity in children under 2 years old.70 The use of reverse vaccinology, with genome sequencing of meningococcus B, may perhaps allow the development of a vaccine that offers wide-ranging protection from serogroup B.
Finally, the auspicious announcement of the licensure of a quadrivalent meningococcal conjugate vaccine for serogroups A, C, W135 and Y at last offers the real possibility of reducing the incidence of meningococcal disease epidemics caused by serogroups A and W135 in Africa, and also the meningococcal disease caused by serogroup Y in the USA. It can also be used as a booster dose for adolescents previously immunized with the meningococcal serogroup C conjugate vaccine, extending protection to the other three serogroups of the vaccine.
References
-
1Word Health Organization. Control of meningococcal epidemic disease. WHO practical guidelines. 2nd ed. Geneva: World Health Organization; 1998. http://www.who.int/csr/resources/publications/meningitis/
- 2. Balmer P, Miller E. Meningococcal disease: how to prevent and how to manage. Curr Opin Infect Dis. 2002;15:275-81.
- 3. Miller E, Salisbury D, Ramsay M. Planning, registration, and implementation of an immunization campaign against meningococcal serogroup C disease in UK: a success story. Vaccine. 2001;20 Suppl 1:S58-67.
- 4. Trotter CL, Andrews NJ, Kaczmarski EB, Miller E, Ramsay ME. Effectiveness of meningococcal serogroup C conjugate vaccine 4 years after introduction. Lancet. 2004;364:365-7.
- 5. Granoff DM, Feavers IM, Borrow R. Meningococcal vaccines. In: Plotkin S, Orenstein WA, editors. Vaccines. 4th ed. Philadelphia: Elsevier; 2004. p. 959-88.
- 6. American Academy of Pediatrics. Meningococcal infections. In: Pickering LK, editor. 2003 red book: report of the Committee on Infectious Diseases. 26th ed. Elk Grove Village: American Academy of Pediatrics; 2003. p. 430-6.
- 7. Hart CA, Rogers TR. Meningococcal disease. J Med Microbiol. 1993;39:3-25.
-
8Word Health Organization (WHO) Working Group, Foundation Marcel Mérieux. Control of epidemic meningococcal disease. WHO practical guidelines. Lyon: Fondation Marcel Mérieux; 1995.
- 9. Bilukha OO, Rosenstein N; National Center for Infectious Diseases, Center for Disease Control and Prevention (CDC). Prevention and control of meningococcal disease. Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2005;54:1-21.
-
10Meningococcal vaccines: polysaccharide and polysaccharide conjugate vaccines. Wkly Epidemiol Rec. 2002;77:331-9.
- 11. Peltola H, Kataja JM, Makela PH. Shift in the age-distribution of meningococcal disease as predictor of an epidemic? Lancet. 1982;2:595-7.
-
12Centro de Vigilância Epidemiológica (CVE), Divisão de Doenças de Transmissão Respiratória. www.cve.saude.sp.gov.br Acesso: 26/01/06.
- 13. Rosenstein N, Perkins B, Stephens D, Popovic T, Hughes J. Meningococcal disease. N Engl J Med. 2001;344:1378-88.
- 14. Connolly M, Noah N. Is group C meningococcal disease increasing in Europe? A report of surveillance of meningococcal infection in Europe 1993-6. European Meningitis Surveillance Group. Epidemiol Infect. 1999;122:41-9.
- 15. Noah N, Henderson B; Communicable Disease Surveillance Centre (PHLS). Surveillance of bacterial meningitis in Europe 1999/00. February, 2002. http://www.hpa.org.uk/hpa/inter/m_surveillance9900.pdf Acesso 20/04/06.
- 16. Rosenstein N, Perkins BA, Stephens DS, Lefkowitz L, Cartter ML, Danila R, et al. The changing epidemiology of meningococcal disease in the United States, 1992-1996. J Infect Dis. 1999;180:1894-901.
- 17. Martin D, Lopez L, McDowell R. The epidemiology of meningococcal disease in New Zealand in 2004. Report prepared for the Ministry of Health by the Institute of Environmental Science and Research Limited. 2005. http://www.moh.govt.nz/moh.nsf/238fd5fb4fd051844c256669006aed57/. Acesso: 20/04/06.
- 18. Oster P, Lennon D, O'Hallahan J, Mulholland K, Reid S, Martin D, et al. MeNZB® : a safe and highly immunogenic tailor-made vaccine against the New Zealand Neisseria meningitidis serogroup B disease epidemic strain. Vaccine. 2005;23:2191-6.
- 19. Moraes JC, Barata RB. A doença meningocócica em São Paulo, Brasil, no século XX: características epidemiológicas Cad Saude Publica. 2005;21:1458-71.
- 20. Ministério da Saúde, Secretaria de Vigilância em Saúde. Boletim epidemiológico. Edição especial. 1999. http://dtr2001.saude.gov.br/svs/pub/pdfs/boletim_epi_edicao_especial.pdf Acesso: 20/04/06.
- 21. Lepow ML, Goldschneider I, Gold R, Randolph M, Gotschlich EC. Persistence of antibody following immunization of children with groups A and C meningococcal polysaccharide vaccines. Pediatrics. 1977;60:673-80.
- 22. Danzig L. Meningococcal vaccines. Pediatr Infect Dis J. 2004;23(12 Suppl):S285-92.
- 23. Lesinski GB, Westerink MA. Novel vaccine strategies to T-independent antigens. J Microbiol Methods. 2001;47:135-49.
- 24. Richmond P, Kaczmarski E, Borrow R, Findlow J, Clark S, McCann R, et al. Meningococcal C polysaccharide vaccine induces immunologic hyporesponsiveness in adults that is overcome by meningococcal C conjugate vaccine. J Infect Dis. 2000;181:761-4.
- 25. MacLennan JM, Shackley F, Heath PT, Deeks JJ, Flamank C, Herbert M, et al. Safety, immunogenicity, and induction of immunologic memory by a serogroup C meningococcal conjugate vaccine in infants: a randomized controlled trial. JAMA. 2000;283:2795-801.
- 26. Borrow R, Goldblatt D, Andrews N, Southern J, Ashton L, Deane S, et al. Antibody persistence and immunological memory at age 4 years after meningococcal group C conjugate vaccination in children in the United Kingdom. J Infect Dis. 2002;186:1353-7.
- 27. McVernon J, Maclennan J, Buttery J, Oster P, Danzig L, Moxon ER. Safety and immunogenicity of meningococcus serogroup C conjugate vaccine administered as a primary or booster vaccination to healthy four-year-old children. Pediatr Infect Dis J. 2002;21:747-53.
- 28. Rennels MB, Edwards KM, Keyserling HL, Reisinger K, Blatter MM, Quataert SA, et al. Safety and immunogenicity of four doses of Neisseria meningitidis group C vaccine conjugated to CRM197 in United States infants. Pediatr Infect Dis J. 2001;20:153-9.
- 29. Richmond P, Borrow R, Miller E, Clark S, Sadler F, Fox A, et al. Meningococcal serogroup C conjugate vaccine is immunogenic in infancy and primes for memory. J Infect Dis. 1999;179:1569-72.
- 30. Costantino P, Viti S, Podda A, Velmonte MA, Nencioni L, Rappuoli R. Development and phase 1 clinical testing of a conjugate vaccine against meningococcus A and C. Vaccine. 1992;10:691-8.
- 31. Twumasi PA Jr, Kumah S, Leach A, O'Dempsey TJ, Ceesay SJ, Todd J, et al. A trial of a group A plus group C meningococcal polysaccharide-protein conjugate vaccine in African infants. J Infect Dis. 1995;171:632-8.
- 32. Leach A, Twumasi PA, Kumah S, Banya WS, Jaffar S, Forrest BD, et al. Induction of immunologic memory in Gambian children by vaccination in infancy with a group A plus group C meningococcal polysaccharide-protein conjugate vaccine. J Infect Dis. 1997;175:200-4.
- 33. Borrow R, Longworth E, Gray SJ, Kaczmarski EB. Prevalence of de-O-acetylated serogroup C meningococci before the introduction of meningococcal serogroup C conjugate vaccines in the United Kingdom. FEMS Immunol Med Microbiol. 2000;28:189-91.
- 34. Arakere G, Frasch CE. Specificity of antibodies to O-acetyl-positive and O-acetyl-negative group C meningococcal polysaccharides in sera from vaccinees and carriers. Infect Immun. 1991;59:4349-56.
- 35. Richmond P, Borrow R, Findlow J, Martin S, Thornton C, Cartwright K, et al. Evaluation of de-O-acetylated meningococcal C polysaccharide-tetanus toxoid conjugate vaccine in infancy: reactogenicity, immunogenicity, immunologic priming, and bactericidal activity against O-acetylated and de-O-acetylated serogroup C strains. Infect Immun. 2001;69:2378-82.
- 36. Richmond P, Goldblatt D, Fusco PC, Fusco JD, Heron I, Clark S, et al. Safety and immunogenicity of a new Neisseria meningitidis serogroup C - tetanus toxoid conjugate vaccine in healthy adults. Vaccine. 1999;18;641-6.
- 37. Richmond P, Borrow R, Goldblatt D, Findlow J, Martin S, Morris R, et al. Ability of 3 different meningococcal C conjugate vaccines to induce immunologic memory after a single dose in UK toddlers. J Infect Dis. 2001;183:160-3.
- 38. Snape MD, Pollard AJ. Meningococcal polysaccharide-protein conjugate vaccines. Lancet Infect Dis. 2005;5:21-30.
- 39. Goldschneider I, Gotschlich EC, Artenstein MS. Human immunity to the meningococcus, I: the role of humoral antibodies. J Exp Med. 1969;129:1307-26.
- 40. Borrow R, Andrews N, Goldblatt D, Miller E. Serological basis for use of meningococcal serogroup C conjugate vaccines in the United Kingdom: reevaluation of correlates of protection. Infect Immun. 2001;69:1568-73.
- 41. Andrews N, Borrow R, Miller E. Validation of serological correlate of protection for meningococcal conjugate vaccine by using efficacy estimates from postlicensure surveillance in England. Clin Diag Lab Immunol. 2003;10:780-6.
- 42. Lakshman R, Jones I, Walker D McMurtrie K, Shaw L, Race G, et al. Safety of a new conjugate meningococcal C vaccine in infants. Arch Dis Child. 2001;85:391-7.
- 43. Expert Working Group on Meningococcal. Group C conjugate vaccines. Safety of meningococcal group C conjugate vaccines. Curr Probl Pharmacovigilance. 2000;26:14.
- 44. Santos AG, Almeida FJ, Arnoni M, Sáfadi MAP, Sanajotta A, Lima R, et al. Eventos adversos da vacina conjugada contra o meningococo C. In: XIII Congresso Brasileiro de Infectologia Pediátrica; 2002; Salvador, Brasil.
- 45. Miller E, Southern J, Kitchin N, Cartwright K, Morris R, Burrage M, et al. Interactions between different meningococcal C conjugate vaccines and the Hib component of concomitantly administered diphtheria/tetanus/pertussis/ Hib vaccines with either whole cell or acellular pertussis antigens. In: Proceedings of the 21st Annual Meeting of the European Society of Pediatric Infectious Diseases; 2003; Sicily, Italy.
- 46. Southern J, Crowley-Luke A, Borrow R, Andrews N, Miller E. Immunogenicity of one, two or three doses of a meningococcal C conjugate vaccine conjugated to tetanus toxoid, given as a three-dose primary vaccination course in UK infants at 2, 3 and 4 months of age with acellular pertussis containing DTP/Hib vaccine. Vaccine. 2006;24:215-9.
- 47. Buttery JP, Ridell A, McVernon J, Chantler T, Lane L, Bowen-Morris J, et al. Immunogenicity and safety of a combination pneumococcal-meningococcal vaccine in infants. JAMA. 2005;293:1751-8.
- 48. Balmer P, Borrow R, Miller E. Impact of meningococcal C conjugate vaccine in the UK. J Med Microbiol. 2002;51:717-22.
- 49. Ramsay ME, Andrews NJ, Trotter CL, Kaczmarski EB, Miller E. Herd immunity from meningococcal serogroup C conjugate vaccination in England: database analysis. BMJ. 2003;326:365-6.
- 50. Maiden MC, Stuart JM; UK Meningococcal Carraige Group. Carriage of serogroup C meningococci 1 year after meningococcal C conjugate polysaccharide vaccination. Lancet. 2002;359:1829-31.
-
51Health Protection Agency. Isolates, England and Wales, by region and group (by epidemiological year), 1989 to 2003. http://www.hpa.org.uk/infections/topics_az/meningo/data_meni_t01.htm Acesso: 25/04/06.
- 52. Perez-Trallero E, Vicente D, Montes M, Cisterna R. Positive effect of meningococcal C vaccination on serogroup replacement in Neisseria meningitidis. Lancet. 2002;360:953.
- 53. Trotter CL, Andrews NJ, Kaczmarski EB, Miller E, Ramsay ME. Effectiveness of meningococcal serogroup C conjugate vaccine 4 years after introduction. Lancet. 2004;364:365-7.
- 54. Snape MD, Kelly DF, Green B, Moxon ER, Borrow R, Pollard AJ. Lack of serum bactericidal activity in preschool children two years after a single dose of serogroup C meningococcal polysaccharide-protein conjugate vaccine. Pediatr Infect Dis J. 2005;24:128-31.
- 55. Borrow R, Goldblatt D, Finn A, Southern J, Ashton L, Andrews N, et al. Immunogenicity of, and immunologic memory to, a reduced primary schedule of meningococcal C-tetanus toxoid conjugate vaccine in infants in the United Kingdom. Infect Immun. 2003;71:5549-55.
- 56. Salleras L, Dominguez A, Cardenosa N. Impact of mass vaccination with polysaccharide conjugate vaccine against serogroup C meningococcal disease in Spain. Vaccine. 2003;21:725-8.
- 57. Cano R, Larrauri A, Mateo S, Alcala B, Salcedo C, Vazquez JA. Impact of the meningococcal C conjugate vaccine in Spain: an epidemiological and microbiological decision. Euro Surveill. 2004;9:11-5.
- 58. de Greeff SC, de Melker HE, Spanjaard L, Schouls LM, van Derende A. Protection from routine vaccination at the age of 14 months with meningococcal serogroup c conjugate vaccine in the Netherlands. Pediatr Infect Dis J. 2006;25:79-80.
-
59United Kingdom, Department of Health. Planned changes to the routine Childhood Immunisation Programme. 2006. http://www.dh.gov.uk/PublicationsAndStatistics/LettersAndCirculars/DearColleagueLe
- 60. Trotter CL, Gay NJ, Edmunds WJ. Dynamic models of meningococcal carriage, disease and the impact of serogroup C conjugate vaccination. Am J Epidemiol. 2005;162:89-100.
- 61. Bilukha OO, Rosenstein N; National Center for Infectious Diseases, Centers for Disease Control and Prevention (CDC). Prevention and control of meningococcal disease. Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2005;54:1-21.
- 62. Campbell JD, Edelman R, King JC Jr, Papa T, Ryall R, Rennels MB. Safety, reactogenicity, and immunogenicity of a tetravalent meningococcal polysaccharide-diphtheria toxoid conjugate vaccine given to healthy adults. J Infect Dis. 2002;186:1848-51.
- 63. Rennels M, King J Jr, Ryall R, Papa T, Froeschle J. Dosage escalation, safety and immunogenicity study of four dosages of a tetravalent meningococcal polysaccharide diphtheria toxoid conjugate vaccine in infants. Pediatr Infect Dis J. 2004;23:429-35.
- 64. Centers for Disease Control and Prevention. Guillain-Barré syndrome among recipients of Menactra meningococcal conjugate vaccine - United States, June-July 2005. MMWR Dispatch. 2005;54:1-3.
- 65. Wyle FA, Artenstein MS, Brandt BL, Tramont EC, Kasper DL, Altieri PL, et al. Immunologic response of man to group B meningococcal polysaccharide vaccines. J Infect Dis. 1972;126:514-21.
- 66. Finne J, Leinonen M, Mäkelä PH. Antigenic similarities between brain components and bacteria causing meningitis: implications for vaccine development and pathogenesis. Lancet. 1983;2:355-7.
- 67. Sierra GV, Campa HC, Varcacel NM, Garcia IL, Izquierdo PL, Sotolongo PF, et al. Vaccine against group B Neisseria meningitidis: protection trial and mass vaccination results in Cuba. NIPH Ann. 1991;14:195-207.
- 68. Bjune G, Hoiby EA, Gronnesby JK, Arnesen O, Fredriksen JH, Halstensen A, et al. Effect of outer membrane vesicle vaccine against group B meningococcal disease in Norway. Lancet. 1991;338:1093-6.
- 69. Rappuoli R. Reverse vaccinology, a genome-based approach to vaccine development. Vaccine. 2001;19:2688-91.
- 70. de Moraes JC, Perkins BA, Camargo MC, Hidalgo NT, Barbosa HA, Sacchi CT, et al. Protective efficacy of a serogroup B meningococcal vaccine in Sao Paulo, Brazil. Lancet. 1992;340:1074-8.
Correspondência:
Publication Dates
-
Publication in this collection
01 Apr 2008 -
Date of issue
July 2006