LETTERS TO THE EDITOR
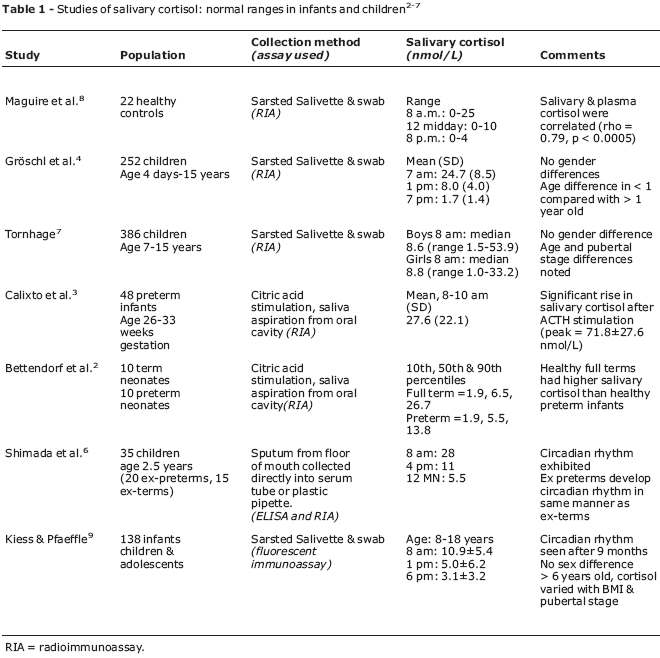
In the recent publication by Silva et al.,1 a mean morning cortisol level of 558 nmol/L (range 77-1,620 nmol/L) in children was reported. This is remarkably higher than any of the previously published ranges for salivary cortisol in children. Although Silva et al. did not report the corresponding serum cortisol levels, their data contradict the notion that only the free cortisol component can pass into saliva, and therefore, under basal conditions, salivary cortisol equates to < 10% of total serum cortisol. Most institutions report morning cortisol levels of a much smaller magnitude, with mean morning salivary cortisol levels of < 30 nmol/L (Table 1).
The authors state that they were unable to locate reference salivary cortisol levels for children in the literature. For readers' information, we have included a summary of the previously published pediatric salivary cortisol literature (Table 1). Our group recently published salivary cortisol reference ranges for healthy children.8 The range for morning cortisol was 0-25 nmol/L.8
Silva et al. used an in-house cortisol radioimmunoassay (RIA), using antibodies to cortisol-3-oxime conjugated with bovine albumin, quoting cross reactivity of 8.5% for cortisone and 7.9% for 11-deoxycortisol. These cross reactivities are higher than in commercially available RIAs. For example, the Orion Diagnostica Spectra Cortisol Coated Tube RIA product information quotes cross reactivity of 0.9% for cortisone and 0.3% for 11-deoxycortisol. However, the increased cross reactivity for cortisone and 11-deoxycortisol cannot explain why the results of Silva et al. are so discrepant from previously published data. This highlights the importance of establishing reference values for all methods and at each institution, as mentioned in the Editorial in the same edition of the journal.9 However, to avoid misleading clinicians, particularly those who are unfamiliar with salivary cortisol, the authors could have elaborated on this in their discussion. Would the authors care to comment on the degree of discrepancy between their reported salivary cortisol levels and those previously reported and summarized in Table 1?
References
1. Silva ML, Mallozi MC, Ferrari GF. Salivary cortisol to assess the hypothalamic-pituitary-adrenal axis in healthy children under 3 years old. J Pediatr (Rio J). 2007;83:121-6.
2. Bettendorf M, Albers N, Bauer J, Heinrich UE, Linderkamp O, Maser-Gluth C. Longitudinal evaluation of salivary cortisol levels in full-term and preterm neonates. Horm Res. 1998;50:303-8.
3. Calixto C, Martinez FE, Jorge SM, Moreira AC, Martinelli CE. Correlation between plasma and salivary cortisol levels in preterm infants. J Pediatr. 2002;140:116-8.
4. Gröschl M, Rauh M, Dörr HG. Circadian rhythm of salivary cortisol, 17alpha-hydroxyprogesterone, and progesterone in healthy children. Clin Chem. 2003;49:1688-91.
5. Kiess W, Meidert A, Dressendorfer RA, Schriever K, Kessler U, Konig A, et al. Salivary cortisol levels throughout childhood and adolescence: relation with age, pubertal stage, and weight. Pediatr Res. 1995;37(4 Pt 1):502-6.
6. Shimada M, Takahashi K, Ohkawa T, Segawa M, Higurashi M. Determination of salivary cortisol by ELISA and its application to the assessment of the circadian rhythm in children. Horm Res. 1995;44:213-7.
7. Tornhage CJ. Reference values for morning salivary cortisol concentrations in healthy school-aged children. J Pediatr Endocrinol Metab. 2003;15:197-204.
8. Maguire AM, Ambler GR, Moore B, Waite K, McLean M, Cowell CT. The clinical utility of alternative, less invasive sampling techniques in the assessment of oral hydrocortisone therapy in children and adolescents with hypopituitarism. Eur J Endocrinol. 2007;156:471-6.
9. Kiess W, Pfaeffle R. Steroid analysis in saliva: a noninvasive tool for pediatric research and clinical practice. J Pediatr (Rio J). 2007;83:97-9.
Ann M. Maguire
Christopher T. Cowell
Institute of Endocrinology and Diabetes, The Children's Hospital at Westmead, Sydney, Australia. Discipline of Paediatrics and Child Health, University of Sydney, Sydney, Australia
Authors' reply
The quantitation of salivary cortisol requires adrenal assessment, which can be performed in a practical and unstressful way, thus being suitable for use in pediatric patients.
Salivary cortisol levels can be determined by various highly sensitive and specific methods. In our study, we used the radioimmunoassay (RIA) method without extraction.1 In this method, after the centrifugation of the saliva samples, the rabbit antibody to cortisol-3-oxime conjugated with bovine albumin produced by Prof. Dr. José Gilberto Vieira (Fleury laboratory) was used, having yielded a sensitivity of up to 5 ng/dL2. The analysis was run in duplicate and the efficacy of this method was based on a coefficient of variation below 12 to 15%.2
Age plays an important role in the determination of salivary cortisol levels, and several studies have shown discrepant results in this respect.3,4
The commitment of authors towards establishing reference values for salivary cortisol levels in healthy children aged less than 3 years arose from the necessity to fill the gap for studies with an adequate number of samples performed on a restricted age range and that require careful sampling.
In this regard, we read with interest the letter from Dr. Ann M. Maguire and Prof. Christopher T. Cowel from The Children's Hospital at Westmead, Sydney and the University of Sydney, Australia, in which they address some aspects related to our paper entitled "Salivary cortisol to assess the hypothalamic-pituitary-adrenal axis in healthy children under 3 years old." Their comments give us the opportunity to broaden the discussion and also clarify some issues that could lead to misinterpretations.
Among the studies mentioned in Table 1 in the letter to the editor written by Dr. Maguire and by Prof. Cowell, we turn our attention to the study by Gröschl et al.,5 in which 252 healthy and well-nourished children were assessed. By analyzing the sample size related to each of the age ranges (< 1 month, n = 13; 1 to 12 months, n = 17; 1 to 2 years, n = 10; 2 to 15 years, n = 212), we obtained a less-than-80% power among infants, with an alpha error greater than 0.05. In another study, by Kiess et al.,3 of 138 patients, 10 were aged between 0 and 1 year and 17 were aged between 1 and 4 years.
In our study, for a variance of 95% and error of 1%, the sample size was calculated to be 71, which is a smaller number than that which was used (n = 91).
With regard to the list of studies on salivary cortisol mentioned by Dr. Maguire and by Prof. Cowell, we would like to add studies that used a similar method to ours, such as Fogaça et al.,6 who assessed 11 children aged 4 to 6 months, and Few et al.7 In the latter study, the authors assessed 106 children aged 1 to 12 months, with cortisol measurements at the following times of the day: morning = 9:30 to 12 hrs and afternoon = 14 to 16 hrs. These intervals are so close that they certainly hamper the analysis of the circadian rhythm. Another important aspect regarding the time of measurement refers to possible differences in morning and afternoon salivary cortisol levels between infants aged less than 1 year and children aged over 1 year.3,5 Thus, the study by Törnhage,8 which only took into consideration the morning cortisol measurement, does not allow assessing any differences across age ranges.
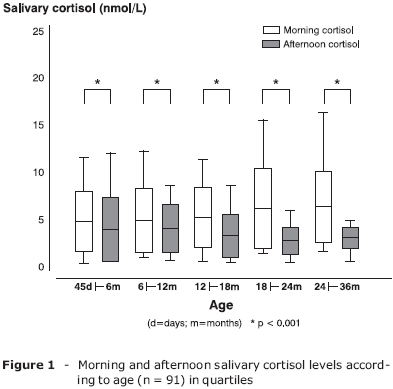
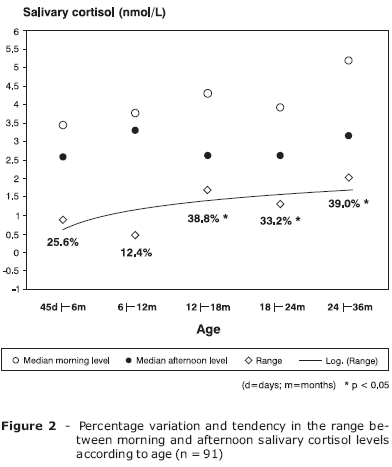
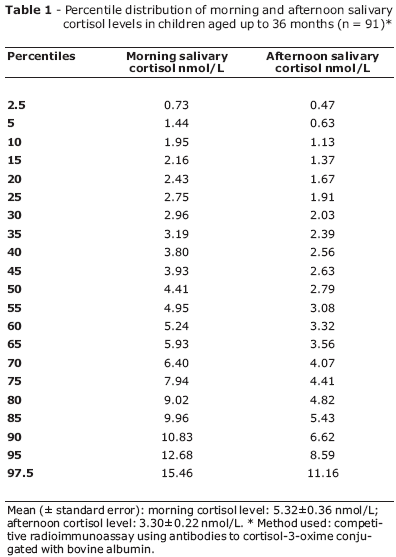
The discrepancy between the values obtained in our study and those mentioned by Dr. Maguire and Prof. Cowell was revised. The results were issued by Fleury Laboratory in ng/dL, and the conversion to nmol/L was based on the value recommended by the Reference Manual for Exams of the Radioimmunoassay Center of São Paulo.9 We requested that the laboratory provide an explanation, and they informed us that the conversion value stipulated in the manual is incorrect.10 The measurements were recalculated, and the mean values (±standard error) expected for the morning levels were 5.32±0.36 nmol/L, with a range of 0.73 to 15.46 nmol/L (2.5th to 97.5th percentiles), and 3.30±0.22 nmol/L, with a range of 0.47 to 11.16 nmol/L (2.5th to 97.5th percentiles) for the afternoon levels.
The corrected table and Figures 1 and 2, including the percentile values, appear below.
References
1. Vieira JGH, Noguti KO, Hidal JT, Russo EMK, Maciel RMB. Ensaio do cortisol na saliva como um método para avaliação da fração livre sérica. Arq Bras Endocrinol Metab. 1984;28:8-10.
2. Hanrahan K, McCarthy AM, Kleiber C, Lutgendorf S, Tsalikian E. Strategies for salivary cortisol collection and analysis in research with children. Appl Nurs Res. 2006;19:95-101.
3. Kiess W, Meidert A, Dressendörfer RA, Schriever K, Kessler U, König A, et al. Salivary cortisol levels throughout childhood and adolescence: relation with age, pubertal stage, and weight. Pediatr Res. 1995;37(4 Pt 1):502-6.
4. Törnhage CJ, Alfvén G. Diurnal salivary cortisol concentrations in school-age children: increased morning cortisol concentration and total cortisol concentration negatively correlated to body mass index in children with recurrent abdominal pain of psychosomatic origin. J Pediatr Endocrinol Metab. 2006;19:843-54.
5. Gröschl M, Rauh M, Dorr HG. Circadian rhythm of salivary cortisol, 17 alpha-hydroxyprogesterone, and progesterone in healthy children. Clin Chem. 2003;49:1688-91.
6. Fogaça MC, Carvalho WB, Peres CA, Lora MI, Hayashi LF, Verreschi ITN. Cortisol salivar como indicador da função adrenal em lactentes sadios com massagem terapêutica. Sao Paulo Med J. 2005;123:215-8.
7. Few JD, Mangat TK, Oppe TE, James VH. Saliva aldosterone concentration in healthy infants. Arch Dis Child. 1986;61:508-9.
8. Törnhage CJ. Reference values for morning salivary cortisol concentrations in healthy school-aged children. J Pediatr Endocrinol Metab. 2002;15:197-204.
9. Di Dio R, Barbério JC, Pradal MG, Menezes MA. Procedimentos hormonais: Central de Radioimunoensaio de São Paulo. 3ª ed. São Paulo: CPD CRIESP; 1995.
10. Olesen H, Ibsen I, Bruunshuus I, Kenny D, Dybkaer R, Fuentes-Arderiu X, et al. Properties and Units in the Clinical Laboratory Sciences. Part XII. Properties and Units in General Clinical Chemistry. (IUPAC Technical Report). Pure Appl. Chem. 2000;72:747-972.
Margarete L. Silva
Mestre, Universidade Estadual Paulista (UNESP), Botucatu, SP, Brasil. Auxiliar de ensino, Disciplina de Pediatria, Faculdade de Medicina do ABC, Santo André, SP, Brasil
Márcia C. Mallozi
Universidade Federal de São Paulo Escola Paulista de Medicina (UNIFESP-EPM), São Paulo, SP, Brasil. Auxiliar de ensino, Disciplina de Pediatria, Faculdade de Medicina do ABC, Santo André, SP, Brasil
Giesela F. Ferrari
Doutora. Professora assistente, Disciplina de Pneumologia Pediátrica, Departamento de Pediatria, Faculdade de Medicina de Botucatu, UNESP, Botucatu, SP, Brasil
Salivary cortisol to assess the hypothalamic-pituitary-adrenal axis in healthy children under 3 years old
Publication Dates
-
Publication in this collection
27 Aug 2007 -
Date of issue
Aug 2007