Abstracts
OBJECTIVE: To assess the effect of phototherapy on serum oxidant and antioxidant status in hyperbilirubinemic full-term newborns. METHOD: Thirty-four full-term infants from 3 to 10 days of age exposed to phototherapy were studied. The serum antioxidant status was assessed by measuring the total antioxidant capacity (TAC) and individual antioxidant components: vitamin C, uric acid, albumin, thiol contents and total bilirubin. The oxidant status was assessed by determining the total oxidant status (TOS), oxidative stress index (OSI) and individual oxidant components: malondialdehyde (MDA), and lipid hydroperoxide levels. RESULTS: Vitamin C, uric acid, total bilirubin and MDA concentration were significantly lower, whereas serum TOS, lipid hydroperoxide and OSI levels were significantly higher after phototherapy (p < 0.05). There were significant positive correlations between serum total bilirubin and MDA (r = 0.434, p = 0.001). CONCLUSIONS: Although the MDA level was reduced after phototherapy, phototherapy has a negative impact on numerous parts of the oxidant/antioxidant defense system in jaundiced full-term newborns, exposing them to potential oxidative stress.
Antioxidants; infant; malondialdehyde; oxidative stress; phototherapy
OBJETIVO: Avaliar o efeito da fototerapia no estado oxidante e antioxidante no soro de recém-nascidos a termo com hiperbilirrubinemia. MÉTODO: Trinta e quatro recém-nascidos a termo com idades entre 3 e 10 dias submetidos a fototerapia foram avaliados. O estado antioxidante do soro foi determinado pela capacidade antioxidante total e por componentes antioxidantes individuais: vitamina C, ácido úrico, albumina, concentração de tiol e bilirrubina total. O estado oxidante foi avaliado através do estado oxidante total, índice de estresse oxidativo e componentes oxidantes individuais: malondialdeído e níveis de hidroperóxido lipídico. RESULTADOS: As concentrações de vitamina C, ácido úrico, bilirrubina total e malondialdeído foram significativamente mais baixas, enquanto que o estado oxidante total, níveis de hidroperóxido lipídico e o índice de estresse oxidativo foram significativamente maiores após a fototerapia (p < 0,05). Houve correlações positivas significativas entre a bilirrubina sérica total e a concentração de malondialdeído (r = 0,434, p = 0,001). CONCLUSÕES: Embora a concentração de malondialdeído tenha diminuído após a fototerapia, esta exerce um efeito negativo sobre as diversas partes do sistema de defesa oxidante/antioxidante em recém-nascidos a termo ictéricos, expondo-os a um possível estresse oxidativo.
Antioxidantes; lactente; malondialdeído; estresse oxidativo; fototerapia
ORIGINAL ARTICLE
Total oxidant/antioxidant status in jaundiced newborns before and after phototherapy
Ali AycicekI; Ozcan ErelII
IMD. Pediatrician, Department of Pediatrics, Children's Hospital at Sanliurfa, Sanliurfa, Turkey
IIMD. Professor, Department of Clinical Biochemistry, Harran University, Medical Faculty, Research Hospital, Sanliurfa, Turkey
Correspondence Correspondence: Ali Aycicek Sanliurfa Cocuk Hastaliklari Hastanesi 63200 Sanliurfa Turkey Tel.:+90 414 313 11 44 Fax:+90 414 313 04 70 Email: ayciceka@hotmail.com; ali_aycicek@yahoo.com.tr
ABSTRACT
OBJECTIVE: To assess the effect of phototherapy on serum oxidant and antioxidant status in hyperbilirubinemic full-term newborns.
METHOD: Thirty-four full-term infants from 3 to 10 days of age exposed to phototherapy were studied. The serum antioxidant status was assessed by measuring the total antioxidant capacity (TAC) and individual antioxidant components: vitamin C, uric acid, albumin, thiol contents and total bilirubin. The oxidant status was assessed by determining the total oxidant status (TOS), oxidative stress index (OSI) and individual oxidant components: malondialdehyde (MDA), and lipid hydroperoxide levels.
RESULTS: Vitamin C, uric acid, total bilirubin and MDA concentration were significantly lower, whereas serum TOS, lipid hydroperoxide and OSI levels were significantly higher after phototherapy (p < 0.05). There were significant positive correlations between serum total bilirubin and MDA (r = 0.434, p = 0.001).
CONCLUSIONS: Although the MDA level was reduced after phototherapy, phototherapy has a negative impact on numerous parts of the oxidant/antioxidant defense system in jaundiced full-term newborns, exposing them to potential oxidative stress.
Keywords: Antioxidants, infant, malondialdehyde, oxidative stress, phototherapy.
Introduction
Phototherapy is the most widely used form of therapy for unconjugated hyperbilirubinemia.1,2 Its noninvasive nature, easy availability, low cost and occurrence of few side effects have initially almost led to the assumption that it is innocuous.3 The possibility that this may not be the case has been raised in several recent publications, which have shown that phototherapy is a photodynamic stress and can induce lipid peroxidation. Increasing appreciation of the causative role of oxidative injury and lipid peroxidation in the development of many severe diseases of the newborn has lent tremendous importance to lipid peroxidation and its possible causes.4
Free radicals and related metabolites have attracted a great deal of attention in recent years.5 They are mainly derived from oxygen and are generated in the body by various endogenous systems, exposure to different physicochemical conditions or pathophysiological states. Free radicals can adversely alter lipids, proteins and DNA, and have been implicated in pulmonary oxygen injury, intraventricular hemorrhage, retinopathy of prematurity, ischemia/reperfusion injury manifested as necrotizing enterocolitis, postasphyxial central nervous system injury, and acute tubular necrosis.6,7 Reactions of bilirubin involving free radicals or toxic oxygen reduction products have been well documented: unconjugated bilirubin scavenges singlet oxygen with high efficiency, reacts with superoxide anions and peroxyl radicals, and serves as a reducing substrate for peroxidases in the presence of hydrogen peroxide or organic hydroperoxides.8,9
We hypothesized that an important factor in the mechanism of oxidative stress in hyperbilirubinemic full-term infants submitted to phototherapy on the first days of life would be increased oxidative stress in relation to antioxidants. This imbalance would be affected by exacerbated oxidative stress, diminished antioxidants, or a combination of both. The aim of this study was to test the validity of this hypothesis by determining the relative roles of oxidative stress and diminished total antioxidant activity.
Methods
Fifty-seven full-term newborn infants from 3 to 10-days of age who were delivered vaginally and admitted to Sanliurfa Children's Hospital because of clinically significant indirect hyperbilirubinemia comprised the subjects of this study. All the infants were being breastfed and had no etiological factor for hyperbilirubinemia. Infants with severe congenital malformation, maternal diabetes, birth asphyxia, sepsis or hemolytic-type hyperbilirubinemia due to blood group (Rh or ABO) incompatibility; those that had received intensive phototherapy; those in which the total serum bilirubin level rose by more than 5 mg/dL per day or was higher than 20 mg/dL within the first 24 hours after birth; and those with signs and symptoms suggestive of serious illness were excluded from the study. Clinically significant indirect hyperbilirubinemia was defined as being present in infants with a serum total bilirubin concentration of more than 13 mg/dL.10 The naked newborn, except for those who wore diapers and eye patches, was placed inside an incubator with a phototherapy system consisting of six white fluorescent tubes (Philips TL 20W/54) 40 cm above. The light energy of the phototherapy unit, measured using a standard photometer (Light Meter VF, Minolta, Japan), was 12-16 µW/cm2/nm. All babies were exposed to continuous phototherapy for 48 h, except while being fed, cleaned and sampled. This period of time was chosen so as to allow sampling simultaneously with routine bilirubin tests, thus avoiding blood sampling solely for the purpose of the study. This study was approved by the local Research Ethics Committee. All parents signed a consent form for the participation of newborns in the study.
Analytical methods
Blood samples were taken from a peripheral vein to determine total bilirubin, direct bilirubin, and antioxidant and oxidant concentrations prior to phototherapy. Second blood samples were taken from 49 infants after 48 h. Blood samples were centrifuged at 1500 x g for 10 minutes within 20 minutes of collection. Serum samples were stored at -80°C and analyzed within 2 months.
Total antioxidant capacity (TAC) levels were measured by Erel's TAC method,11,12 which is based on the bleaching of the characteristic color of a more stable 2,2'-azino-bis (3-ethylbenz-thiazoline-6-sulfonic acid) (ABTS) radical cation by antioxidants. The results were expressed in mmol Trolox equiv./L. Serum thiol (total SH group) content was measured by using dithionitrobenzoic acid (DTNB).13 Vitamin C concentration was measured using the FRASC method.14 Uric acid, albumin and total bilirubin, which are individual serum antioxidants, were measured using commercial kits (Abbott). Total oxidant status (TOS) serum concentrations were measured using Erel's TOS method,15 which is based on the oxidation of ferrous ion to ferric ion in the presence of various oxidative species in acidic medium and the measurement of the ferric ion by xylenol orange. The results were expressed in µmol H2O2/L. Erel's TAC and TOS methods are colorimetric and automated and the precision of this assay is excellent lower than 3%.13,16 Serum malondialdehyde (MDA) levels were measured using a fluorimetric method.17,18 Lipid hydroperoxide serum concentrations were also measured using the automated ferric-xylenol orange method.19 The TOS to TAC ratio was regarded as the oxidative stress index (OSI).20,21 To perform the calculation, the result unit of TAC, mmol Trolox equivalent/L, was changed to µmol Trolox equivalent/L, and the OSI value was calculated as follows: OSI = [(TOS, µmol/L)/(TAC, µmol Trolox equivalent/L) /100].
Statistical analysis
The statistical analysis of the data was carried out with the Statistical Package for the Social Sciences, version 11.0, for Windows (SPSS, Inc). The Student's t test for paired samples was used to compare the blood samples before and after phototherapy, assuming a 95% confidence interval. Bivariate associations between continuous variables were assessed by Pearson's correlation test. P-values less than 0.05 were considered to be statistically significant.
Results
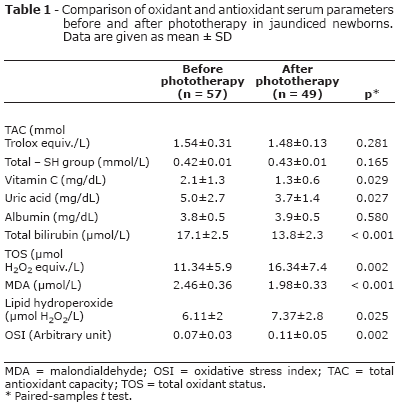
The infants' mean age was 6±3 days, their mean length was 50±3.8 cm, their mean body weight was 3.1±1.4 kg, their mean head circumference ranged from 36.2±1.9 cm, and 29 of them were male, whereas 28 were female. Serum antioxidant/oxidant parameters before and after phototherapy are shown in Table 1. Serum TAC, thiol content and albumin levels were not altered after phototherapy. Serum vitamin C, uric acid, total bilirubin and MDA concentrations were significantly lower after phototherapy than before it (p < 0.05). Conversely, serum TOS, lipid hydroperoxide and OSI levels were significantly higher after phototherapy than before it (p < 0.05). There were significantly positive correlations between serum total bilirubin and MDA (r = 0.434, p = 0.001). Furthermore, there was no correlation between total bilirubin and other parameters.
Discussion
In the present study, vitamin C levels and uric acid, which are well known antioxidants, were significantly lower after phototherapy than before it; in contrast, TOS, lipid hydroperoxide and OSI levels were significantly higher after phototherapy than before it. Interestingly, MDA, which is known as an end product of lipid peroxidation and as an oxidant marker, was decreased after phototherapy. There was also a significant positive correlation between MDA and total bilirubin. All the published studies are related to the oxidative effects of phototherapy, especially lipid peroxide (which is a TBARs), and antioxidant enzyme activities, but not to serum nonenzymatic total antioxidant capacity.22-27 This is the first report showing an association between these serum oxidant/antioxidant parameters in phototherapy-treated full-term nonhemolytic hyperbilirubinemic infants.
In a healthy human being, the formation and inactivation of reactive oxygen species are balanced at a level at which the compounds can play their physiological role without any toxic effects. This balance can be unstable in the neonatal period following rapid changes in tissue oxygen concentration, immature antioxidant mechanism and considerable neonatal developmental changes in antioxidants. This deterioration is especially evident in the presence of oxidative stress such as phototherapy.4
Although phototherapy is now widely used for the treatment of neonatal hyperbilirubinemia, there are concerns regarding the possibility of photodynamic tissue damage.28 The exposure of infants to phototherapy in the presence of a sensitizer (bilirubin) resulted in oxidative injury to the red cell membrane manifested by a significant increase in concentrations of products of lipid peroxidation in the membrane and in hemolysis.29 Another study reported that phototherapy in the presence of bilirubin induced a dramatic decrease in ATPase activity and an increased susceptibility to lipid peroxidation in neonatal erythrocytes.4 Our study showed that phototherapy remarkably increased serum lipid hydroperoxides and TOS in jaundiced newborns.
It was shown that plasma MDA concentrations in neonates with nonhemolytic jaundice were significantly higher than those in healthy infants.22,23 Ozture et al. reported that MDA concentrations were significantly lower after phototherapy than before it, and no significant correlation was found between plasma MDA and plasma bilirubin concentrations before and after phototherapy.23 Yigit et al. reported that there was no significant correlation between MDA and bilirubin levels.22 They also reported that both bilirubin and MDA were increased in jaundiced infants, and both parameters were reduced after phototherapy, but they did not find a significant correlation between the opposite oxidant/antioxidants parameters. The results of our study confirmed those findings, but we also investigated serum TOS, lipid hydroperoxide and OSI levels. In our study, serum MDA concentrations significantly decreased after phototherapy, and a significant correlation was found between plasma MDA and bilirubin concentrations before and after phototherapy (r = 0.434, p = 0.001). These results suggest that oxidative stress was not caused by phototherapy. However, TOS, lipid hydroperoxide and OSI levels were significantly increased after phototherapy. The apparent cause of these conflicting data is the fact that MDA measurement is not a specific method for lipid peroxidation, and that it is positively affected by bilirubin and some aldehyde structures.15 MDA is an end product of the lipid peroxidation process, and lipid hydroperoxide, which is our lipid peroxidation parameter, is an early marker of the oxidation chain of lipids, and in the latter method, there is no interference with other structures.
Previous studies examined the effect of phototherapy on erythrocyte antioxidant enzyme activity (glutathione peroxidase, superoxide dismutase, etc.) in jaundiced infants. We investigated serum nonenzymatic total antioxidant capacity and individual antioxidants. Bohles et al. reported a significant decrease in serum uric acid during phototherapy.30 The decrease in serum uric acid concentration is discussed as an effect of direct photodecomposition on the one hand, and as an inhibitory effect of riboflavin deficiency on uric acid formation on the other hand. We found that TAC levels were not significantly altered by phototherapy, but vitamin C and uric acid concentrations were significantly reduced. However, TAC levels were not reduced, and the oxidant/antioxidant balance shifted significantly to the oxidant side, because other indicators of oxidative status, i.e. lipid hydroperoxides, TOS and OSI levels, were significantly increased in jaundiced newborns exposed to phototherapy.
In conclusion, phototherapy has a negative impact on numerous parts of the oxidant/antioxidant defense system in newborn hyperbilirubinemic infants, exposing them to potent oxidative stress.
References
Manuscript received May 08 2006, accepted for publication March 12 2007.
- 1. Porter ML, Dennis BL. Hyperbilirubinemia in the term newborn. Am Fam Physician. 2002;65:599606.
- 2. Tan KL. Phototherapy for neonatal jaundice. Acta Paediatr. 1996;85:2779.
- 3. Tan KL. Phototherapy for neonatal jaundice. Clin Perinatol. 1991;18:42339.
- 4. Gathwala G, Sharma S. Oxidative stress, phototherapy and the neonate. Indian J Pediatr. 2000;67:8058.
- 5. Devasagayam TP, Tilak JC, Boloor KK, Sane KS, Ghaskadbi SS, Lele RD. Free radicals and antioxidants in human health: current status and future prospects. J Assoc Physicians India. 2004;52:794804.
- 6. Warner BB, Wispe JR. Free radicalmediated diseases in pediatrics. Semin Perinatol. 1992;16:4757.
- 7. Halliwell B. Free radicals, antioxidants, and human disease: curiosity, cause, or consequence. Lancet. 1994;344:7214.
- 8. Stocker R, Ames BN. Potential role of conjugated bilirubin and copper in the metabolism of lipid peroxides in bile. Proc Natl Acad Sci USA. 1987;84:81304.
- 9. Stocker R, Glazer AN, Ames BN. Antioxidant activity of albuminbound bilirubin. Proc Natl Acad Sci USA. 1987;84:591822.
- 10. Newman TB, Maisels MJ. Evaluation and treatment of jaundice in the term newborn: a kinder, gentler approach. Pediatrics. 1992;89(5 Pt 1):80918.
- 11. Erel O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin Biochem. 2004;37:27785.
- 12. Erel O. A novel automated method to measure total antioxidant response against potent free radical reactions. Clin Biochem. 2004;37:1129.
- 13. Hu ML, Louie S, Cross CE, Motchnik P, Halliwell B. Antioxidant protection against hydrochlorous acid in human plasma. J Lab Clin Med. 1993;121:25762.
- 14. Chung WY, Chung JK, Szeto YT, Tomlinson B, Benzie IF. Plasma ascorbic acid: measurement, stability and clinical utility revisited. Clin Biochem. 2001;34:6237.
- 15. Erel O. A new automated colorimetric method for measuring total oxidant status. Clin Biochem. 2005;38:110311.
- 16. Ahn MR, Kumazawa S, Hamasaka T, Bang KS, Nakayama T. Antioxidant activity and constituents of propolis collected in various areas of Korea. J Agric Food Chem. 2004;52:728692.
- 17. Kamal AA, Gomaa A, el Khafif M, Hammad AS. Plasma lipid peroxides among workers exposed to silica or asbestos dusts. Environ Res. 1989;49:17380.
- 18. Erel O, Kocyigit A, Avci S, Aktepe N, Bulut V. Oxidative stress and antioxidative status of plasma and erythrocytes in patients with vivax malaria. Clin Biochem. 1997;30:6319.
- 19. Arab K, Steghens JP. Plasma lipid hydroperoxides measurement by an automated xylenol orange method. Anal Biochem. 2004;325:15863.
- 20. Harma M, Harma M, Erel O. Oxidative stress in women with preeclampsia. Am J Obstet Gynecol. 2005;192:6567.
- 21. Aycicek A, Erel O, Kocyigit A. Decreased total antioxidant capacity and increased oxidative stress in passive smoker infants and their mothers. Pediatr Int. 2005;47:6359.
- 22. Yigit S, Yurdakok M, Kilic K, Oran O, Erdem G, Tekinalp G. Serum malondialdehyde concentration in babies with hyperbilirubinaemia. Arch Dis Child Fetal Neonatal Ed. 1999;80:F2357.
- 23. Ozture H, Duman M, Duman N, Ozkan H. How phototherapy affects the relation between serum bilirubin and plasma malondialdehyde in neonates. Arch Dis Child Fetal Neonatal Ed. 2000;82:F171.
- 24. Turgut M, Basaran O, Cekmen M, Karatas F, Kurt A, Aygun AD. Oxidant and antioxidant levels in preterm newborns with idiopathic hyperbilirubinaemia. J Paediatr Child Health. 2004;40:6337.
- 25. Akisu M, Coker C, Tuzun S, Yilmaz D, Kultursay N. Serum malondialdehyde levels in preterm and fullterm infants undergoing phototherapy. Acta Paediatr. 1998;87:6056.
- 26. Akisu M, Yilmaz D, Tuzun S, Kultursay N. Antioxidant defense systems in newborns undergoing phototherapy. Indian J Pediatr. 1999;66:6515.
- 27. Ostrea EM Jr., Cepeda EE, Fleury CA, Balun JE. Red cell membrane lipid peroxidation and hemolysis secondary to phototherapy. Acta Paediatr Scand. 1985;74:37881.
- 28. McDonagh AF. The role of singlet oxygen in bilirubin photooxidation. Biochem Biophys Res Commun. 1971;44:130611.
- 29. Vreman HJ, Wong RJ, Stevenson DK. Phototherapy: current methods and future directions. Semin Perinatol. 2004;28:32633.
- 30. Bohles H, Schnall B. [The effect of phototherapy on serum uric acid (author"s transl)]. Klin Padiatr 1981;193:30810.
Publication Dates
-
Publication in this collection
27 Aug 2007 -
Date of issue
Aug 2007
History
-
Received
08 May 2006 -
Accepted
12 Mar 2007