Abstracts
OBJECTIVE: Our aim was to determine whether endogenous mononuclear leukocyte DNA strand is a target of phototherapy. METHODS: The study included 65 term infants aged between 3-10 days that had been exposed to intensive (n = 23) or conventional (n = 23) phototherapy for at least 48 hours due to neonatal jaundice, and a control group (n = 19). DNA damage was assayed by single-cell alkaline gel electrophoresis (comet assay). Plasma total antioxidant capacity and total oxidant status levels were also measured, and correlation between DNA damage and oxidative stress was investigated. RESULTS: Mean values of DNA damage scores in both the intensive and conventional phototherapy groups were significantly higher than those in the control group (p < 0.001). Mean values and standard deviation were 32 (9), 28 (9), 21 (7) arbitrary unit, respectively. Total oxidant status levels in both the intensive and conventional phototherapy groups were significantly higher than those in the control group (p = 0.005). Mean (standard deviation) values were 18.1 (4.2), 16.9 (4.4), 13.5 (4.2) µmol H2O2 equivalent/L, respectively. Similarly, oxidative stress index levels in both the intensive and conventional phototherapy groups were significantly higher than those in the control group (p = 0.041). Plasma total antioxidant capacity and total bilirubin levels did not differ between the groups (p > 0.05). There were no significant correlations between DNA damage scores and bilirubin, total oxidant status and oxidative stress levels in either phototherapy group (p > 0.05). CONCLUSIONS: Both conventional phototherapy and intensive phototherapy cause endogenous mononuclear leukocyte DNA damage in jaundiced term infants.
DNA damage; comet assay; hyperbilirubinemia; oxidative stress; phototherapy
OBJETIVO: Determinar se a fita de DNA de leucócitos mononucleares endógenos é alvo de fototerapia. MÉTODOS: O estudo incluiu 65 recém-nascidos a termo com idades entre 3 e 10 dias que haviam sido expostos a fototerapia intensiva (n = 23) ou convencional (n = 23) por pelo menos 48 horas devido à icterícia neonatal, além de um grupo controle (n = 19). Dano ao DNA foi avaliado por eletroforese alcalina em gel de célula única (ensaio cometa). A capacidade antioxidante total plasmática e os níveis de estado oxidativo total também foram medidos, e a correlação entre danos ao DNA e estresse oxidativo foi investigada. RESULTADOS: Os valores médios de escores de danos ao DNA nos grupos de fototerapia intensiva e convencional foram significativamente maiores do que os do grupo controle (p < 0,001). Os valores médios e desvio padrão foram 32 (9), 28 (9), 21 (7) unidades arbitrárias, respectivamente. Os níveis de estado oxidativo nos grupos de fototerapia intensiva e convencional foram significativamente maiores do que os do grupo controle (p < 0,005). Os valores médios (desvio padrão) foram 18, 1 (4,2), 16.9 (4,4), 13,5 (4,2) µmol H2O2 equivalente/L, respectivamente. De maneira semelhante, os níveis de estresse oxidativo nos grupos de fototerapia intensiva e convencional foram significativamente maiores do que os do grupo controle (p = 0,041). A capacidade antioxidante total plasmática e os níveis de bilirrubina total não diferiram entre os grupos (p > 0,05). Não houve correlações significativas entre escores de danos ao DNA e bilirrubina, estado oxidante total e níveis de estresse oxidativo entre os grupos de fototerapia (p > 0,05). CONCLUSÕES: Tanto a fototerapia intensiva quanto a convencional causam danos ao DNA dos leucócitos mononucleares endógenos em recém-nascidos a termo com icterícia.
Danos ao DNA; ensaio cometa; hiperbilirrubinemia; estresse oxidativo; fototerapia
ORIGINAL ARTICLE
Phototherapy causes DNA damage in peripheral mononuclear leukocytes in term infants
Ali AycicekI; Abdurrahim KocyigitII; Ozcan ErelII; Hakan SenturkIII
IMD. Pediatrics Department, Medical Faculty, Harran University, Sanliurfa, Turkey
IIMD. Clinical Biochemistry Department, Medical Faculty, Harran University, Sanliurfa, Turkey
IIIMD. Pediatrics Department, Sanliurfa Children's Hospital, Sanliurfa, Turkey
Correspondence Correspondence: Ali Aycicek Pediatrics Department, Medical Faculty, Harran University 63300 - Sanliurfa - Turkey Tel.: +90 (414) 314.8410 Fax: +90 (414) 313.9615 Email: ayciceka@hotmail.com, ali_aycicek@yahoo.com.tr
ABSTRACT
OBJECTIVE: Our aim was to determine whether endogenous mononuclear leukocyte DNA strand is a target of phototherapy.
METHODS: The study included 65 term infants aged between 3-10 days that had been exposed to intensive (n = 23) or conventional (n = 23) phototherapy for at least 48 hours due to neonatal jaundice, and a control group (n = 19). DNA damage was assayed by single-cell alkaline gel electrophoresis (comet assay). Plasma total antioxidant capacity and total oxidant status levels were also measured, and correlation between DNA damage and oxidative stress was investigated.
RESULTS: Mean values of DNA damage scores in both the intensive and conventional phototherapy groups were significantly higher than those in the control group (p < 0.001). Mean values and standard deviation were 32 (9), 28 (9), 21 (7) arbitrary unit, respectively. Total oxidant status levels in both the intensive and conventional phototherapy groups were significantly higher than those in the control group (p = 0.005). Mean (standard deviation) values were 18.1 (4.2), 16.9 (4.4), 13.5 (4.2) µmol H2O2 equivalent/L, respectively. Similarly, oxidative stress index levels in both the intensive and conventional phototherapy groups were significantly higher than those in the control group (p = 0.041). Plasma total antioxidant capacity and total bilirubin levels did not differ between the groups (p > 0.05). There were no significant correlations between DNA damage scores and bilirubin, total oxidant status and oxidative stress levels in either phototherapy group (p > 0.05).
CONCLUSIONS: Both conventional phototherapy and intensive phototherapy cause endogenous mononuclear leukocyte DNA damage in jaundiced term infants.
Keywords: DNA damage, comet assay, hyperbilirubinemia, oxidative stress, phototherapy.
Introduction
Phototherapy is the most widely used form of therapy for newborn infants with hyperbilirubinemia in order to decrease the body burden of neurotoxic bilirubin.1-3 Wide clinical experience suggests that long-term adverse biological effects of phototherapy are absent, minimal, or unrecognized. However, those using phototherapy should remain alert to these possibilities and avoid any unnecessary use because untoward effects on DNA have been demonstrated in vitro.4
Peroxidases can also catalytically generate nitrogen dioxide (NO2) using H2O2 and nitrite as substrates.5 Excess amounts of reactive oxygen and nitrogen species (ROS and RNS) can cause injury to host cells and may induce DNA strand breaks. Accumulation of DNA damage with time can lead to gene modifications in cells that may be mutagenic or carcinogenic.6 As yet, although there are some studies on oxidative status in phototherapy treated infants,7-10 there is no report available on DNA damage in these patients. We previously reported that phototherapy has a negative impact on numerous parts of the oxidant/antioxidant defense system in newborn hyperbilirubinemic infants and exposes them to potent oxidative stress.11 In this study, we measured endogenous mononuclear leukocyte DNA strand breaks, and investigated the correlation between DNA damage score and plasma total antioxidant capacity (TAC), total oxidant status (TOS), and oxidative stress index (OSI) in jaundiced term infants treated with conventional and intensive phototherapy.
Methods
Subjects
Sixty-five term newborn (38-41 weeks) 3- to 10-day-old infants who were delivered vaginally and admitted to Sanliurfa Children's Hospital because of clinically significant indirect hyperbilirubinemia comprised the subjects of this study. All the infants were being breastfed and had no etiological factor for hyperbilirubinemia. Infants with severe congenital malformation, prematurity or postmaturity, maternal diabetes, birth asphyxia, sepsis or hemolytic-type hyperbilirubinemia due to blood group (Rh or ABO) incompatibility, those that had received conventional or intensive phototherapy before blood drawing, those in which within the first 24 hours of birth the total plasma bilirubin level had risen by more than 5 mg/dL per day or was higher than 24 mg/dL; and those with signs and symptoms suggestive of serious illness were excluded from the study. Clinically significant indirect hyperbilirubinemia was defined as being present in infants with a plasma total bilirubin concentration of more than 13 mg/dL.12
Conventional phototherapy systems consisted of six white fluorescent tubes (Philips TL 52/20W) placed 40 cm above the infant. Intensive phototherapy systems consisted of 12 white fluorescent tubes (Philips TL03) placed within 20 cm under and above the infant's front and back. The infants were placed naked, except for a diaper and eye patches, in an incubator or cradle, or intensive phototherapy unit (Bilicrystal, Medestime). The light energy of the phototherapy units was measured using a standard photometer (Light Meter VF, Minolta, Japan), conventional phototherapy units were 12-16 µW/cm2/nm, and intensive phototherapy units were 30-34 µW/cm2/nm. For bilirubin levels greater than 22 mg/dL, intensive phototherapy was applied; otherwise, lower than 22 mg/dL, randomized conventional or intensive phototherapy was applied. Intensive and conventional phototherapy were exposed to continuous at least for 48 hours (maximum 72 hours, intensive mean = 58 hours, conventional = 61 hours), except during feeding, cleaning, and sampling. The control group comprised 19 infants whose plasma total bilirubin concentrations were 12.9-19.1 mg/dL but who did not receive phototherapy (before phototherapy). DNA damage score and serum total antioxidants/oxidants levels were measured in samples drawn from hyperbilirubinemic infants after treated with phototherapy (intensive or conventional groups) and before phototherapy (controls). Maximum 2.5 mL blood samples were taken per infant for all parameters. This time was chosen to allow sampling simultaneous routine bilirubin tests, thus avoiding a blood-taking procedure solely for the purpose of the study. Controls were not included in phototherapy groups. All babies were breast-fed. Local ethics committee approved this study. The parents gave their consent for the newborns' involvement in the study.
Sample preparation
Blood samples were collected from a peripheral vein into heparinized tubes, stored at 10 ºC in the dark to prevent further DNA damage, and were processed within 2 hours. Mononuclear leukocyte isolation for the comet assay was performed by use of Histopaque 1077 (Sigma); 1 mL of heparinized whole blood was carefully layered over 1 mL of Histopaque and centrifuged for 35 min at 500 x g and 25 ºC. The interface band containing mononuclear leukocytes was washed with phosphate-buffered saline (PBS) and then collected after 15 min centrifugation at 400 x g. The resulting pellets were resuspended in PBS and the cells were counted in a Neubauer chamber. Membrane integrity was assessed by means of the trypan blue exclusion method. The remaining blood was centrifuged at 1,500 x g for 10 min to obtain the plasma. The separated plasma was divided into two parts, one of which was used to measure total and direct bilirubin, while the other one was stored at -80 ºC until further analysis of TAC and TOS.
DNA damage determination by alkaline comet assay
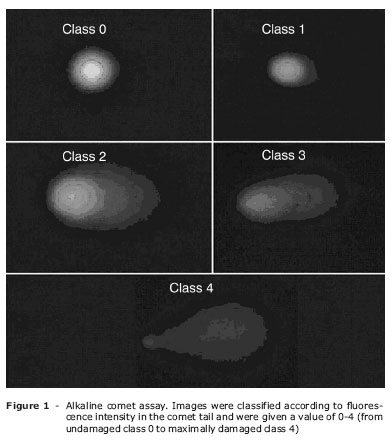
Endogenous DNA damage in peripheral mononuclear leukocytes was analyzed by alkaline comet assay according to Singh et al.13 with minor modification.14 All of the analysis steps were conducted under red light or without direct light to prevent additional DNA damage. The images of 100 randomly chosen nuclei (50 nuclei from each of two replicate slides) were analyzed visually for each subject. Each image was classified according to the intensity of the fluorescence in the comet tail and was given a value of 0, 1, 2, 3, or 4 (from undamaged, class 0, to maximally damaged, class 4) (Figure 1), so that the total score of a slide could be between 0 and 400 arbitrary units (AU). Photomicrographs of representative samples were depicted in Figures 2 and 3. The same biochemistry staff completed all procedures and a single observer unaware of the subject's group determined DNA damage score.
TAC and TOS levels were measured by Erel's methods.15,16 The percentage of TOS level to TAC level was regarded as the OSI.17,18 To perform the calculation, the result unit of TAC, mmol Trolox equivalents per liter, was changed to µmol Trolox equivalent per liter, and the OSI value was calculated as follows: OSI = [(TOS, µmol per liter/(TAC, µmol Trolox equivalent per liter)/100].
Statistical analysis
Testing for normality of variances was accomplished using Levene statistical tests. Variances in this assay were homogeneous. The data were compared using one-way analysis of variance with Tukey's honestly significant difference multiple comparison test. Sex ratio was compared using a chi-square test. Bivariate associations between variables were assessed by Pearson's correlation test. The data were expressed as mean + standard deviation and differences were considered statistically significant at p < 0.05. Statistical analysis was conducted using SPSS for Windows Release 11.5 (SPSS, Chicago, IL, USA).
Results
Intensive phototherapy group's mean age was 7+3 days, mean height was 50+3.2 cm, mean body weight was 3.1+1.6 kg, mean duration of phototherapy was hours 54+6, and sex ratio was 12/11 (M/F). Conventional phototherapy group's mean age was 7+4 days, mean height was 50+3.6 cm, mean body weight was 3.2+1.2 kg, mean duration of phototherapy was hours 61+10, and sex ratio was 13/10 (M/F). Control group's mean age was 5+2 days, mean height was 50+2.9 cm, mean body weight was 3+1.1 kg, and sex ratio was 10/9 (M/F). We found no significant differences between the three groups in terms of age, length, weight, duration of phototherapy (between intensive and conventional), gestational age (all groups were 39+1) or male/female distribution (p > 0.05).
DNA damage scores, and TAC, TOS, OSI, and total bilirubin levels are shown in Table 1. DNA damage scores were 32+9 in the intensive group, 28+9 in the conventional group, and 21+7 in the control group (p < 0.001). TOS and OSI levels were significantly higher in the intensive and conventional groups than they were in the control group (p < 0.05). DNA damage scores and TOS and OSI levels did not differ significantly between the conventional and intensive phototherapy groups. Plasma TAC and total bilirubin levels did not differ between the groups (p >0.05). There were no significant correlations between DNA damage scores and bilirubin, TOS and OSI levels in the conventional or in the intensive phototherapy groups (p > 0.05).
Discussion
The major results of the present study are that endogenous mononuclear leukocyte DNA strand breaks, which are a well-known type of DNA damage, were significantly increased in both conventional and intensive phototherapy-treated infants when compared to the controls. Interestingly, there was no significant correlation between DNA damage scores and TOS levels in the intensive or conventional phototherapy groups. This is the first report showing an association between mononuclear leukocytes DNA damage in intensive or conventional phototherapy-treated jaundiced term infants.
Human peripheral mononuclear leukocytes have been widely used to monitor environmentally induced genetic damage by a variety of methods, such as micronucleus, chromosome aberration, and sister-chromatid exchange assays.19 Among the various assays for measuring DNA damage, the single cell gel electrophoresis (comet) assay is a sensitive and powerful method for determining DNA strand breaks.20,21 It has also been reported that strand breaks arise from DNA damage generated by oxidative stress.14,22 Measuring DNA damage with the comet assay in the human epidermis from phototherapy-treated infants is not possible due to the difficulty in obtaining viable single cells from this tissue in infants. We therefore used this method to measure DNA damage in circulating mononuclear leukocytes. However, future assessments of DNA oxidation in the epidermis may shed further light on the role of oxidative stress in phototherapy-treated infants.
Although we have demonstrated presence of DNA damage in the form of direct strand breaks, we cannot rule out base oxidation. The repair of strand-break ligation (halftime of approximately 30 min) is a much faster process than that of oxidized base lesions by base excision repair (halftime of approximately 3 hours).23 Thus, the repair procedures already existed for phototherapy 48 hours later. Similarly, the partial inhibition of repair induced by the DNA polymerase inhibitor after phototherapy may represent interference with the rejoining of direct strand breaks. Although presence of comets after phototherapy may also indicate that strand breaks were induced as primary lesions, strand breaks measured by the comet assay immediately after phototherapy could just as well represent nicks arising between the incision and rejoining stages of excision repair processes initiated during solar irradiation.24
Photoreactions are able to induce mutagenic photoproducts or lesions in DNA among adjacent pyrimidines in the form of dimmers.25 Although wavelength from 245 to 290 nm is absorbed maximally by DNA,26 as seen in this study, at the wavelength 460 nm DNA damage resulted. This result may be caused by the greater wavelength penetrating a deep layer of the tissue, especially in the newborn's vulnerable soft skin.27 It appears that under phototherapy rate of damage exceeds repair capacity. This study showed that mononuclear leukocyte DNA damages are greater in both conventional phototherapy and intensive phototherapy-treated infants than in not treated phototherapy term jaundiced infants. However, in this study, the main limitation is not using the same patients as their own control (before and during phototherapy). In alkaline comet assay is very complex and samples waiting are not possible.14 Also, laboratory could accept blood samples for study of comet assay only at particular times of the week. These difficulties caused lost of cases and not using the same patients as their own control (before and during phototherapy). We cope with these difficulties owing to using different patients treated with phototherapy for at least 48 hours and controls (not exposure phototherapy). We think that new studies are needed to determine whether this changes in similar cases.
Free radicals can adversely alter lipids, proteins, and DNA.28,29 Reactions of bilirubin involving free radicals or toxic oxygen reduction products have been well documented: unconjugated bilirubin scavenges singlet oxygen with high efficiency, reacts with superoxide anions and peroxyl radicals, and serves as a reducing substrate for peroxidases in the presence of hydrogen peroxide or organic hydroperoxides.30,31 This deterioration is especially evident in the presence of oxidative stress such as phototherapy.7 Bohles et al. reported a significant decrease in antioxidants during phototherapy.32 However, our study showed that TAC levels were not altered significantly by phototherapy. The oxidative/antioxidative balance shifted significantly to the oxidative side, because other indicators of oxidative status, TOS and OSI level, were significantly increased in infants exposed to conventional and intensive phototherapy. We also found that there were no significant correlations between DNA damage ratios and TOS and OSI levels (p > 0.05). In this study, these results indicate that DNA damage was not only caused by oxidative stress but also by other factors such as direct interaction of phototherapy light with DNA. Interestingly, intensive and conventional phototherapy cause similar degrees of DNA damage and oxidative stress. There may be a threshold effect of phototherapy on these parameters.
It is stated that phototherapy's noninvasive nature, easy availability, low cost, and few side effects reported initially have almost led to the assumption that it is innocuous.33 However, those using phototherapy should remain alert to these possibilities and avoid any unnecessary use because untoward effects on DNA have been demonstrated in vitro.3,4,11 Our study showed that both intensive and conventional phototherapy cause unfortunate effects on DNA in vivo.
In conclusion, both conventional phototherapy and intensive phototherapy cause mononuclear leukocyte DNA damage in jaundiced term infants.
Acknowledgements
We are most grateful to the nurses of the Neonatal Service at Sanliurfa Children's Hospital and Harran University Medical School Biochemistry Department for their assistance in conducting this study.
References
Manuscript received Oct 22 2007, accepted for publication Dec 19 2007.
No conflicts of interest declared concerning the publication of this article.
- 1. Porter ML, Dennis BL. Hyperbilirubinemia in the term newborn. Am Fam Physician. 2002;65:599-606.
- 2. Tan KL. Phototherapy for neonatal jaundice. Acta Paediatr. 1996;85:277-9.
- 3. Lamola AA, Blumberg WE, McClead R, Fanaroff A. Photoisomerized bilirubin in blood from infants receiving phototherapy. Proc Natl Acad Sci U S A. 1981;78:1882-6.
- 4. Babara JS, Robert MK. Jaundice and hyperbilirubinemia in the newborn. In: Behrman RE, Kliegman RM, Jenson HB, editors. Nelson Textbook of Pediatrics. 17th ed. Philadelphia, PA: Saunders; 2004. p. 592-7.
- 5. Brennan ML, Wu W, Fu X, Shen Z, Song W, Frost H, et al. A tale of two controversies: defining both the role of peroxidases in nitrotyrosine formation in vivo using eosinophil peroxidase and myeloperoxidase-deficient mice, and the nature of peroxidase-generated reactive nitrogen species. J Biol Chem. 2002;277:17415-27.
- 6. Valko M, Izakovic M, Mazur M, Rhodes CJ, Telser J. Role of oxygen radicals in DNA damage and cancer incidence. Mol Cell Biochem. 2004;266:37-56.
- 7. Gathwala G, Sharma S. Oxidative stress, phototherapy and the neonate. Indian J Pediatr. 2000;67:805-8.
- 8. Turgut M, Basaran O, Cekmen M, Karatas F, Kurt A, Aygun AD. Oxidant and antioxidant levels in preterm newborns with idiopathic hyperbilirubinaemia. J Paediatr Child Health. 2004;40:633-7.
- 9. Akisu M, Yilmaz D, Tuzun S, Kultursay N. Antioxidant defense systems in newborns undergoing phototherapy. Indian J Pediatr. 1999;66:651-5.
- 10. McDonagh AF. The role of singlet oxygen in bilirubin photo-oxidation. Biochem Biophys Res Commun. 1971;44:1306-11.
- 11. Aycicek A, Erel O. Total oxidant/antioxidant status in jaundiced newborns before and after phototherapy. J Pediatr (Rio J). 2007;83:319-22.
- 12. Newman TB, Maisels MJ. Evaluation and treatment of jaundice in the term newborn: A kinder, gentler approach. Pediatrics. 1992;89:809-18.
- 13. Singh NP, McCoy MT, Tice RR, Schneider EL. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res. 1988;175:184-91.
- 14. Kocyigit A, Keles H, Selek S, Guzel S, Celik H, Erel O. Increased DNA damage and oxidative stress in patients with cutaneous leishmaniasis. Mutat Res. 2005;585:71-8.
- 15. Erel O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin Biochem. 2004;37:277-85.
- 16. Erel O. A new automated colorimetric method for measuring total oxidant status. Clin Biochem. 2005;38:1103-11.
- 17. Aycicek A, Erel O, Kocyigit A. Increased oxidative stress in infants exposed to passive smoking. Eur J Pediatr. 2005;164:775-8.
- 18. Harma M, Harma M, Erel O. Oxidative stress in women with preeclampsia. Am J Obstet Gynecol. 2005;192:656-7.
- 19. Cole J, Skopek TR. International Commission for Protection Against Environmental Mutagens and Carcinogens. Working paper no. 3. Somatic mutant frequency, mutation rates and mutational spectra in the human population in vivo. Mutat Res. 1994;304:33-105.
- 20. Moller P, Knudsen LE, Loft S, Wallin H.The comet assay as a rapid test in biomonitoring occupational exposure to DNA-damaging agents and effect of confounding factors. Cancer Epidemiol Biomarkers Prev. 2000;9:1005-15.
- 21. Garcia O, Mandina T, Lamadrid AI, Diaz A, Remigio A, Gonzalez Y, et al. Sensitivity and variability of visual scoring in the comet assay. Results of an inter-laboratory scoring exercise with the use of silver staining. Mutat Res. 2004;556:25-34.
- 22. Rojas E, Lopez MC, Valverde M. Single cell gel electrophoresis assay: methodology and applications. J Chromotogr B Biomed Sci Appl. 1999;722:225-54.
- 23. Sparrow JR, Zhou J, Cai B. DNA is a target of the photodynamic effects elicited in A2E-laden RPE by blue-light illumination. Invest Ophthalmol Vis Sci. 2003;44:2245-51.
- 24. Arlett CF, Lowe JE, Harcourt SA, Waugh AP, Cole J, Roza L, et al. Hypersensitivity of human lymphocytes to UV-B and solar irradiation. Cancer Res. 1993;53:609-14.
- 25. Matsumura Y, Ananthaswamy HN. Toxic effects of ultraviolet radiation on the skin. Toxicol Appl Pharmacol. 2004;195:298-308.
- 26. Tornaletti S, Pfeifer GP. UV damage and repair mechanisms in mammalian cells. Bioessays 1996;18:221-8.
- 27. Meulemans CC, Werner M. Skin penetration depth optical radiation. http://www.solarcsystems.com/pdfs/philips_light_sources_for_phototherapy.pdf Acesso: 19/9/2007.
- 28. Warner BB, Wispe JR Free radical mediated diseases in pediatrics. Semin Perinatol. 1992;16:47-57.
- 29. Halliwell B. Free radicals, antioxidants, and human disease: curiosity, cause, or consequence? Lancet. 1994;344:721-4.
- 30. Stocker R, Ames BN. Potential role of conjugated bilirubin and copper in the metabolism of lipid peroxides in bile. Proc Natl Acad Sci USA. 1987; 84:8130-4.
- 31. Stocker R, Glazer AN, Ames BN. Antioxidant activity of albumin bound bilirubin. Proc Natl Acad Sci USA. 1987;84:5918-22.
- 32. Bohles H, Schnall B. The effect of phototherapy on serum uric acid. Klin Padiatr. 1981;193:308-10.
- 33. Tan KL. Phototherapy for neonatal jaundice. Clin Perinatol. 1991;18:423-39.
Publication Dates
-
Publication in this collection
14 Apr 2008 -
Date of issue
Apr 2008
History
-
Accepted
19 Dec 2007 -
Received
22 Oct 2007