Abstracts
OBJECTIVES: To determine the incidence of extubation failure in preterm newborns with birth weight < 1,250 g extubated to nasal continuous positive airway pressure and to identify the main risk factors associated with the need for reintubation in this population. METHODS: A retrospective review of eligible infants admitted and mechanically ventilated between July 2002 and June 2004 was performed. Extubation failure was defined as the need for reintubation within 7 days after the first extubation attempt. RESULTS: Of the 52 patients included in the study, 13 died before the first extubation attempt. Of the remaining 39 patients, only nine failed extubation (23.1%) Comparing the two groups (failure vs. successful), there was a statistically significant difference regarding birth weight, gestational age and 5-minute Apgar score. After logistic regression, only gestational age was significant. Other secondary outcomes showed significant difference between the groups: intracranial hemorrhage grade III and/or IV, patent ductus arteriosus and death. CONCLUSIONS: The incidence of extubation failure in our population was similar to the rate reported in the literature. The main risk factor for extubation failure was prematurity (≤ 28 weeks). In this population of extreme preterm infants, implementation of strategies for early extubation, use of methylxanthines, prevention of patent ductus arteriosus, and use of different modes of assisted ventilation after extubation may improve the outcomes.
Prematurity; mechanical ventilation; extubation failure; bronchopulmonary dysplasia
OBJETIVOS: Determinar a incidência de falha de extubação em recém-nascidos prematuros com peso de nascimento < 1.250 g extubados para pressão positiva contínua nas vias aéreas nasais e identificar os principais fatores de risco que possam estar associados à necessidade de reintubação nessa população. MÉTODOS: Análise retrospectiva dos prontuários de pacientes internados e ventilados mecanicamente durante o período de julho de 2002 a junho de 2004. Falha na extubação foi definida como necessidade de reintubação nos primeiros 7 dias após a primeira tentativa de extubação. RESULTADOS: Entre 52 pacientes estudados, 13 faleceram antes da primeira tentativa de extubação. Do restante, apenas nove falharam na extubação (23,1%). Comparando esses recém-nascidos com aqueles extubados com sucesso, houve diferença estatisticamente significativa em relação a peso de nascimento, idade gestacional e escore de Apgar no 5º minuto. Após a regressão logística, apenas a idade gestacional se manteve significativa. Alguns resultados secundários também foram significativamente diferentes: incidência de hemorragia intracraniana graus III e/ou IV, persistência do canal arterial e óbito. CONCLUSÕES: Nosso estudo demonstrou uma incidência de falha na extubação semelhante à da literatura. O principal fator de risco para falha nessa população foi a prematuridade (≤ 28 semanas). Nesses prematuros extremos, a implementação de estratégias para extubação precoce, o uso de metilxantinas, a prevenção da abertura do canal arterial e o uso de outros métodos de assistência ventilatória pós-extubação podem contribuir para a melhora desses resultados.
Prematuridade; ventilação mecânica; falha na extubação; displasia broncopulmonar
ORIGINAL ARTICLE
Incidence and main risk factors associated with extubation failure in newborns with birth weight < 1,250 grams
Fernanda HermetoI; Bianca M. R. MartinsII; José R. M. RamosIII; Carlos A. BheringIII;Guilherme M. Sant'AnnaIV
IFellow, Medicina Neonatal-Perinatal, Departamento de Pediatria, McMaster University, Hamilton, ON, Canada
IIMestre em Saúde da Criança e da Mulher, Instituto Fernandes Figueira (IFF), Fundação Oswaldo Cruz (FIOCRUZ), Rio de Janeiro, RJ, Brazil
IIIDoutor em Ciências, IFF, FIOCRUZ, Rio de Janeiro, RJ, Brazil
IVProfessor associado, Departamento de Pediatria, McMaster University, Hamilton, ON, Canada
Correspondence
ABSTRACT
OBJECTIVES: To determine the incidence of extubation failure in preterm newborns with birth weight < 1,250 g extubated to nasal continuous positive airway pressure and to identify the main risk factors associated with the need for reintubation in this population.
METHODS: A retrospective review of eligible infants admitted and mechanically ventilated between July 2002 and June 2004 was performed. Extubation failure was defined as the need for reintubation within 7 days after the first extubation attempt.
RESULTS: Of the 52 patients included in the study, 13 died before the first extubation attempt. Of the remaining 39 patients, only nine failed extubation (23.1%) Comparing the two groups (failure vs. successful), there was a statistically significant difference regarding birth weight, gestational age and 5-minute Apgar score. After logistic regression, only gestational age was significant. Other secondary outcomes showed significant difference between the groups: intracranial hemorrhage grade III and/or IV, patent ductus arteriosus and death.
CONCLUSIONS: The incidence of extubation failure in our population was similar to the rate reported in the literature. The main risk factor for extubation failure was prematurity (≤ 28 weeks). In this population of extreme preterm infants, implementation of strategies for early extubation, use of methylxanthines, prevention of patent ductus arteriosus, and use of different modes of assisted ventilation after extubation may improve the outcomes.
Keywords: Prematurity, mechanical ventilation, extubation failure, bronchopulmonary dysplasia.
Introduction
Invasive mechanical ventilation (MV) is a mode of assisted ventilation often used in intensive care. In very low birth weight (VLBW) infants, this type of assisted ventilation is associated with complications such as pneumothorax, pneumonia, bronchopulmonary dysplasia (BPD), upper airway trauma, neurodevelopment impairment and death.1-4 The main objective in decreasing the duration of invasive ventilation is to reduce these complications.
The respiratory system of preterm infants has peculiar characteristics such as higher instability of upper airway and chest wall, which makes it difficult to perform a successful extubation.5,6 The need for reintubation has also been associated with complications such as longer duration of MV, hospital stay, and higher mortality rate. In adults, approximately 50% of the patients who need to be reintubated present evidence of hypoxemia and hypercapnia or signs of increased work of breathing.7 Therefore, it is extremely important to identify those patients with higher chances of successful extubation.
The estimated incidence of extubation failure in VLBW infants ranges from 20 to 50% depending on birth weight (BW), gestational age (GA), mode of ventilatory support used after extubation and failure criteria.6 Newborns who receive nasal continuous positive airway pressure (CPAP) or noninvasive ventilation after extubation have higher chances of being successfully extubated.8-10
Since there are no measurements or tests with satisfactory levels of sensitivity and specificity to predict the optimal moment for extubation in preterm newborns,11-14 it is important to identify the main factors associated with extubation failure in this population. The objective of the present study is to determine the incidence and the main risk factors associated with the need for reintubation in our neonatal intensive care unit (NICU) in a population of preterm newborns who are extubated to bubble CPAP.
Methods
Population
All newborns with BW ≤ 1,250 g admitted to our NICU and mechanically ventilated from July 2002 to June 2004 were included in the present study. Only the first extubation attempt was considered. Data were retrospectively collected from medical records and using a predefined spreadsheet. Two investigators (F.H. and B.M.) were responsible for data collection. They were assisted by two medical residents. Newborns with congenital malformations or those who were transferred before the first extubation attempt were excluded. During the study, all newborns were extubated to bubble CPAP (5 cm H2O). The study was conducted after being approved by the Research Ethics Committee of the institution.
Ventilatory assistance
All newborns were ventilated using the conventional mode (intermittent mandatory ventilation), with microprocessor controlled volume ventilator model Inter 3 (Intermed®, São Paulo, Brazil), inspiratory time between 0.35 and 0.5 seconds, peak inspiratory pressure between 12 and 20 cm H2O, positive end-expiratory pressure between 4 and 5 cm H2O and ventilatory rate between 15 and 60 bpm. The inspired oxygen fraction (FiO2) was adjusted to keep the oxygen saturation (SaO2) between 88 and 92% (alarms: 85-95%). Those newborns with mean upper airway pressure of approximately 7-8 cm H2O, ventilatory rate ≤ 20 bpm and FiO2 ≤ 0.3 were considered for extubation.
Extubation failure
Extubation failure was defined as the need for reintubation within 7 days after extubation. The following criteria were used to define the need for reintubation: a) respiratory acidosis (partial pressure of carbon dioxide in the arterial blood or PaCO2 > 65 mmHg and pH ≤ 7.25; b) significant number of apnea episodes (> 6 during 6 hours); c) apnea episodes requiring resuscitation; d) consistent increase (> 2 hours) in the need for oxygen higher than 50% in CPAP to keep SaO2 within the desired range. A secondary analysis, using the period of 72 hours as a definition of failure and different weight ranges (≤ 1,000 g and 1,000-1,249 g), was also performed.
Definitions
The following definitions were considered in the present study:
- BPD. We used the definition based on the disease severity.15 All newborns who require supplementary oxygen for ≥ 28 days are diagnosed as having BPD. These newborns are reassessed at 36 weeks of corrected age (if GA ≤ 32 weeks), or on the 56th day of life (if GA ≥ 32 weeks), or at hospital discharge. During reassessment, the newborns kept in room air are classified as having mild BPD, those who are still receiving FiO2 ≤ 0.3 are considered to have moderate BPD, and those with FiO2 > 0.3 and/or CPAP and/or MV are classified as having severe BPD.
- Patent ductus arteriosus (PDA). It is defined based on the presence of clinical signs such as systolic murmur, wide pulse and/or hyperdynamic precordium and need of clinical and/or surgical treatment, or based on diagnosis confirmation via echocardiogram. Indomethacin was administered only as a pharmacologic treatment of PDA.
- Intracranial hemorrhage (ICH). It was diagnosed based on the findings of the cranial ultrasound and classified according to Papile.16
- Retinopathy of prematurity (ROP). It was defined according to the international classification.17
Variables
- Perinatal characteristics: pregnancy-induced hypertension, time of membrane rupture, use of antenatal steroids, mode of delivery, BW, GA, sex, 1- and 5-minute Apgar score and small for GA newborn.
- Ventilatory characteristics: administration of surfactant and age (minutes), number of doses, pre-extubation data (mean airway pressure, FiO2, pH and PaCO2), age at extubation, time interval between extubation and need for reintubation.
- Secondary outcomes: BPD, ICH grade III/IV, ROP, PDA, length of hospital stay, and death.
Statistical analysis
The statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS) version 9.0. Continuous variables were analyzed using Student's t test, and the categorical variables were analyzed using chi-square or Fisher's exact test. Finally, we performed a binary logistic regression analysis including the main risk factors for extubation failure.
Results
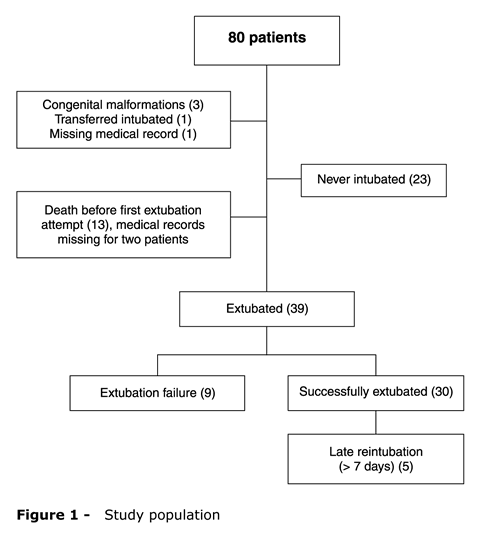
During the study period, 80 infants with BW ≤ 1,250 g were admitted to our NICU. Five newborns were excluded: three had congenital malformations, one was transferred to another hospital ward and one did not have the complete medical record available. Of the 75 remaining patients, 23 (30.6%) were not intubated and 13 died before the first extubation attempt (two patients did not have the complete medical records available). Thus, 39 newborns were assessed: 30 (76.9%) were successfully extubated and nine (23.1%) needed to be reintubated within 7 days after extubation (Figure 1).
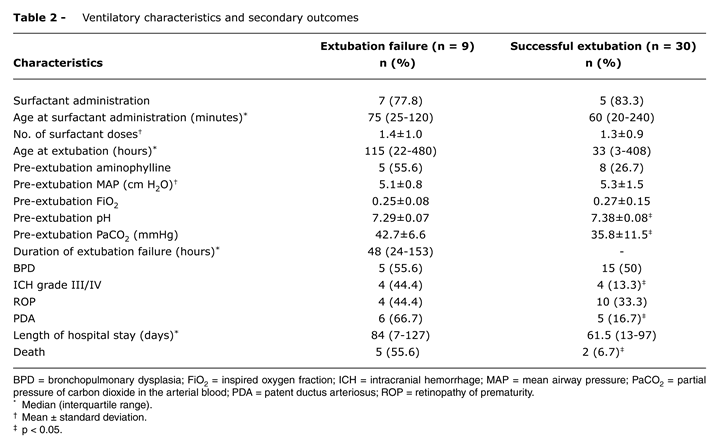
The main characteristics of the population are shown in Table 1. Almost all newborns were ventilated due to hyaline membrane disease (94.8%). There was only one case of pulmonary hypertension associated with pneumothorax (2.6%) and one case of apnea (2.6%). There was no significant difference between cause of MV and extubation failure (p = 0.729). Most of the 13 newborns that died before the first extubation attempt were males, with similar BW and GA to the group of newborns in which extubation failed. Successfully extubated newborns presented significantly higher BW, GA and 5-minute Apgar than those who failed extubation (p ≤ 0.05), were extubated at a younger age (33 hours, ranging from 3 to 408 vs. 115 hours, ranging from 22 to 480; p = 0.07), had higher pH values (7.38±0.08 vs. 7.29±0.07; p ≤ 0.05) and lower PaCO2 levels (35.8±11.5 vs. 42.7±6.6 mmHg; p ≤ 0.05) at pre-extubation blood gas analysis (Table 2). Those newborns for whom extubation failed were reintubated approximately 48 hours after extubation (median: 48 hours; interquartile range: 24-153 hours). Successfully extubated newborns had a lower incidence of PDA, ICH grade III/IV and death (Table 2).
Extubation success rate ranged according to the criterion used: 72 hours = 82.1% and 7 days = 76.9%. For the newborns with BW ≤ 1,000 g, these percentages were 70 and 65%, and for those between 1,000 and 1,249 g, the percentages were 94.8 and 89.5%, respectively (Figure 2). The main reasons for extubation failure were: apnea (66.7%), pneumothorax (22.2%) and increased work of breathing (11.1%). In the logistic regression analysis for the main risk factors, only GA was significant (≤ 28 weeks).
Discussion
Our study showed an incidence of extubation failure of 23% in a population of newborns with BW ≤ 1,250 g who were ventilated using the conventional mode and extubated to nasal CPAP under 5 cm of water seal. The main risk factor associated with this outcome was GA at birth. The several aspects related to extubation success for these preterm infants are discussed next.
Incidence and causes of extubation failure
Data from the literature report a wide range on the rate of extubation failure due to important differences in the populations studied (BW and GA), failure criteria and modes of ventilatory support used after extubation.6,8,13,18 Davis et al.,19 in a study involving preterm infants with BW between 600 and 1,250 g, showed a significantly lower incidence of failure in newborns extubated to nasal CPAP (34%) in comparison with those extubated to oxyhood (60%). Recent studies have demonstrated an even lower incidence in newborns extubated to noninvasive ventilation in comparison with those extubated to nasal CPAP.8,20 In our study, all patients were extubated to nasal CPAP. Using 7 days as a period of time to define extubation failure, we found a rate of 35% in newborns with BW ≤ 1,000 g. Stefanescu et al.,6 in a population with GA and BW similar to our study, and using similar definitions, reported a failure incidence of 8% in newborns with BW > 1,000 g and 40% in those with BW ≤ 1,000 g. In our study, 18% of the newborns were reintubated within 72 hours after extubation. Barrington et al.,8 in a population with lower GA and BW, and using the same 72 hours as failure definition, reported an incidence rate of 44%. An important limitation of our study was the death of 13 newborns before any extubation attempt. Considering that these infants had BW and GA similar to those who had failed extubation, if all of them had been included in this group, our failure incidence would have increased from 23 to 42% even so, it would fall within the percentage range of failure reported in the literature for newborns with BW ≤ 1,250 g extubated to nasal CPAP.
Apnea, pneumothorax, and increased work of breathing were the main causes of reintubation in our sample. Other studies have confirmed our results; the major identified causes were apnea, increased work of breathing and increased oxygen requirements.11,13,19
Risk factors
In a study conducted by Dimitriou et al.,11 gestational age (GA ≤ 30 weeks) and postnatal age were risk factors more significant than the measurements of respiratory muscle strength and respiratory load. Szymankiewicz et al.13 assessed the pulmonary mechanics of VLBW newborns before they were extubated and demonstrated that patients who had successful extubation had significantly better results of pulmonary function and similar clinical characteristics in comparison with patients who failed. In our study, such measures were not routinely taken before extubation. Among the data collected, we found that the following factors had an influence on the percentage of extubation failure: BW (p = 0.03), GA (p ≤ 0.01) and 5-minute Apgar score (p ≤ 0.01). However, after performing the logistic regression analysis, only GA was significant between the groups studied.
Weaning form MV
Slow weaning, with delayed first extubation attempt, is associated with a higher failure rate and a prolonged use of MV. Danan et al.,12 using optimized MV, delayed this first extubation attempt for 36 hours without achieving any improvement of the success rate. Thomson et al.21 compared extubation in 24 hours with extubation on the 5th day of life in preterm babbons and demonstrated that the animals extubated later had increased number of apneic episodes, duration of MV and increased oxygen requirements, with more severe lung injury and increased number of inflammatory markers. In our study, we found an important, although non-significant, difference in the postnatal age at extubation (success = 33 hours vs. failure = 115 hours; p = 0.07). We have recently demonstrated, in a population with similar BW and GA, the introduction of a MV protocol, which established evidence-based criteria for extubation and reintubation, was able to provide a significant reduction in the incidence of extubation failure in newborns who were extubated earlier (5 days vs. 1.2 day).22
The use of methylxanthines also reduces time on MV, mainly in newborns with BW ≤ 1,000 g who are extubated in the first week of life.23 Recently, Schmidt et al.24 demonstrated a significant decrease in the time of ventilatory support in newborns who received caffeine at 3-4 days of life while on MV. In our study, methylxanthines were not used as a routine treatment and/or as a prophylactic procedure.
Secondary outcomes
Inappropriate weaning from MV may cause atelectasis, episodes of hypoxia and, as a consequence, brain injury.7,25 Therefore, it is extremely important to identify the moment when the patient has higher chances of being successfully extubated. In our study, we found significant differences in rates of ICH grade III/IV (p = 0.04), PDA (p ≤ 0.01) and death (p ≤ 0.01) between the patients who were successfully extubated and those who needed reintubation. Due to the retrospective design of our study, it is difficult to determine the causality between these outcomes and extubation failure. Nevertheless, for most extreme preterm infants, ICH and PDA are events that occur during the first days of life. These intercurrent events may be caused by newborns' higher immaturity (lower GA) and/or severity, contributing to the delayed ventilatory weaning and the consequent increase in failure incidence. On the other hand, delayed extubation, as mentioned above, is associated with worsening of the lung injury and increased number of inflammatory markers. On its turn, increase in the inflammatory activity has been associated with a higher incidence of ICH and reopening of the ductus arteriosus.22,26,27
Conclusion
Our study demonstrated an incidence of extubation failure similar to the rates reported in the literature for a NICU where the newborns are ventilated in a conventional mode and extubated to bubble CPAP. We used specific criteria for oxygenation, extubation and reintubation, and the main risk factor for failure was extreme prematurity (≤ 28 weeks). In this population, the implementation of ventilatory weaning protocols, administration of methylxanthines (caffeine), prevention of PDA and use of alternative methods of post-extubation ventilatory assistance (nasal ventilation) may contribute to improve such outcomes.
Acknowledgements
The authors thank Dr. Daniele C. Alexandre and Dr. Marcia T. da Silva for their help with data collection.
References
- 1. Finer NN, Carlo WA, Duara S, Fanaroff AA, Donovan EF, Wright LL, et al. Delivery room continuous positive airway pressure/positive end-expiratory pressure in extremely low birth weight infants: a feasibility trial. Pediatrics. 2004;114:651-7.
- 2. Walsh MC, Morris BH, Wrage LA, Vohr BR, Poole WK, Tyson JE, et al. Extremely low birthweight neonates with protracted ventilation: mortality and 18-month neurodevelopmental outcomes. J Pediatr. 2005;146:798-804.
- 3. Carlo WA, Stark AR, Wright LL, Tyson JE, Papile LA, Shankaran S, et al. Minimal ventilation to prevent bronchopulmonary dysplasia in extremely-low-birth-weight infants. J Pediatr. 2002;141:370-4.
- 4. Ambalavanan N, Van Meurs KP, Perritt R, Carlo WA, Ehrenkranz RA, Stevenson DK, et al. Predictors of death or bronchopulmonary dysplasia in preterm infants with respiratory failure. J Perinatol. 2008;28:420-6
- 5. De Paoli AG, Morley C, Davis PG. Nasal CPAP for neonates: what do we know in 2003? Arch Dis Child Fetal Neonatal Ed. 2003;88:F168-72.
- 6. Stefanescu BM, Murphy WP, Hansell BJ, Fuloria M, Morgan TM, Aschner JL. A randomized, controlled trial comparing two different continuous positive airway pressure systems for the successful extubation of extremely low birth weight infants. Pediatrics. 2003;112:1031-8.
- 7. Rothaar RC, Epstein SK. Extubation failure: magnitude of the problem, impact on outcomes, and prevention. Curr Opin Crit Care. 2003;9:59-66.
- 8. Barrington KJ, Bull D, Finer NN. Randomized trial of nasal synchronized intermittent mandatory ventilation compared with continuous positive airway pressure after extubation of very low birth weight infants. Pediatrics. 2001;107:638-41.
- 9. Davis PG, Henderson-Smart DJ. Nasal continuous positive airways pressure immediately after extubation for preventing morbidity in preterm infants. Cochrane Database Syst Rev. 2000;(3):CD000143.
- 10. Kamlin CO, Davis PG, Morley CJ. Predicting successful extubation of very low birthweight infants. Arch Dis Child Fetal Neonatal Ed. 2006;91:F180-3.
- 11. Dimitriou G, Greenough A, Endo A, Cherian S, Rafferty GF. Prediction of extubation failure in preterm infants. Arch Dis Child Fetal Neonatal Ed. 2002;86:F32-F35.
- 12. Danan C, Durrmeyer X, Brochard L, Decobert F, Benani M, Dassieu G. A randomized trial of delayed extubation for the reduction of reintubation in extremely preterm infants. Pediatr Pulmonol. 2008;43:117-24.
- 13. Szymankiewicz M, Vidyasagar D, Gadzinowski J. Predictors of successful extubation of preterm low-birth-weight infants with respiratory distress syndrome. Pediatr Crit Care Med. 2005;6:44-9.
- 14. Davis PG, Henderson-Smart DJ. Extubation from low-rate intermittent positive airways pressure versus extubation after a trial of endotracheal continuous positive airways pressure in intubated preterm infants. Cochrane Database Syst Rev. 2000;(2):CD001078.
- 15. Jobe AH, Bancalari E. Bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2001;163:1723-9.
- 16. Papile LA, Burstein J, Burstein R, Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J Pediatr. 1978;92:529-34.
- 17. International Committee for the Classification of Retinopathy of Prematurity. The International Classification of Retinopathy of Prematurity Revisited. Arch Ophthalmol. 2005;123:991-9.
- 18. Davis P, Henderson-Smart D. Post-extubation prophylactic nasal continuous positive airway pressure in preterm infants: systematic review and meta-analysis. J Paediatr Child Health. 199;35:367-71.
- 19. Davis P, Jankov R, Doyle L, Henschke P. Randomised, controlled trial of nasal continuous positive airway pressure in the extubation of infants weighing 600 to 1250 g. Arch Dis Child Fetal Neonatal Ed. 1998;79:F54-7
- 20. Khalaf MN, Brodsky N, Hurley J, Bhandari V. A prospective randomized, controlled trial comparing synchronized nasal intermittent positive pressure ventilation versus nasal continuous positive airway pressure as modes of extubation. Pediatrics. 2001;108:13-7.
- 21. Thomson MA, Yoder BA, Winter VT, Giavedoni L, Chang LY, Coalson JJ. Delayed extubation to nasal continuous positive airway pressure in the immature baboon model of bronchopulmonary dysplasia: lung clinical and pathological findings. Pediatrics. 2006;118:2038-50.
- 22. Hermeto F, Bottino MN, Vaillancourt K, Sant'Anna GM. Implementation of a respiratory therapist-driven protocol for neonatal ventilation: impact on the premature population. Pediatrics. 2009;123:e907-16.
- 23. Henderson-Smart DJ, Davis PG. Prophylactic methylxanthines for extubation in preterm infants. Cochrane Database Syst Rev. 2003;(1):CD000139.
- 24. Schmidt B, Roberts RS, Davis P, Doyle LW, Barrington KJ, Ohlsson A, et al. Caffeine therapy for apnea of prematurity. N Engl J Med. 2006;354:2112-21.
- 25. Epstein SK, Ciubotaru RL. Independent effects of etiology of failure and time to reintubation on outcome for patients failing extubation. Am J Respir Crit Care Med. 1998;158:489-93.
- 26. Rocha G, Proenca E, Quintas C, Rodrigues T, Guimarães H. Chorioamnionitis and brain damage in the preterm newborn. J Matern Fetal Neonatal Med. 2007;20:745-9.
- 27. Goldenberg RL, Andrews WW, Faye-Petersen OM, Cliver SP, Goepfert AR, Hauth JC. The Alabama preterm birth study: corticosteroids and neonatal outcomes in 23- to 32-week newborns with various markers of intrauterine infection. Am J Obstet Gynecol. 2006;195:1020-4.
Correspondência:
Publication Dates
-
Publication in this collection
30 Oct 2009 -
Date of issue
Oct 2009
History
-
Accepted
19 June 2009 -
Received
22 Apr 2009