Abstracts
OBJECTIVES: To present evidence-based recommendations for the use of antibiotics for the treatment of the most common acute respiratory infections (ARI) and the available information on the importance of this type of management. SOURCES: MEDLINE and LILACS databases, technical publications by international organizations, national and international directives. The search terms acute respiratory infection, otitis, sinusitis, tonsillitis, pneumonia, antibiotic, guidelines and bacterial resistance were used. Articles cited by the articles selected were analyzed for information of interest. SUMMARY OF THE FINDINGS: Bacterial resistance has grown, to the extent that today it is recognized as a global public health problem. ARI are the most common cause of antibiotic usage within the community; yet a large proportion of these cases, compromising the upper (otitis, sinusitis, tonsillitis) or the lower airways (pneumonia), are the result of viral infections. Recommendations to rationalize the use of antibiotics in patients with ARI have the common objective of minimizing unnecessary antibiotic use, since "antibiotic pressure" is one of the factors triggering bacterial resistance. CONCLUSIONS: It is of great importance to differentiate among ARI patients those who will benefit from the use of antibiotics. The establishment of recommendations for the prescription of antibiotics is one strategy for minimizing the frequency of bacterial resistance.
Antibiotics; bacterial resistance; otitis; sinusitis; tonsilitis; pneumonia
OBJETIVOS: Apresentar as recomendações baseadas em evidência para uso de antibióticos no tratamento das infecções respiratórias agudas (IRA) mais freqüentes e as informações disponíveis sobre a importância desse tipo de conduta. FONTES DOS DADOS: Bases de dados MEDLINE, LILACS, publicações técnicas de organizações internacionais, diretrizes nacionais e internacionais. Foram utilizados os unitermos acute respiratory infection, otitis, sinusitis, tonsillitis, pneumonia, antibiotic, guidelines, bacterial resistance. Artigos citados pelos artigos incluídos foram analisados quanto à apresentação de informação de interesse. SÍNTESE DOS DADOS: A resistência bacteriana tem crescido, sendo atualmente reconhecida como problema mundial de saúde pública. As IRA são a causa mais freqüente para uso de antibiótico na comunidade; grande parte desses casos, tanto nas vias aéreas superiores (otite, sinusite, faringoamidalite) como nas inferiores (pneumonia), são decorrente de infecção viral. As recomendações para racionalizar o uso de antibióticos nos pacientes com IRA têm como objetivo comum minimizar o uso desnecessário de antibióticos, visto que a "pressão antibiótica" é um dos fatores desencadeantes da resistência bacteriana. CONCLUSÕES: É de grande importância a distinção, entre os pacientes com IRA, daqueles que podem se beneficiar do uso de antibióticos. O uso das recomendações para a prescrição de antibióticos é uma estratégia para minimizar a freqüência de resistência bacteriana.
Antibióticos; resistência bacteriana; otite; sinusite; faringoamidalite; pneumonia
REVIEW ARTICLE
Outpatient antibiotic therapy as a predisposing factor for bacterial resistance: a rational approach to airway infections
Cristiana M. Nascimento-Carvalho
Doutora. Professora adjunta, Departamento de Pediatria, Faculdade de Medicina, Universidade Federal da Bahia (UFBA), Salvador, BA, Brasil
Correspondence Correspondence: Cristiana Nascimento de Carvalho Rua Prof. Aristides Novis, 105/1201B CEP 40210-730 - Salvador, Bahia - Brazil Tel./Fax: +55 (71) 3235.7869 E-mail: nascimentocarvalho@hotmail.com
ABSTRACT
OBJECTIVES: To present evidence-based recommendations for the use of antibiotics for the treatment of the most common acute respiratory infections (ARI) and the available information on the importance of this type of management.
SOURCES: MEDLINE and LILACS databases, technical publications by international organizations, national and international directives. The search terms acute respiratory infection, otitis, sinusitis, tonsillitis, pneumonia, antibiotic, guidelines and bacterial resistance were used. Articles cited by the articles selected were analyzed for information of interest.
SUMMARY OF THE FINDINGS: Bacterial resistance has grown, to the extent that today it is recognized as a global public health problem. ARI are the most common cause of antibiotic usage within the community; yet a large proportion of these cases, compromising the upper (otitis, sinusitis, tonsillitis) or the lower airways (pneumonia), are the result of viral infections. Recommendations to rationalize the use of antibiotics in patients with ARI have the common objective of minimizing unnecessary antibiotic use, since "antibiotic pressure" is one of the factors triggering bacterial resistance.
CONCLUSIONS: It is of great importance to differentiate among ARI patients those who will benefit from the use of antibiotics. The establishment of recommendations for the prescription of antibiotics is one strategy for minimizing the frequency of bacterial resistance.
Key words: Antibiotics, bacterial resistance, otitis, sinusitis, tonsilitis, pneumonia.
Introduction
Acute respiratory infections (ARI) are common, cause significant morbidity and contribute significantly to the overall disease load on the community.1 In addition to their important social impact, ARI are a frequent cause of medical care and consumption of antibiotics.2It is estimated that 20-40% of outpatient consultations and 12-35% of hospital admissions to general pediatric hospitals are the result of ARI.3 In Finland, an evaluation of diagnoses made during 1 week at 30 health centers (target population 819,000 people) revealed that 74% of 7,777 consultations were the result of respiratory infections. Taking just children aged less than five years, 84% of infections were of the respiratory airways.4 Although the incidence of ARI is greatest during the first 5 years of life,5 these infections have also been documented as common among adolescents from 10 years onwards, progressing with substantial co-morbidity of upper and lower airways.6 In a recent study undertaken in Pakistan with children aged 2 months to 5 years, the estimated incidence of ARI was 44%.7
Pneumonia stands out from among the other ARI, causing 2 million child deaths per year globally (20% of all child deaths).8 Seventy percent of these deaths occurred in Africa and Southeast Asia, in which regions the incidence of pneumonia is 2 to 10 times greater (7-40% per year) than in the United States.9 In Latin America, the evolution of mortality due to pneumonia has varied greatly between 1990 and 2000: reducing in the majority of countries, as in Chile and Uruguay, while increasing or remaining high in others, for example Bolivia, Peru and Guyana.10 The available data indicate that estimated annual incidence in developed countries is 10 to 15 cases/1,000 children, with annual incidence of hospital admissions due to pneumonia of from 1 to 4/1,000.11 Around 95% of children with pneumonia are given antibiotics.12
Acute otitis media (AOM) is the most common ARI responsible for medical consultations and in these cases antibiotics are widely prescribed.13 In a recent study undertaken in Valencia, in Spain, the incidence of AOM was 40,014 episodes/100,000 children under 5 (95%CI 39,700-40,300), and the frequency of antibiotic use was 94.5%.14 In Israel, the mean duration (days) of antibiotic courses given to children with AOM was 9.0±5.6. 15 AOM is the infection for which it is most common to prescribe antibiotics, which results in more than 20 million antibiotic prescriptions annually.16
It is estimated that patients aged 3 to 17 years attended 7.3 million medical consultations in the United States during 2003 because of sore throats, with antibiotics prescribed at 54% of consultations.17 A population-based study carried out in Sweden, found an annual rate of ARI episodes of 166 per every 1,000 inhabitants; 18% were diagnosed as acute tonsillitis (AT). Overall, the total annual number of antibiotic prescriptions for ARI was 78 for every 1,000 inhabitants.18
While involvement of the sinuses is common in viral respiratory infections, the true incidence of bacterial sinusitis in children is unknown. It is estimated that around 10% of upper airway infections are complicated by acute bacterial sinusitis (ABS).19 A study that evaluated the frequency of nasal congestion and daytime coughing for longer than 9 days in 1,307 children aged 1 to 5 years seen at pediatric clinics in Seattle, USA, found a point prevalence of 9.3% (95%CI 7.7%-10.9%) for ABS; excluding comorbidity with AOM, 5% of the 1,307 children received antibiotics due to ABS.20 A Finnish multicenter study demonstrated that antibiotics are prescribed 2 to 5 times more frequently than the true incidence of ABS warrants.21 ABS has been described as the fifth most common cause of antibiotic prescriptions.22
Use of antibiotics for airway infections
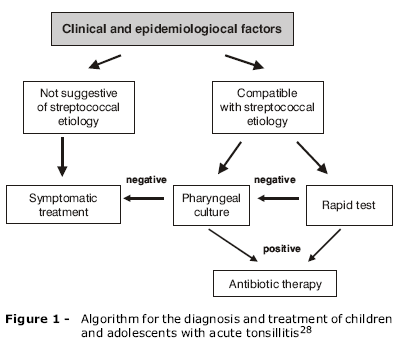
The majority of ARI cases, whether involving the upper airways (AOM, AT, ABS)23 or the lower respiratory tract (pneumonia),24 have viral etiology. The principal etiologic agent of bacterial AT is Streptococcus pyogenes,25 and streptococcal AT can course with suppurative complications, such as peritonsillar abscess, or non-suppurative complications, such as rheumatic fever, which can be prevented with correct use of antibiotics to treat the primary condition (streptococcal AT).26 Strategies for the diagnosis and treatment of patients with AT are aimed to distinguish children with viral AT, who will not benefit from antibiotics, from children with streptococcal AT, for whom antibiotic is beneficial.27 The Infectious Diseases Department of the Brazilian Society of Pediatrics (SBP - Sociedade Brasileira de Pediatria), in accord with the recommendations of the American Academy of Pediatrics and the Infectious Diseases Society of America, have developed an algorithm for the diagnosis and treatment of these patients (Figure 1).28 The distinction between viral and bacterial etiology in children with AT is crucial to minimizing antibiotic use.27
The bacterial etiologic agents most common among patients with pneumonia, AOM and ABS are Streptococcus pneumoniae, Haemophilus influenzae and Moraxella catarrhalis.22,29,30 Among pneumonia patients the significance of bacterial etiology is due to the lethality of these agents.31 A compilation made of studies undertaken in developed countries to investigate the etiology of pneumonia in children, using specific and noninvasive methods, showed that, for those patients where it was possible to determine etiology, the cause was viral in 69% and bacterial in 61% of hospitalized patients, and viral in 48% and bacterial in 65% of those seen in an outpatient setting.30 The World Health Organization (WHO) estimates that up to 1 million children under 5 years old die annually from pneumonia, meningitis or pneumococcal sepsis, and the great majority of these deaths attributed to pneumonia occurs in developing countries.32 Despite the lack of parity in the information available on the frequency of etiologic agents among children with pneumonia in different regions around the world, S. pneumoniae is considered the most common bacterial pathogen among children aged less than 5 years.33
In order to contain the high mortality from pneumonia in developing countries, the WHO has suggested an algorithm (Figure 2) for the management of patients with ARI. This algorithm is based on easily-detected clinical signs, such as tachypnea and subcostal retraction.34 The same algorithm has been recommended by the SBP as a strategy to rationalize the use of antibiotics in children with ARI,35 and there is evidence that the algorithm has a substantial impact on mortality from pneumonia.36 On the other hand, the use of this algorithm (Figure 2) has resulted in a considerable number of children being incorrectly diagnosed with pneumonia (specificity 49-89%).37-39 In a case-control study conducted in Haiti, the majority of children with recurrent pneumonia diagnosed according to the WHO algorithm actually had acute asthma crises.40 Bronchospasm crises also present with tachypnea and, for this reason, it is possible that use of the WHO algorithm occasions unnecessary antibiotic use.41
It is important to point out that the objective of the algorithm for management of patients with ARI is to distinguish from among children with respiratory complaints, those who have lower airway (LA) involvement, with antibiotics being recommended when the LA are affected.42 In contrast with what has been described with relation to AT, the algorithm for managing patients with pneumonia does not make any distinction between viral and bacterial etiology. This is because of the difficulty involved in establishing the etiology of pneumonia and the fact that the high mortality from pneumonia occurs in regions where there are operational limitations in terms of basic technological resources.9-10 The gold standard for the diagnosis of pneumonia is the detection of pulmonary infiltrates on chest X-ray.43 Many attempts have been made to distinguish radiologically between children with pulmonary infiltrate whose etiology is exclusively viral and those whose etiology is bacterial, of which the bacterial pneumonia score (BPS) is an example.44 This type of attempt aims to increase the rational use of antibiotics, reducing their unnecessary utilization. Nevertheless, a Finnish study, using specific and noninvasive methods to determine pneumonia etiology in children, demonstrated that bacterial infections can present both alveolar infiltration and interstitial infiltrate.45 A Brazilian study has shown that X-ray findings can be influenced by the age of the child involved, possibly as a result of the differences in the inflammatory response of infants when compared with older children.46 Therefore, the presence of radiological signs such as consolidation, pleural fluid and pneumatocele can be seen as having an association with bacterial etiology, but the absence of these signs does not signify that etiology is not bacterial.45
The importance of antibiotic therapy for patients with AOM is related to the complications and sequelae that may take hold if etiology is bacterial. Mastoiditis and conductive deafness are two examples.47 The introduction of antibiotics during the 1930s and 1940s led to a significant decline in the incidence, both of complications and of sequelae.48 Nevertheless, in 1972, Howie & Ploussard49 demonstrated that spontaneous resolution of AOM is possible and that the defining factor is the etiologic agent: 48% for H. influenzae and 18% for S. pneumoniae. A recent meta-analysis of 63 studies demonstrated that the symptoms of AOM improve within 24 hours without antibiotics in 61% of children (95%CI 50-72%), increasing to 80% in 2 to 3 days (95%CI 69-90%). The frequency of suppurative complications when antibiotics are used immediately (0.24%) is comparable to the frequency of complications when antibiotics are used after an initial observation period (0.12%).50
Evidence of the growing frequency of bacterial resistance among AOM pathogens,51 the knowledge that this resistance is strongly linked with antimicrobial use52 and the difficulties, in practice, of identifying the etiologic agent of AOM (need for tympanocentesis and a culture of middle ear fluid) have resulted in repeated recommendations with the objective of reducing the use of antibiotics in children with AOM.51,53-55
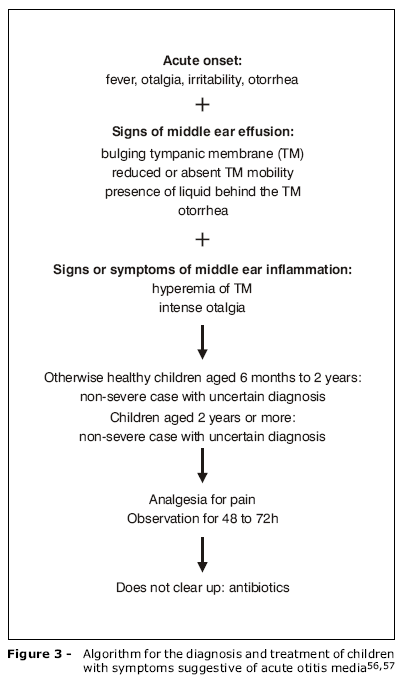
The most recent recommendations were published in 2004 and presented by the AAP56 and the American Academy of Family Physicians (AAFP - Figure 3).57 It is important to point out that these recommendations emphasize the need to increase diagnostic precision when assessing patients with symptoms compatible with AOM and strongly favor the conservative treatment option, i.e., observation without introduction of antibiotics, with symptomatic medication in uncomplicated cases of AOM. These recommendations are innovative in that they propose a rethink of the routine use of antibiotics for patients with AOM, in the expectation that this will prevent the continuing increase of bacterial resistance,58 without failing to treat those cases that would in fact benefit from antibiotics.
There is less evidence-based information available on the subject of patients with ABS than on the conditions described so far.59 In 2001, the AAP published recommendations for the management of patients with clinical status suggestive of ABS based on a consensus among specialists, in order to supplement issues on which evidence was lacking (Figure 4).60 It is important to mention that the recommendations were based on five randomized clinical trials, on eight studies of case series and the opinions of specialists. According to these recommendations, antibiotics are used for the treatment of uncomplicated ABS with the objective of achieving the quickest possible clinical cure. Antibiotics are emphatically recommended for patients suspected or confirmed of complications.
Recent studies carried out in Scandinavian countries have demonstrated that antibiotics are prescribed 2 to 5 times more than the true incidence of ABS (10%).21 Physicians feel under pressure to prescribe antibiotics for ABS, both because of patient expectations and because of the difficulty in precisely diagnosing ABS.61 Although it is difficult to define the precise moment when viral infections become less likely than bacterial ones, the risk of bacterial infection is greater, the longer symptoms last.62 For this reason, diagnosis of uncomplicated ABS for which antibiotics will be beneficial is based on duration of upper airway infection symptoms (nasal congestion and/or discharge, posterior rhinorrhea, coughing) for 10 days, or their worsening after 5 to 7 days. Nasal discharge can be clear, mucoid or purulent, and coughing (wet or dry) should be present during the day, although it will often worsen at night (Figure 4).62
Antimicrobial resistance of respiratory pathogens
Recently, the results of in vitro surveillance of respiratory pathogens in the United States demonstrated that around 45% of S. pneumoniae strains were resistant to penicillin (16% intermediate resistance and 29% absolute resistance), while 33% and 43% of strains were resistant to macrolides and trimethoprim-sulfamethoxazole, respectively.63 Resistance to multiple drugs was significantly more common among strains resistant than among strains susceptible to penicillin.63 Of the H. influenzae strains, 28.6% were beta-lactamase producers, 70.2% were susceptible to amoxicillin, 99.8% to a combination of amoxicillin/clavulanic acid, 82.8% to cefuroxime axetil, 78.1% to trimethoprim-sulfamethoxazole, 23.2% to cefprozil and 5% to cefaclor, azithromycin and clarithromycin.63 Almost all strains of M. catarrhalis were resistant to amoxycillin, cefaclor, cefprozil and trimethoprim-sulfamethoxazole, and beta-lactamase production was the mechanism of resistance.63
Although antibiotic resistance has been spreading over recent years, there is great variation between different locations that have been studied, with high and low penicillin resistance rates being reported in nearby geographical regions.64 This is a very worrying situation, in view of the great ease of transport nowadays, meaning that the movement of people between different areas can rapidly contribute to the dissemination of resistant strains.65 In Brazil, national surveillance data record that, from 1993 to 2004, the frequency of S. pneumoniae strains resistant to penicillin grew from 10.2% to 27.9%.66 Proportions of intermediate and high levels resistance were 9.1% and 1.1% in 1993, reaching 22.0% and 5.9% in 2004.66 Of a total of 1,712 invasive strains of H. influenzae isolated in ten Brazilian states, the prevalence of beta-lactamase production varied from 6.6% to 57.5% between 1996 and 2000, and the general prevalence was 18.4%.67
There is no record of S. pyogenes strains resistant to penicillin, but the same is not true for macrolides. A study undertaken in Spain, evaluating bacteria pathogenic in the respiratory tract, demonstrated that 33.2% of 3,174 strains of S. pyogenes exhibited resistance to erythromycin.68 In France, the rate of resistance to erythromycin was 22.4% in 322 strains of S. pyogenes, being primarily associated with a small number of emm type 28 clones.69 This resistance has increased significantly in certain European countries since the last years of the nineties. In Greece between 1999 and 2002, 24% of 1,577 strains were resistant to erythromycin.70
Evidence that the use of antibiotics is a factor linked to the antibiotic resistance of respiratory pathogens
The respiratory pathogens commented on here are part of the flora that colonize the upper airways and as such are frequently exposed to antibiotics.71 Patients previously exposed to the use of antibiotics have higher incidence of non-susceptible strains than patients who have not previously received antibiotics.72 Nowadays, bacterial resistance is not only a problem in nosocomial infections, but also in community-acquired infections.52 The majority of antibiotics consumed are used to treat community infections and the principal selective pressure to generate changes to the frequency of resistance is the volume of antibiotics consumed.73 What has become known as "antibiotic pressure" is the raw relationship between the quantities of drugs employed and the selection of resistant strains.74 In the United States, 160 million antibiotic prescriptions are filled every year, corresponding to 22.7 million kg, 50% for human consumption and 50% for use on animals, in agriculture and fisheries.75 For a population of 275 million people, this is the equivalent of 30 prescriptions for every 100 people and 4.1 kg of antibiotics for every 100 people annually.74
Antibiotic pressure is different for each drug.64 In Spain, Garcia-Rey et al. studied 1,684 strains of S. pneumoniae isolated from patients with community-acquired respiratory infections and demonstrated that the correlation between the use of antibiotics and bacterial resistance was ascending when penicillins, cephalosporins and macrolides were analyzed separately.76 Three important measurements can be used to predict the level of bacterial resistance at a given location: the proportion of organisms introduced from elsewhere, the proportion selected spontaneously or by antibiotic pressure and the proportion that are transmitted from person to person.74 It is thus possible to understand that the use of antibiotics is extremely important to the process of selection of resistant strains and that more rational and limited use could contain the progression in rates of resistance.
Notwithstanding, reductions in resistance are not influenced to such an extent by reductions in antibiotic use. In Sweden, between 1997 and 2003, an absence of correlation was demonstrated between the frequency of resistant S. pneumoniae and reductions in antibiotic use.77 It is possible that this finding could be explained as a function of the remaining factors, such as introduction and dissemination of resistant strains into the community. In the United States, epidemiological surveillance data have demonstrated a significant fall in the frequency of infections by resistant S. pneumoniae associated with large-scale use of the heptavalent conjugate pneumococcal vaccine.78 These data allow us to suppose that, in the short term, the principal impact factor for the reduction of bacterial resistance will be the reduction of circulation of bacteria in the community. The technical difficulties involved in producing vaccines for all bacteria that cause community respiratory infections are a strong reason for compliance, in clinical practice, with these recommendations for the rational use of antibiotics.
Final comments
Bacterial resistance among respiratory pathogens is recognized as a growing problem and one of great relevance. The use of antibiotics for ARI is one of the factors that influence the occurrence of this phenomenon. While reduction in antibiotic use doesn't appear to be associated with reductions in bacterial resistance over the short term, one of the easiest measures to implement, in the expectation of, at least, reducing the accelerated increase in resistance rates, is the rational use of antibiotics.
References
- 1. Leder K, Sinclair MI, Mitakakis TZ, Hellard ME, Forbes A. A community-based study of respiratory episodes in Melbourne, Australia. Aust N Z J Public Health. 2003;27:399-404.
- 2. Shoham Y, Dagan R, Givon-Lavi N, Liss Z, Shagan T, Zamir O, et al. Community-acquired pneumonia in children: quantifying the burden on patients and their families including decrease in quality of life. Pediatrics. 2005;115:1213-9.
- 3. Jain N, Lodha R, Kabra SK. Upper respiratory tract infections. Indian J Pediatr. 2001;68:1135-8.
- 4. Rautakorpi UM, Klaukka T, Honkanen P, Makela M, Nikkarinen T, Palva E, et al. Antibiotic use by indication: a basis for active antibiotic policy in the community. Scand J Infect Dis. 2001;33:920-6.
- 5. Facanha MC, Pinheiro AC. Distribution of acute respiratory diseases in Brazil from 1996 to 2001, Brazil. Rev Saude Publica. 2004;38:346-50.
- 6. Karevold G, Kvestad E, Nafstad P, Kvaerner KJ. Respiratory infections in schoolchildren: co-morbidity and risk factors. Arch Dis Child. 2006;91:391-5.
- 7. Nizami SQ, Bhutta ZA, Hasan R. Incidence of acute respiratory infections in children 2 months to 5 years of age in periurban communities in Karachi, Pakistan. J Pak Med Assoc. 2006;56:163-7.
- 8. Mulholland K. Global burden of acute respiratory infections in children: implications for interventions. Pediatr Pulmonol. 2003;36:469-74.
- 9. Singh V. The burden of pneumonia in children: an Asian perspective. Paediatr Respir Rev. 2005;6:88-93.
- 10. Fuchs SC, Fischer GB, Black RE, Lanata C. The burden of pneumonia in children in Latin America. Paediatr Respir Rev. 2005;6:83-7.
- 11. Farha T, Thomson AN. The burden of pneumonia in children in the developed world. Paediatr Respir Rev. 2005;6:76-82.
- 12. Weigl JA, Bader HM, Everding A, Schmitt HJ. Population-based burden of pneumonia before school entry in Schleswig-Holstein, Germany. Eur J Pediatr. 2003;162:309-16.
- 13. Cripps AW, Otczyk DC, Kyd JM. Bacterial otitis media: a vaccine preventable disease? Vaccine. 2005;23:2304-10.
- 14. Garces-Sanchez M, Diez-Domingo J, Alvarez de Labiada T, Planelles V, Graullera M, Baldo JM, et al. Epidemiology and burden of acute otitis media in Valencia (Spain). An Pediatr (Barc). 2004;60:125-32.
- 15. Greenberg D, Bilenko N, Liss Z, Shagan T, Zamir O, Dagan R. The burden of acute otitis media on the patient and the family. Eur J Pediatr. 2003;162:576-81.
- 16. Bernius M, Perlin D. Pediatric ear, nose, and throat emergencies. Pediatr Clin North Am. 2006;53:195-214.
- 17. Linder JA, Bates DW, Lee GM, Finkelstein JA. Antibiotic treatment of children with sore throat. JAMA. 2005;294:2315-22.
- 18. Engstrom S, Molstad S, Nilsson G, Lindstrom K, Borgquist L. Data from electronic patient records are suitable for surveillance of antibiotic prescriptions for respiratory tract infections in primary health care. Scand J Infect Dis. 2004;36:139-43.
- 19. Wald ER, Guerra N, Byers C. Upper respiratory tract infection in young children: duration of and frequency of complications. Pediatrics. 1991;87:129-33.
- 20. Aitken M, Taylor JA. Prevalence of clinical sinusitis in young children followed up by primary care pediatricians. Arch Pediatr Adolesc Med. 1998;152:244-8.
- 21. Varonen H, Rautakorpi UM, Huikko S, Honkanen PO, Klaukka T, Laippala P, et al. Management of acute maxillary sinusitis in Finnish primary care. Results from the nationwide MIKSTRA study. Scand J Prim Health Care. 2004;22:122-7.
- 22. Bishai WR. Issues in the management of bacterial sinusitis. Otolaryngol Head Neck Surg. 2002;127(6 Suppl):S3-9.
- 23. West JV. Acute upper airway infections. Br Med Bull. 2002;61:215-30.
- 24. Klig JE. Current challenges in lower respiratory infections in children. Curr Opin Pediatr. 2004;16:107-12.
- 25. Gieseker KE, Roe MH, MacKenzie T, Tood JK. Evaluating the American Academy of Pediatrics diagnostic standard for Streptococcus pyogenes pharyngitis: backup culture versus repeat rapid antigen testing. Pediatrics. 2003;111:e666-70.
- 26. Vincet MT, Celestin N, Hussain AN. Pharyngitis. Am Fam Physician. 2004;69:1465-70.
- 27. Gerber MA. Diagnosis and treatment in children. Pediatr Clin North Am. 2005;52:729-47, vi.
- 28. Nascimento-Carvalho CM, Marques HH. The Brazilian Society of Pediatrics, Department of Infectious Diseases guidelines for the management of children and adolescents with acute pharyngitis. J Pediatr (Rio J). 2006;82:79-80.
- 29. Cripps AW, Otczyk DC, Kyd JM. Bacterial otitis media: a vaccine preventable disease? Vaccine. 2005;23:2304-10.
- 30. Nascimento-Carvalho CM. Etiology of childhood community-acquired pneumonia and its implication for vaccination. Braz J Infect Dis. 2001;5:87-97.
- 31. Shann F. Etiology of severe pneumonia in children in developing countries. Pediatr Infect Dis J. 1986;5:247-52.
- 32. World Health Organization. Pneumococcal vaccines. Wkly Epidemiol Record. 2003;14:110-9.
- 33. Burgner D, Richmond P. The burden of pneumonia in children: an Australian perspective. Paediatr Respir Rev. 2005;6:94-100.
- 34. Cashat-Cruz M, Morales-Aguirre JJ, Mendoza-Azpiri M. Respiratory tract infections in children in developing countries. Semin Pediatr Infect Dis. 2005;16:84-92.
- 35. Nascimento-Carvalho CM, Marques HHS. Recomendação da Sociedade Brasileira de Pediatria para antibioticoterapia em crianças e adolescentes com pneumonia comunitária. Rev Panam Salud Publica. 2004;15:380-7.
- 36. Qazi SA, Rehman GN, Khan MA. Standard management of acute respiratory infections in a children's hospital in Pakistan: impact on antibiotic use and case fatality. Bull World Health Organ. 1996;74:501-7.
- 37. Perkins BA, Zucker JR, Otieno J, Jafari HS, Paxton L, Redd SC, et al. Evaluation of an algorithm for integrated management of childhood illness in an area of Kenya with high malaria transmission. Bull World Health Organ. 1997;75(Suppl 1):33-42.
- 38. Simoes EA, Desta T, Tessema T, Gerbresellassie T, Dagnew M, Gove S. Performance of health workers after training in integrated management of childhood illness in an area with seasonal malaria in the Gambia. Bull World Health Organ. 1997;75 Suppl 1:43-53.
- 39. Weber MW, Mulholland EK, Jaffar S, Troedsson H, Gove S, Greenwood BM. Evaluation of an algorithm for the integrated management of childhood illness in an area with seasonal malaria in the Gambia. Bull World Health Organ. 1997;75 Suppl 1:25-32.
- 40. Heffelfinger JD, Davis TE, Gebrian B, Bordeau R, Schwartz B, Dowell SF. Evaluation of children with recurrent pneumonia diagnosed by World Health Organization criteria. Pediatr Infect Dis J. 2002;21:108-12.
- 41. Torzillo PJ. Wheezing and the management algorithms for pneumonia in developing countries. Indian Pediatr. 2001;38:821-6.
- 42. Benguigui Y, Stein F. Integrated management of childhood illness: an emphasis on the management of infectious diseases. Semin Pediatr Infect Dis. 2006;17:80-98.
-
43World Health Organization. Standardization of interpretation of chest radiographs for the diagnosis of pneumonia in children. Geneva: WHO; 2001.
- 44. Moreno L, Krishman JA, Duran P, Ferrero F. Development and validation of a clinical prediction rule to distinguish bacterial from viral pneumonia in children. Pediatr Pulmonol. 2006;41:331-7.
- 45. Korppi M, Kiekara O, Heiskanen-Kosma T, Soimakallio S. Comparison of radiological findings and microbial aetiology of childhood pneumonia. Acta Paediatr. 1993;82:360-3.
- 46. Nascimento-Carvalho CM, Alves NN, Athayde LA, Caldas RM, Barberino MG, Duarte J, et al. Is there any association of a specific chest x-ray pattern and bacteremia in children with pneumonia? J Trop Pediatr. 2002;48:253-4.
- 47. Leskinen K. Complications of acute otitis media in children. Curr Allergy Asthma Rep. 2005;5:308-12.
- 48. Agrawal S, Husein M, MacRae D. Complications of otitis media: an evolving state. J Otolaryngol. 2005;34 Suppl 1:S33-9.
- 49. Howie VM, Ploussard JH. Efficacy of fixed combination antibiotics versus separate components in otitis media. Effectiveness of erythromycin estolate, triple sulfonamide, ampicillin, erythromycin-estolate-triple sulfonamide and placebo in 280 patients with acute otitis media under two and one-half years of age. Clin Pediatr. 1972;11:205-14.
- 50. Rosenfeld RM, Kay D. Natural history of untreated otitis media. Laryngoscope. 2003;113:1645-57.
- 51. Leibovitz E. Acute otitis media in pediatric medicine: current issues in epidemiology, diagnosis, and management. Paediatr Drugs. 2003;5 Suppl 1:1-12.
- 52. Tenover FC, McGowan JE Jr. Reasons for the emergence of antibiotic resistance. Am J Med Sci.1996;311:9-16.
- 53. Hoberman A, Marchant CD, Kaplan SL, Feldman S. Treatment of acute otitis media consensus recommendations. Clin Pediatr (Phila). 2002;41:373-90.
- 54. Dowell SF, Schwartz B, Phillips WR. Appropriate use of antibiotics for URIs in children: Part I. Otitis media and acute sinusitis. The pediatric URI Consensus Team. Am Fam Physician. 1998;58:1113-8, 1123.
- 55. Paradise JL. Treatment guidelines for otitis media: the need for breadth and flexibility. Pediatr Infect Dis J. 1995;14:429-35.
- 56. American Academy of Pediatrics Subcommittee on Management of Acute Otitis Media. Diagnosis and management of acute otitis media. Pediatrics. 2004;113:1451-65.
- 57. American Academy of Family Physicians. Guideline on Diagnosis and Management of Acute Otitis Media. Am Fam Physician. 2004;69:2713-5.
- 58. Pelton SI. Otitis media: re-evaluation of diagnosis and treatment in the era of antimicrobial resistance, pneumococcal conjugate vaccine, and evolving morbidity. Pediatr Clin North Am. 2005;52:711-28.
- 59. Ioannidis JP, Lau J. Technical report: evidence for the diagnosis and treatment of acute uncomplicated sinusitis in children: a systematic overview. Pediatrics. 2001;108:E57.
- 60. American Academy of Pediatrics. Subcommittee on Management of Sinusitis and Committee on Quality Improvement. Pediatrics. 2001;108:798-808.
- 61. Varonen K, Sainio S. Patients' and physicians' view on the management of acute maxillary sinusitis. Scand J Prim Health Care. 2004;22:22-6.
- 62. Poole MD, Jacobs MR, Anon JB, Marchant CD, Hoberman A, Harrison CJ. Antimicrobial guidelines for the treatment of acute bacterial rhinosinusitis in immunocompetent children. Int J Pediatr Otorhinolaryngol. 2002;63:1-12.
- 63. Jacobs MR, Bajaksouzian S, Windau A. Good CE, Lin G, Pankuch GA, et al. Susceptibility of Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis to 17 oral antimicrobial agents based on pharmacodynamic parameters: 1998-2001 US Surveillance Study. Clin Lab Med. 2004;24:503-30.
- 64. Cardozo DM, Nascimento-Carvalho CM, Souza FR, Silva NM. Nasopharyngeal colonization and penicillin resistance among Pneumococcal Strains: a world-wide 2004 update. Braz J Infect Dis (in press).
- 65. Schito GC. Is antimicrobial resistance also subject to globalization? Clin Microbiol Infect. 2002;8 Suppl 3:1-8, 33-5.
- 66. Brandileone MC, Casagrande ST, Guerra ML, Zanella RC, Andrade AL, Di Fabio JL. Increase in numbers of beta-lactam-resistant invasive Streptococcus pneumoniae in Brazil and the impact of conjugate vaccine coverage. J Med Microbiol. 2006;55:567-74.
- 67. Casagrande ST, Landgraf IM, Kobata AMM, Zanella RC, Bonkermann S. Antimicrobial resistance among invasive Haemophilus influenzae strains: results of a Brazilian study carried out from 1996 through 2000. Braz J Med Biol Res. 2002;35:1293-300.
- 68. Perez-Trallero E, Garcia-de-la-Fuente C, Garcia-Rey C, Baquero F, Aguilar L, Dal-Re R, et al. Geographical and ecological analysis of resistance, coresistance, and coupled resistance to antimicrobials in respiratory pathogenic bacteria in Spain. Antimicrob Agents Chemother. 2005;49:1965-72.
- 69. Bingen E, Bidet P, Mihaila-Amrouche L, Doit C, Forcet S, Brahimi N, et al. Emergence of macrolide-resistant Streptococcus pyogenes strains in French children. Antimicrob Agents Chemother. 2004;48:3559-62.
- 70. Grivea IN, Al-Lahham A, Katopodis GD, Syrogiannopoulos GA, Reinert RR. Resistance to erythromycin and telithromycin in Streptococcus pyogenes isolates obtained between 1999 and 2002 from Greek children with tonsillopharyngitis: phenotypic and genotypic analysis. Antimicrob Agents Chemother. 2006;50:256-61.
- 71. Jacobs MR. Streptococcus pneumoniae: epidemiology and patterns of resistance. Am J Med. 2004;117 Suppl 3A:3S-15S.
- 72. Sokol W. Epidemiology of sinusitis in the primary care setting: results from the 1999-2000 respiratory surveillance program. Am J Med. 2001;111 Suppl 9A:19S-24S.
- 73. Austin DJ, Kristinsson KG, Anderson RM. The relationship between the volume of antimicrobial consumption in human communities and the frequency of resistance. Proc Natl Acad Sci. 1999;96:1152-6.
- 74. Wenzel RP, Edmond MB. Managing antibiotic resistance. N Engl J Med. 2000;343:1961-3.
- 75. Levy SB. Antibiotic resistance: an ecological imbalance. In: Antibiotic resistance: origins, evolution, selection and spread. Ciba Foundation Symposium 207. Chichester, England: John Wiley; 1997. p. 1-14.
- 76. Garcia-Rey C, Aguilar L, Baquero F, Casal J, Dal-Re R. Importance of local variations in antibiotic consumption and geographical differences of erythromycin and penicillin resistance in Streptococcus pneumoniae J Clin Microbiol. 2002;40:159-64.
- 77. Hogberg L, Ekdahl K, Sjostrom K, Olsson-Liljequist B, Walder M, Melander E, et al. Penicillin-resistant pneumococci in Sweden 1997-2003: increased multiresistance despite stable prevalence and decreased antibiotic use. Microb Drug Resist. 2006;12:16-22.
- 78. Levine OR, O'Brien KL, Knoll M, Adegbola RA, Black S, Cherian T, et al. Pneumococcal vaccination in developing countries. Lancet. 2006;367:1880-2.
Correspondence:
Publication Dates
-
Publication in this collection
23 Feb 2007 -
Date of issue
Nov 2006