Abstracts
OBJECTIVE: To develop a predictive model capable of identifying which premature infants have the greatest probability of presenting bronchopulmonary dysplasia (BPD), based on assessment at the end of their first week of life. METHODS: Data were collected retrospectively from January 1998 to July 2001, and prospectively from August 2001 to July 2003. All children born at the institution with gestational age < 34 weeks and birth weight < 1,500 g were included. The principal risk factors for BPD were subjected to univariate analysis followed by logistic regression. Significant variables were used to construct a formula to calculate the probability of BPD. The model was calibrated and its discriminative power assessed using receiver operating characteristic (ROC) curves. Between August 2003 and July 2005 the model was then applied to a different population for validation. RESULTS: The sample comprised 247 children, of whom 68 developed BPD, classified as follows: mild = 35 (51.4%), moderate = 20 (29.4%) and severe = 8 (11.7 %). Four variables maintained significance with relation to BPD: gestational age < 30 weeks, persistent ductus arteriosus, mechanical ventilation > 2 days and loss of > 15% of birth weight on the seventh day of life. Where patients exhibited all of these variables, the model had a 93.7% probability of being correct. The model was further validated when using another sample of 61 newborns; similar figures were obtained. CONCLUSIONS: At the end of the first week of life, the predictive model developed from our population was capable of identifying newborn infants at increased risk of developing BPD with a high degree of sensitivity.
Bronchopulmonary dysplasia; predictive model; chronic neonatal lung disease; mechanical ventilation; prematurity
OBJETIVO: Desenvolver um modelo preditivo capaz de identificar, ao final da primeira semana de vida, os recém-nascidos prematuros com maior probabilidade de evoluir para displasia broncopulmonar (DBP). MÉTODOS: Os dados foram coletados retrospectivamente entre janeiro de 1998 e julho de 2001, e prospectivamente de agosto de 2001 a julho de 2003. Foram incluídas todas as crianças nascidas na Instituição, com idade gestacional < 34 semanas e peso de nascimento < 1.500 g. Os principais fatores de risco foram submetidos inicialmente a uma análise univariada, seguida de regressão logística. As variáveis significativas foram utilizadas na montagem da fórmula para cálculo da probabilidade de ocorrência de DBP. O modelo foi calibrado, e a discriminação avaliada pela curva ROC. De agosto de 2003 a julho de 2005, o modelo foi aplicado em outra população para validação. RESULTADOS: Foram incluídas 247 crianças, das quais 68 evoluíram para DBP, sendo divididas da seguinte maneira: leve = 35 (51,4%), moderada = 20 (29,4%) e grave = oito (11,7 %). Quatro variáveis mantiveram significância em relação à DBP: idade gestacional < 30 semanas, persistência do canal arterial, ventilação mecânica > 2 dias e perda de > 15% do peso de nascimento no sétimo dia de vida. Nos pacientes com todas as variáveis presentes, o modelo permitiu uma probabilidade de acerto de 93,7%. Valores semelhantes foram obtidos com as 61 crianças utilizadas na validação do modelo. CONCLUSÕES O modelo preditivo desenvolvido em nossa população foi capaz de identificar com elevado grau de sensibilidade, ao final da primeira semana de vida, os recém-nascidos sob maior risco de evoluir para DBP.
Displasia broncopulmonar; modelo preditivo; doença pulmonar crônica do recém-nascido; ventilação mecânica; prematuridade
ORIGINAL ARTICLE
Bronchopulmonary dysplasia prediction modelfor 7-day-old infants
Carlos A. BheringI; Christieny C. MochdeceII; Maria E. L. MoreiraIII; José R. RoccoIV; Guilherme M. Sant'AnnaV
IProfessor titular, Universidade Severino Sombra (USS), Vassouras, RJ, Brasil. Doutor, Instituto Fernandes Figueira, Fundação Oswaldo Cruz (Fiocruz), Rio de Janeiro, RJ, Brasil
IINeonatologista. Bolsista, Instituto Fernandes Figueira, Fiocruz, Rio de Janeiro, RJ, Brasil
IIIDoutora. Pesquisadora, Instituto Fernandes Figueira, Fiocruz, Rio de Janeiro, RJ, Brasil IVProfessor adjunto, Universidade Federal do Rio de Janeiro (UFRJ), Rio de Janeiro, RJ, Brasil
VDoutor, Instituto Fernandes Figueira, Fiocruz, Rio de Janeiro, RJ, Brasil. Professor associado, Departamento de Pediatria, McMaster University, Hamilton, Ontario, Canada
Correspondence Correspondence: Guilherme Mendes Sant'Anna McMaster Children's Hospital. Hamilton Health Sciences 1200 Main Street West, 4G40B L8S4J9 Hamilton, ON Canada Tel.: +1 (905) 521.2100, ext. 76487 Email: santang@mcmaster.ca
ABSTRACT
OBJECTIVE: To develop a predictive model capable of identifying which premature infants have the greatest probability of presenting bronchopulmonary dysplasia (BPD), based on assessment at the end of their first week of life.
METHODS: Data were collected retrospectively from January 1998 to July 2001, and prospectively from August 2001 to July 2003. All children born at the institution with gestational age < 34 weeks and birth weight < 1,500 g were included. The principal risk factors for BPD were subjected to univariate analysis followed by logistic regression. Significant variables were used to construct a formula to calculate the probability of BPD. The model was calibrated and its discriminative power assessed using receiver operating characteristic (ROC) curves. Between August 2003 and July 2005 the model was then applied to a different population for validation.
RESULTS: The sample comprised 247 children, of whom 68 developed BPD, classified as follows: mild = 35 (51.4%), moderate = 20 (29.4%) and severe = 8 (11.7 %). Four variables maintained significance with relation to BPD: gestational age < 30 weeks, persistent ductus arteriosus, mechanical ventilation > 2 days and loss of > 15% of birth weight on the seventh day of life. Where patients exhibited all of these variables, the model had a 93.7% probability of being correct. The model was further validated when using another sample of 61 newborns; similar figures were obtained.
CONCLUSIONS: At the end of the first week of life, the predictive model developed from our population was capable of identifying newborn infants at increased risk of developing BPD with a high degree of sensitivity.
Keywords: Bronchopulmonary dysplasia, predictive model, chronic neonatal lung disease, mechanical ventilation, prematurity.
Introduction
Bronchopulmonary dysplasia (BPD) is still the most common cause of morbidity among extremely low birth weight (ELBW) newborns, although the incidence, risk factors and severity of the disease have changed substantially since the introduction of new treatments and mechanical ventilation techniques.1-3 Pathogenesis is the result of multiple factors including immaturity, infection, barotrauma or volutrauma and oxygen toxicity. Excessive ingestion of liquids and sodium have also been suggested as additional risk factors.1
Strategies and interventions that might reduce the incidence of BPD have been widely investigated. Recently, Schmidt et al.3 reported on a multicenter study whose results demonstrated that administration of caffeine during the first days of life was capable of reducing BPD incidence in a population of very low birth weight (VLBW) newborns.4 Nevertheless, in order that such measures achieve greater efficacy, it is necessary to identify early and precisely the newborn population that is at greatest risk of developing the condition.5 Working from this principle, several predictive models have been developed over the last 25 years achieving sensitivity rates that varied from 64 to 92.7%.6-9 However, one factor that has limited widespread adoption of any of these models is the fact that they were developed in specific centers and populations and were not tested prospectively on other populations.
The principal objective of this study was to develop and apply a model to predict BPD at a neonatal intensive care unit in Brazil, using a local population of VLBW infants assessed at the end of their first week of life.
Methods
The study was carried out at the Instituto Fernandes Figueira (IFF), part of the Fundação Oswaldo Cruz (FIOCRUZ), after approval by the Institute's Research Ethics Committee.
Study period
The study employs data from two distinct periods. From January 1998 to July 2003, a predictive model was developed through analysis of the principle risk factors associated with BPD. Data were collected retrospectively, during the period January 1998 to July 2001, by means of direct consultation of patient records and the computerized database at the Department of Neonatology. The data were collected prospectively from August 2001 to July 2003, by means of a spreadsheet. During the second period, August 2003 to July 2005, the predictive model that had been developed was validated. All of the data, whether retrospective or prospective, were collected by just two of the researchers involved in the study (C.A.B. and C.C.M.).
Study population
All children born at the IFF with gestational ages (GA) < 34 weeks and birth weight (BW) < 1,500 g were included if they were free from complex genetic malformations and congenital infections, their mother's were not HIV-positive, and they survived past their 28th day of life.
Definition of bronchopulmonary dysplasia
This study employed the BPD definition based on disease severity proposed by Jobe & Bancalari10 and later validated by Ehrenkranz et al.11 Infants were thus defined as suffering from BPD if they were on oxygen support ≥ 28 days. These newborns were then reassessed when they reached 36 weeks' corrected GA (if GA < 32 weeks) or on the 56th day of life (if GA ≥ 32 weeks) or at hospital discharge. Those who were on room air at the time of reevaluation were classified as having had mild BPD. Those receiving less than 30% fraction of inspired oxygen (FiO2) were classified as having moderate BPD and those on FiO2 > 30% and/or continuous positive airway pressure (CPAP) and/or mechanical ventilation were classified as having severe BPD.
Variables and definitions employed
During the initial phase of developing the model, countless variables considered to be possible risk factors for the development of BPD were included. Percentage birth weight lost was assessed on the seventh day and expressed as a percentage (> 10%, > 15% or > 20%). Time on mechanical ventilation during the first week of life was categorized into number of days (> 2 days, > 3 days, > 4 days or > 5 days). Any day on which a child was ventilated for more than 12 hours was counted as a day on ventilation. Scores on the clinical risk index for babies (CRIB) were split into degrees (degree 1 = 0-5; degree 2 = 6-10; degree 3 = 11-15; degree 4 >15). Persistent ductus arteriosus (PDA) was defined by the presence of clinical signs (systolic murmur, wide pulse and hyperdynamic precordium) and the need for clinical and/or surgical treatment was confirmed using echocardiography. Sepsis was defined as the presence of at least one positive blood culture (confirmed sepsis) or clinical signs indicative of sepsis (suspected sepsis), and classified as either early (up to 72 h) or late (> 72 h).
Development of the predictive model, sample size calculation and statistical analysis
Initially, each possible risk factor was assessed in isolation (n = 66) in order to identify variables that had a significant relationship with BPD. The 36 most significant variables (where p < 0.25) were then selected for the logistic regression model, which was produced using the stepwise forward method with the cutoff for significance set at p < 0.05. Only variables that were capable of providing results before the end of the early neonatal period were tested (n = 25).
Once logistic regression had been performed, four variables maintained a significant relationship with the outcome and were used to construct the formula to calculate the probability of BPD [Probability (%) = (elogit) / (1+(elogit), where logit is the β value for each variable multiplied by the corresponding variable minus the constant (logit = β x variable (0/1) + β x variable (0/1) constant) and e is the base for natural logarithms with a value of 2.71828].
The model was calibrated using the Hosmer & Lemeshow12 test by means of the goodness of fit statistic, with the aim of describing the precision of predicted and observed results as a whole. For this test, patients are grouped by degree of risk and categorized by diagnosis. The program uses the C statistic which selects groups of different degrees of risk that contain approximately the same number of cases. From this, the observed number is compared with the predicted number for each group and differences are summed to calculate the chi-square test p value. In this case, in contrast with usual interpretations, the larger the value of p, the better calibrated the model. Next the model's discriminatory power was evaluated by calculating the area under the receiver operating characteristic (ROC) curve, which identifies the degree of sensitivity and specificity with which the model discriminates between patients who will or will not develop the outcome of interest.
A logistic regression model is considered reliable if the number of patients is 10 to 20 times the number of variables associated with the outcome of interest.13 In our model, containing four significant variables, the size of the BPD patient sample was calculated at between 40 and 80 individuals. Univariate frequency analyses and logistic regression were performed using the statistical package SPSS 11.0 (SPSS Inc., Chicago, IL, USA). The univariate analysis employed the chi-square or Fisher tests for categorical variables and the Mann-Whitney or Student's t tests to compare mean values. Values of p < 0.05 were considered significant.
Results
During the study period, 386 children meeting the inclusion criteria were born. One hundred and twenty-four (124) children were excluded due to death before the 28th day of life (32.1% of the total), 12 due to congenital malformations, two because of congenital infections and one whose mother was HIV-positive. Excluding infants born with malformations, the mortality rate was 29% (n = 112). The final sample, therefore, included 247 children with a mean BW of 1,083±237 g and mean GA of 29.1±2.4 weeks.
Sixty-eight newborns (27.5%) developed BPD, with the following classifications: mild, n = 35 (51.4%); moderate, n = 20 (29.4%); and severe, n = 8 (11.7 %). Five (7.3 %) children could not be classified since, although they were diagnosed with BPD, they died before the classification point. Twenty-eight (11.3 %) were still dependent on oxygen at 36 weeks' corrected GA. Table 1 lists the principal characteristics of the sample during the perinatal and neonatal periods, broken down by whether or not patients developed BPD, together with the significance of the differences between the two sets.
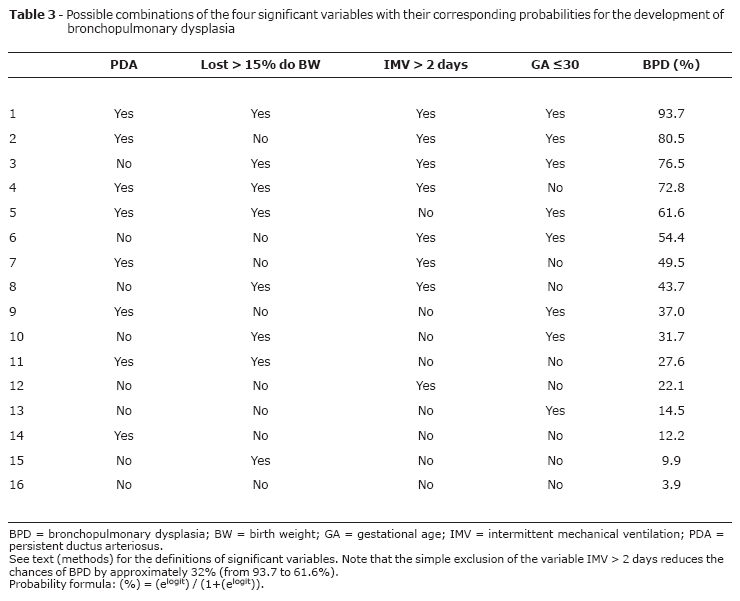
Four of the variables analyzed in the logistic regression maintained their significance with relation to BPD: GA <30 weeks, PDA and mechanical ventilation for > 2 days during the first week of life and loss of > 15% of BW by the seventh day of life (Table 2). The model was calibrated using the following values: C^ test = 8.0067; degrees of freedom = 5 and p = 0.1559. As evaluated by the ROC curve, the model's discriminatory power was excellent (Figure 1A) with an area under the curve of 0.906±0.021 (95%CI 0.864-0.947). Where all four variables were present, the model allowed for a maximum correct identification probability of 93.7% (Table 3).
In order to validate the model, we applied it to 61 newborns who had not participated in the initial sample and who exhibited similar characteristics (with and without BPD): GA (31.6±1.4 and 29.3±2.8 weeks), birth weight (1,229±218 and 926±270 g), male sex (n = 40, 55%), CRIB (2.7±3.3 and 9.8±3.8), PDA (17 and 55%), time on IMV (0.7±9 and 10.1±1.8), % of birth weight lost (11.5±5 and 19.8±5.3%). For this group, calibration values were: C^ test = 7.6957; degrees of freedom = 5 and p = 0.1738. The area under the ROC curve was 0.935±0.0032 (95%CI 0.873-0.997), confirming the sensitivity of the predictive model developed (Figure 1B).
Discussion
In our population, assessed at 7 days, the probability model developed here demonstrated an elevated level of sensitivity for the identification of those newborns with the greatest chance of developing BPD. This early identification is essential to the development of strategies that may act in a preventative manner, reducing the incidence of this disease. Nevertheless, our model exhibits certain differences that should be discussed, particularly in relation to the most significant variables. For the sake of clarity, significant variables and the existing probabilistic models and their relevance are discussed separately.
Significant variables in the model
GA < 30 weeks
In the model, prematurity was one of the four principal risk factors associated with the development of BPD. In other predictive models, which employed similar variables in their multivariate analyses, GA was the most significant independent predictive factor.14 Recently, Henderson-Smart et al.15 applied a logistic regression model to 11,453 children and found a significantly higher chance of BPD from 30 weeks' GA.
Indeed, the most marked characteristic of BPD since the introduction of surfactant and of new mechanical ventilation techniques is the alteration to normal alveolar development, by which the pulmonary maturation process is interrupted.16 Therefore, the more premature an infant is, the greater the chances it has of developing BPD. It is estimated that, from 31 weeks' GA, for each week less, the chances of BPD increase by approximately 2 times.15
Mechanical ventilation for more than 48 h
Ventilatory support is a well-known potential mechanism of pulmonary injury in preterm infants. Nevertheless, the incidence of BPD increases when invasive forms of ventilatory support are employed, such as intermittent mechanical ventilation. Ammari et al.17 report a very much reduced incidence of BPD among infants who were started and maintained on nasal CPAP from the moment of birth, even when extremely premature. This more conservative conduct is currently being evaluated in a multicenter study, the first phase of which has confirmed the viability of this intervention.18
In our study, all of the variables related to invasive ventilatory support exhibited significant differences between subsets, with mechanical ventilation and time on ventilation being the most important (p < 0.001). Those infants who required mechanical ventilation for more than 2 days during the first week of life had a greater risk of developing BPD. Indeed, this was the most expressive variable in the entire model. Its simple removal (with the other three variables present) reduced the percentage chance of developing BPD from 93.7 to 61.6% (Table 3).
Persistent ductus arteriosus
The association between PDA and BPD is well delineated in the literature.19,20 In our study, infants with PDA at the end of the first week of life exhibited an increased risk of developing BPD (OR = 13.5; 95%CI 6.4-28.3). Despite this robust association, prophylactic use of indomethacin or ibuprofen reduced the incidence of PDA without modifying the incidence of BPD.19 Nevertheless, it is important to point out that in none of these studies the principal object was to assess the effects of this treatment on the incidence of BPD. Although some hypotheses have been developed in an attempt to explain these conflicting results,20,21 it is believed that treatment that may be effective at reducing the incidence of BPD should be directed to the abnormal development of immature lungs, and not at other factors associated with prematurity, such as PDA.22
Loss of > 15% of birth weight on seventh day of life
In the post-surfactant era, three studies have assessed a possible correlation between fluids and weight loss and the development of BPD.1,20,23 In these studies, percentage weight loss was similar on the fifth day of life,23 significantly lower between the sixth and ninth days of life1 or significantly lower on the seventh day of life.20 The most important differences between these studies and ours was that, in the first two, infants who developed BPD were given significantly more liquids throughout the evaluation period.1,23 In the third study, prophylactic indomethacin was used and the volume of liquids administered was not provided.20 In our population, the volume of liquids administered was similar in both groups and indomethacin was not used prophylactically. Furthermore, those infants who did develop BPD lost a much greater percentage of birth weight by the end of the first week than in the studies cited (~ 20%).
Calorie intake of the infants who developed BPD was significantly lower on the sixth (p = 0.01) and seventh days (p = 0.03) of life. These children's scant reserves associated with inadequate nutritional support, may have contributed to a state of catabolism and protein deficiency, which has been described as capable of altering both pulmonary growth and healing and control of the inflammatory response, promoting pulmonary injuries.24-26 Furthermore, although just 11% of the total number of patients were treated with diuretics, these were started after the fourth day of life and in a significant manner among the subset of children who would develop BPD.
In conclusion, in a population of infants with GA < 30 weeks receiving similar amounts of fluids throughout the first week of life, the increased use of diuretics from the fourth day on, as well as the reduced calorie intake on the sixth and seventh days, was responsible for the greater percentage loss of birth weight by the end of that first week. This basically reflects the greatest severity of the subset (see CRIB) and the reason for the significant association with the development of BPD.
Predictive models and their relevance
Other probabilistic models have been proposed for BPD. Cohen et al.27 used BW (< 1,250 g) and mechanical ventilation at 48 h and obtained an estimated risk of 61%. In the same study, infants that were on FiO2 > 60% for > 2 h within the first 48 h of life had a risk of 81%. Other authors have used birth weight and ventilatory data to predict the risk of BPD at 12 h to 10 days of life. A sensitivity of 75% was obtained using FiO2 on the 10th day and MAP on the fifth day, in association with birth weight and GA.28 Palta et al.29 obtained a maximum probability of 85% with a logistic model that used the variables: severity score, X ray assessment and BW. In our model, the estimation possibilities varied from 3.9% (all four variables absent) to 93.7% (all variables present), indicating a maximum predictive capacity superior to those of all of the studies published to date.
Our study involves some important limitations, such as the retrospective data collection during the first period. Nevertheless, it is important to point out that the patients whose data were collected retrospectively were the same patients used in another prospective study performed at our institution.3 As a result, the proportion of data that could not be collected was extremely small, having no effect on analysis of the results. Another limitation is the incapacity of the model to detect the degree of BPD that an infant will develop (mild, moderate or severe). While this was not the objective of the study, it is known that infants with mild BPD are breathing room air by 36 weeks' corrected GA, which is evidence of lesser severity, and therefore are of a different clinical relevance when compared with infants with moderate or severe BPD. The capacity to predict severity could be of aid when making critical decisions, such as the use of corticosteroids during the postnatal period. Doyle et al.5 performed a regressive meta-analysis demonstrating that postnatal corticosteroids could be more beneficial than detrimental when given to infants at greatest risk of developing moderate or severe BPD.
Recently, Ballard et al.30 demonstrated that administering nitric oxide to preterms on mechanical ventilation between 7 and 21 days of life reduced the incidence of BPD, an effect only observed when treatment was started between 7 and 14 days of life. One limitation of that study was the fact that the authors had included all preterms on mechanical ventilation without selecting a population at greater risk of developing moderate or severe BPD, which according to the authors themselves could have significantly modified the final results.
Conclusions
BPD is still a common cause of morbidity among ELBW infants, and the risk factors related with this disease are countless. At the end of their first week of life, the predictive model developed in this study was capable of identifying those infants with the greatest probability of developing BPD with a high degree of sensitivity. When the model was prospectively applied to a different population, this sensitivity was confirmed, validating the model. This type of identification makes it possible to intervene in specific populations in a timely manner. Based on our model, reducing the time spent on mechanical ventilation and optimizing fluid and nutrition management could contribute to reducing BPD incidence in our population. Furthermore, the model could also be used to select target populations for further studies of BPD that will be carried out at our institution.
References
Manuscript received Jul 12 2006, accepted for publication Nov 07 2006.
- 1. Oh W, Poindexter BB, Perrit R, Lemons JA, Bauer CR, Ehrenkranz RA, et al. Association between fluid intake and weight loss during the first ten days of life and risk of bronchopulmonary dysplasia in extremely low birth weight infants. J Pediatr. 2005;147:786-90.
- 2. Monte LF, Silva Filho LV, Miyoshi MH, Rozov T. Displasia broncopulmonar. J Pediatr (Rio J). 2005;81:99-110.
- 3. de Mello RR, Dutra MV, Lopes JM. Morbidade respiratória no primeiro ano de vida de prematuros egressos de uma unidade pública de tratamento intensivo neonatal. J Pediatr (Rio J). 2004;80:503-10.
- 4. Schmidt B, Roberts RS, Davis P, Doyle LW, Barrington KJ, Ohlsson A, et al. Caffeine therapy for apnea of prematurity. N Engl J Med. 2006;354:2112-21.
- 5. Doyle LW, Halliday HL, Ehrenkranz RA, Davis PG, Sinclair JC. Impact of postnatal systemic corticosteroids on mortality and cerebral palsy in preterm infants: effect modification by risk of chronic lung disease. Pediatrics. 2005;115:655-61.
- 6. Palta M, Gabbert D, Weinstein MR, Peters ME. Multivariate assessment of traditional risk factors for chronic lung disease in very low birth weight neonates. The Newborn Lung Project. J Pediatr. 1991;119:285-92.
- 7. Ehrenkranz R, Younes N, Fanaroff A, Wright LL, Stevenson DK, Shankaran S, et al. Predicting BPD and oxygen dependence at 36 weeks post-conceptional age. Pediatr Res. 1995;37:330A.
- 8. Rozycki HJ, Narla L. Early versus late identification of infants at high risk of developing moderate to severe bronchopulmonary dysplasia. Pediatr Pulmonol. 1996;21;345-52.
- 9. Romagnoli C, Zecca E, Tortorolo L, Vento G, Tortorolo G. A scoring system to predict the evolution of respiratory distress syndrome into chronic lung disease in preterm infants. Intensive Care Med. 1998;24:476-80.
- 10. Jobe AH, Bancalari E. Bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2001;163:1723-9.
- 11. Ehrenkranz RA, Walsh MC, Vohr BR, Jobe AH, Wright LL, Fanaroff AA, et al. Validation of the National Institute of Health consensus definition of bronchopulmonary dysplasia. Pediatrics. 2005;116:1353-60.
- 12. Lemeshow S, Hosmer DW Jr. A review of goodness of fit statistics for use in the development of logistic regression models. Am J Epidemiol. 1982;115:92-106.
- 13. Harrell FE. Regression modeling strategies with applications to linear models, logistic regression, and survival analysis. New York: Springer; 2001.
- 14. Costeloe K, Hennessy E, Gibson AT, Marlow N, Wilkinson AR. The EPICure study: outcomes to discharge from hospital for infants born at the threshold of viability. Pediatrics. 2000;106:659-71.
- 15. Henderson-Smart DJ, Hutchinson JL, Donoghue DA, Evans NJ, Simpson JM, Wright I, et al. Prenatal predictors of chronic lung disease in very preterm infants. Arch Dis Child Fetal Neonatal Ed. 2006;91:F40-5.
- 16. Bourbon J, Boucherat O, Chailley-Heu B, Delacourt C. Control mechanisms of lung alveolar development and their disorders in bronchopulmonary dysplasia. Pediatr Res. 2005;57:38R-46R.
- 17. Ammari A, Suri M, Milisavljevic V, Sahni R, Bateman D, Sanocka U, et al. Variables associated with the early failure of nasal CPAP in very low birth weight infants. J Pediatr. 2005;147:341-7.
- 18. Finer NN, Carlo WA, Duara S, Fanaroff AA, Donovan EF, Wright LL, et al. Delivery room continuous positive airway pressure/positive end-expiratory pressure in extremely low birth weight infants: a feasibility trial. Pediatrics. 2004;114:651-7.
- 19. Schmidt B, Wright LL, Davis P, Solimano A, Roberts RS; Indomethacin Prophylaxis in Preterms Investigators. Ibuprofen prophylaxis in preterm neonates. Lancet. 2002;360:492.
- 20. Schmidt B, Roberts RS, Fanaroff A, Davis P, Kirpalani HM, Nwaesei C, et al. Indomethacin prophylaxis, patent ductus arteriosus, and the risk of bronchopulmonary dysplasia: further analysis from the Trial of Indomethacin Prophylaxis in Preterms (TIPP). J Pediatr. 2006;148:730-4.
- 21. Bose CL, Laughon M. Treatment to prevent patency of the ductus arterious: beneficial or harmful? J Pediatr. 2006;148:713-4.
- 22. Jobe AH. Indomethicin and BPD. J Pediatr. 2006;148:2.
- 23. Marshall DD, Kotelchuck M, Young TE, Bose CL, Kruyer L, O'Shea TM. Risk factors for chronic lung disease in the surfactant era: a North Carolina population-based study of very low birth weight infants. North Carolina Neonatologists Association. Pediatrics. 1999;104:1345-50.
- 24. Sosenko IR, Frank L. Nutritional influences on lung development and protection against chronic lung disease. Semin Perinatol. 1991;15:462-8.
- 25. Kalenga M, Tschanz SA, Burri PH. Protein deficiency and the growing rat lung. I. Nutritional findings and related lung volumes. Pediatr Res. 1995;37:783-8.
- 26. Mataloun MM, Rebello CM, Mascaretti RS, Dohlnikoff M, Leone CR. Pulmonary responses to nutritional restriction and hyperoxia in premature rabbits. J Pediatr (Rio J). 2006;82:179-85.
- 27. Cohen A, Taeusch HW Jr. Prediction of risk of bronchopulmonary dysplasia. Am J Perinatol. 1983;1:21-2.
- 28. Sinkin RA, Cox C, Phelps DL. Predicting risk of bronchopulmonary dysplasia: selection criteria for clinical trials. Pediatrics. 1990;86:728-36.
- 29. Palta M, Gabbert D, Fryback D, Widjaja I, Peters ME, Farrell P, et al. Development and validation of an index for scoring baseline respiratory disease in the very low birth weight neonate. Severity Index Development and Validation Panels and Newborn Lung Project. Pediatrics. 1990;86:714-21.
- 30. Ballard RA, Truog WE, Martin RJ, Ballard PL, Cnaan A, Merrill JD, et al. Improved outcome with inhaled nitric oxide in preterm infants mechanically ventilated at 7-21 days of age. Pediatr Res. 2006;31:53A.
Publication Dates
-
Publication in this collection
10 May 2007 -
Date of issue
Apr 2007
History
-
Accepted
07 Nov 2006 -
Received
12 July 2006