Abstracts
OBJECTIVE: To measure the impact of atopic dermatitis (AD) on the quality of life of pediatric patients and their families, establishing correlations with scores of disease severity. METHODS: This was an observational study of the correlations between clinical indicators of severity and two questionnaires on quality of life: IDQOL and DFI. The study also included scoring of eczema severity - EASI. Forty-two children with AD, fulfilling established diagnostic criteria, and 44 children with other dermatologic diseases were investigated for the effect of eczema on quality of life. Pearson's correlation was used for the correlation analysis and the comparison between the groups was carried out using the Mann-Whitney test. RESULTS: Data analysis demonstrated significant differences between the scores for the two groups. The mean score in the eczema group was 9.2 (range 1-19) for IDQOL and 8.5 (range 0-17) for DFI. The highest scoring questions for IDQOL referred to itching and scratching, mood changes and problems caused by treatment. For the FDI, the highest impact domains were treatment-related expenditure and sleep disturbance affecting family members. CONCLUSIONS: AD has a negative impact on the quality of life of pediatric patients and their families. Data obtained in studies of quality of life in AD should be used to guide clinical practice in order to identify individual treatment strategies and should lead to the adoption of measures to reduce the impact of the disease on patients and their families.
Quality of life; atopic dermatitis; children
OBJETIVO: Avaliar o impacto da dermatite atópica (DA) sobre a qualidade de vida de pacientes pediátricos e de seus familiares, estabelecendo correlações com escores de severidade da doença. MÉTODOS: Estudo observacional com correlação entre indicador clínico de gravidade e dois questionários sobre qualidade de vida: o Índice de Qualidade de Vida da Dermatite Atópica em Crianças (IDQOL) e o Impacto da Dermatite Atópica na Família (DFI). O estudo incluiu também um escore de gravidade do eczema (Índice de Gravidade do Eczema por Área). Foram investigadas 42 crianças com DA e 44 crianças com outras dermatoses em relação ao impacto do eczema na qualidade de vida. Na análise de correlação, utilizou-se o teste de correlação de Pearson, e a comparação entre os dois grupos foi realizada através do teste de Mann-Whitney. RESULTADOS: A análise dos dados demonstrou significativa diferença entre a pontuação dos dois grupos. A média de pontuação no grupo com DA foi de 9,2 (variando de 1 a 19) para o IDQOL e de 8,5 (variando de 0 a 17) para o DFI. Os domínios mais comprometidos no IDQOL foram os relativos ao prurido, alterações de humor e problemas com o tratamento. Quanto ao DFI, os domínios de maior impacto foram os relacionados a despesas com o tratamento e a perturbação do sono de membros da família. CONCLUSÕES: A DA interfere negativamente na qualidade de vida dos pacientes pediátricos e dos seus familiares. Dados obtidos nos estudos de qualidade de vida em DA devem nortear a prática clínica de modo a individualizar as estratégias de tratamento e levar à adoção de medidas que possam minimizar o impacto da doença sobre pacientes e familiares.
Qualidade de vida; dermatite atópica; criança
ORIGINAL ARTICLE
Quality of life in pediatric patients with atopic dermatitis
Tassiana M. M. AlvarengaI; Antônio P. CaldeiraII
IMestranda, Programa de Pós-Graduação em Ciências da Saúde, Universidade Estadual de Montes Claros (UNIMONTES), Montes Claros, MG, Brazil
IIDoutor, Medicina, Universidade Federal de Minas Gerais (UFMG). Docente, Programa de Pós-Graduação em Ciências da Saúde, UNIMONTES, Montes Claros, MG, Brazil
Correspondence
ABSTRACT
OBJECTIVE: To measure the impact of atopic dermatitis (AD) on the quality of life of pediatric patients and their families, establishing correlations with scores of disease severity.
METHODS: This was an observational study of the correlations between clinical indicators of severity and two questionnaires on quality of life: IDQOL and DFI. The study also included scoring of eczema severity EASI. Forty-two children with AD, fulfilling established diagnostic criteria, and 44 children with other dermatologic diseases were investigated for the effect of eczema on quality of life. Pearson's correlation was used for the correlation analysis and the comparison between the groups was carried out using the Mann-Whitney test.
RESULTS: Data analysis demonstrated significant differences between the scores for the two groups. The mean score in the eczema group was 9.2 (range 1-19) for IDQOL and 8.5 (range 0-17) for DFI. The highest scoring questions for IDQOL referred to itching and scratching, mood changes and problems caused by treatment. For the FDI, the highest impact domains were treatment-related expenditure and sleep disturbance affecting family members.
CONCLUSIONS: AD has a negative impact on the quality of life of pediatric patients and their families. Data obtained in studies of quality of life in AD should be used to guide clinical practice in order to identify individual treatment strategies and should lead to the adoption of measures to reduce the impact of the disease on patients and their families.
Keywords: Quality of life, atopic dermatitis, children.
Introduction
For a long time the impact of disease on people was measured simply in terms of mortality indexes. As the concept of health has widened, other measures have been developed and applied in order to gauge the degree to which certain diseases compromise the lives of individual people. It was within this context that the concepts of quality of life and health-related quality of life emerged.1,2 Nowadays, these measures are widely used in healthcare and are considered indispensable for approving and defining treatments and for assessing the cost-benefit relationship of care provided.3
Worldwide interest in the subject has led to the development of many instruments; some generic intended to assess the quality of life of populations in general and others specific designed for groups of individuals suffering from a specific acute or chronic disease.4 In general, chronic diseases are the primary focus of quality of life assessments, since they have a major impact on patients' lives.5,6 Prominent among these are dermatological diseases, since they have a profound effect on people's lives, whether due to the itching, the anti-esthetic appearance or to restrictions caused by lesions or by treatment. It has been proven that they have a significant impact on social relations, on psychological status and the day-to-day activities of patients.7 As a result, many different instruments for measuring quality of life have been developed specifically for patients with dermatological diseases, aimed at both the adult and pediatric populations.8,9
Against this background, and as a result of its chronic and recidivist character, the intense pruritis involved, the disturbance of sleep and day-to-day activities and the potential association with bronchial asthma, atopic dermatitis (AD) is a socially and psychologically relevant disease that affects the patients themselves and, decisively, their entire family and professional environments.10 In conjunction with the damage caused by the disease itself, there is also a social, emotional and financial burden on patients' families. The parents of affected children report difficulties disciplining and caring for their children, primarily due to sleep deprivation, exhaustion and problems with both the cost and administration of topical and systemic medication.11,12 The overload caused by caring for these patients generates conflict between parents and also with any eczema-free siblings, altering the family structure.13
In view of the relevance of this subject and the lack of Brazilian studies, this research was carried out with the objective of assessing the quality of life of pediatric patients with AD and their families, tracing correlations between disease severity scales.
Methods
This study was undertaken in Montes Claros, which is a municipality in the North of the Brazilian state of Minas Gerais. It is the principal urban center in the region and has a population of approximately 360,000 inhabitants. This is a deprived area with socioeconomic indicators that are comparable with the poorest areas of Brazil.
Data collection was carried-out for a period of 1 year (September/2007 to August/2008), during which patients aged from 6 to 59 months were recruited at the Dermatology Clinic at the Hospital Universitário Clemente de Faria belonging to the Universidade Estadual de Montes Claros (UNIMONTES), Montes Claros, Brazil. These patients were classified into two groups: the first comprising children with AD, diagnosed according to criteria published by Hanifin & Rakja,14 and the second comprising children with other types of dermatosis, used as a control group. Patients were excluded if they were suffering from other chronic conditions.
No sample size calculation was performed. The municipality's health professionals (Family Health Program physicians, pediatricians and dermatologists) were informed of the project and invited to refer patients. After the project had been publicized, all patients presenting at the clinic for a first consultation and fitting the diagnostic criteria were recruited. All of these patients were being treated on the Brazilian National Health Service (Sistema Único de Saúde, SUS), and their parents or guardians gave permission for them to take part. This type of sample selection process, in which investigators make use of the most accessible elements of a population, is called sampling by convenience, or non-probabilistic accidental sampling, and is appropriate for situations such as this, in which the event being investigated in uncommon and/or does not allow for probabilistic sampling.
During the data collection phase, patients underwent a dermatological clinical examination performed by one of the investigators and then their parents or guardians answered two structured quality of life questionnaires.
Disease severity was assessed using the Eczema Area and Severity Index (EASI), which was developed by Hanifin et al.15 and measures the severity of the condition based on combinations of clinical signs erythema, papules, abrasions and lichenification and the surface area of the body affected. Possible scores are from 0 to 72, with higher scores correlating with greater disease severity. In this study, patients with EASI scores less than or equal to 20 were classified as mild AD patients, and those with EASI scores above 20 were classified as severe AD patients.
We used the following instruments to measure quality of life: Infant's Dermatitis Quality of Life Index (IDQOL) and the Dermatitis Family Impact Questionnaire (DFI), both validated for use in Portuguese.16,17
The IDQOL covers the following items: sleep and mood disturbances, difficulties in taking part in recreational activities or family life and discomfort when bathing, dressing and eating. It comprises ten questions, answered by the patient's parents on the basis of the previous week.16,18
The DFI also comprises ten questions about the effects of AD on family life domains emotional disorders, sleep disturbances, impact on cleanliness at home, on food, on leisure activities and on the relationship between the patient's parents and the impact of treatment costs and the questions are also about the previous week.12,16 It should be pointed out that no numerical cutoffs have been proposed for either instrument, but there is a direct relationship between the scores obtained and prejudice to quality of life.
Statistical analysis was performed using SPSS, version 15.0. The relationship between the disease severity indexes and the quality of life indexes was assessed using Pearson's correlation. The Mann-Whitney test was used to compare perceived quality of life between the group of patients with AD and the group with other types of dermatosis. The significance level adopted for this study was 5% (p ≤ 0.05).
The research protocol was approved by the Research Ethics Committee at UNIMONTES, and the patients' parents or guardians signed free and informed consent forms.
Results
We recruited 42 children with AD for the study; 19 (45.2%) males and 23 (54.8%) females. For the control group we enrolled 44 children with other types of dermatosis; 20 (45.5%) males and 24 (54.5%) females. The median age of the children who took part in the study was 25.2 months. There were also no statistically significant differences between the two groups in terms of the distribution of ages or classification of socioeconomic status. The most common dermatological conditions in the control group were dermatomycoses, impetigo, molluscum contagiosum, verruca vulgaris and sweat gland disorders.
Table 1 contains the classifications of disease severity according to the parents' perceptions; a clear difference can be observed between the two study groups.
Figure 1 illustrates the correlation between quality of life score according to the IDQOL and dermatosis severity according to the dermatological assessment (EASI) and the correlation between the impact of AD on the family according to the DFI and the dermatological assessment (EASI). The Pearson correlation coefficients for these analyses were r = 0.219 (EASI x IDQOL) and r = 0.337 (EASI x DFI).
The scores calculated for IDQOL, DFI and EASI are given in Table 2, together with their minimum and maximum values, means and standard deviations.
Clinical assessment of the severity of AD, according to EASI, demonstrated that the majority of patients had mild forms of dermatosis, with scores ≤ 20 (mean 9.2). Analysis of the parents' perspective of the disease showed that the majority considered that their children had a case of average severity.
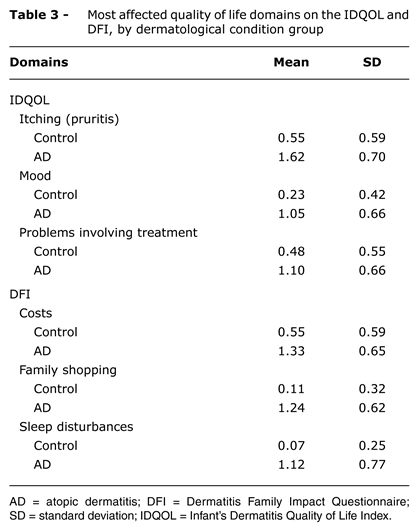
Analysis of the IDQOL questionnaires for the patients with AD revealed that the domains most affected were those that covered itching, mood changes, problems with treatment and discomfort while bathing. The domains related to interference with participation in family activities and the time that children took to be able to go to sleep were scored lowest. The lowest score was 1 and the highest was 19 (mean 9.2).
Analysis of the DFI questionnaire scores indicated the greatest impact in domains assessing treatment costs, the effect on the family shopping, the effect on other family members' sleep and eczema as a cause of tiredness or exhaustion for family members. The domains that scored lowest were those related to the effects on the parents' relationship with each other, on housework and on food preparation. The lowest score was 1 and the highest 17 (mean 8.5).
The highest IDQOL scores in the control group were for the domains of itching, problems with treatment and time taken to go to sleep. The lowest scores were in the following domains: sleep disturbances, interference with participation in family activities and problems at mealtimes. The lowest score was 0 and the highest was 9 (mean 2.0). The highest scores on the DFI scale were related with the costs of the disease.
The domains in which the AD patients' and control group patients' quality of life suffered the greatest impacts are shown in Table 3.
Discussion
AD is a chronic cutaneous inflammatory disease of a genetic nature that is characterized by recurrent episodes of eczema associated with itching, which has a substrate of immunological cutaneous involvement leading to inflammation, and which can occur in association with other forms of allergic diseases, such as asthma and allergic rhinitis.19 While the effects of AD on children and their families have been well-established in many countries, studies of this type remain rare in Brazil, especially with children less than 5 years old. Our study has concentrated on assessing this population, using quality of life questionnaires to determine the impact of AD on patients and their families.
It should be emphasized that the results of this study should be interpreted with care, due to certain limitations. The first of these is related to the sample of convenience selected for the study from a recently-established specialist centre. Nevertheless, it should also be pointed out that this is the normal sampling process employed when investigating uncommon events. In such cases, the validity of the study is not judged on the basis of the sample size, but on the conditional hypothesis that the sample selected behaves in a similar manner, irrespective of whether that process is random or not.20 This group of patients had mild AD affecting the majority of their limbs and, to this extent, are representative of all patients with this disease. In this case, the bias of non-randomized sampling is be to reduce the magnitude of observed results. In other words, in real-life situations, it is probable that the impact of this disease on patients and their families is greater than described in this study. When the statistical power provided by this sample was calculated the result proved satisfactory (over 80%), which confers reliability on the results. The fact that this study was restricted to SUS patients is also a limitation, since it precludes the inclusion of patients with higher socioeconomic status. Notwithstanding the limitations described, the profile described here is compatible with the majority of AD patients in other studies.21,22
The comparison between clinical disease severity and parent-assessed severity indicated a weak correlation (r = 0.306). The clinical assessments found that the majority of these patients had mild AD (EASI = 20), while the majority of their parents stated that their children's AD was of average severity. This discrepancy has been detected in other studies, one of which was that undertaken by Eiser et al.,23 which reported that parents generally have a more negative view of the disease (course, progression) and of its effects on their children. The same study also found evidence that parents are better able to judge domains related to physical symptoms (pain, itching, tiredness, etc.), but are less accurate when judging emotional and psychological domains (anxiety, sadness, etc.).23
Analysis of Figure 1 reveals that, although there was a positive correlation between severity according to EASI and family perception of quality of life, no significant inclination of curves is seen (IDQOL or DFI). This fact, which has previously been reported in another Brazilian study,24 may be a result of the restrictive character of the sample (just 42 patients) or may be the result of the fact that in the majority of cases patients had AD that was classified as mild.
The majority of recently published studies that have employed the IDQOL state that the highest-scoring questions, therefore considered those relating to the greatest impact on quality of life, are those about itching/scratching, mood changes and sleep disorders. The questions revealing less impact relate to the interference of AD in family activities and discomfort when dressing and undressing children.21,22 In our study, the IDQOL domain that scored highest was also that related to itching, which is in agreement with international literature that considers the correlation between itching and quality of life to be well-established. Night time itching affects both the quality and quantity of patients' sleep, causing tiredness, irritability, problems with concentration and learning and poor performance at school.10 In a study carried out in Porto Alegre in 2004, Weber et al. demonstrated that this symptom was present daily in 74.2% of their study population, and that patients with severe AD exhibited more intense and more frequent itching than patients with mild and moderate forms of the disease.25
There was also a significant impact on the quality of life of our study population in the domain covering the influence of AD on mood. This is in agreement with international studies, which report that mood changes, hyperreactivity, irritability and crying during the daytime and when topical and systemic medications are administered are all observed.26,27
With relation to the DFI, the domain in which the greatest impact on quality of life of family members was registered was that covering the financial costs of the disease (treatment, medical consultations, special clothing, etc.). This result is in contrast with the results of some other studies which indicate that sleep disorders and tiredness/exhaustion are the domains where the greatest impact is felt.28,29 This difference may be the result of the socioeconomic and regional characteristics of our sample, since it is made up of patients treated on the SUS from one of the poorest regions of the country.
Although it is not the factor causing the greatest impact on quality of life, the cost of management of these patients is significant and is comparable to the costs of diseases such as psoriasis and asthma, and has been estimated at around one billion annually in the United States.12,30
There was also a significant impact within the domains covering tiredness/exhaustion of parents and impact on the sleep of other family members. An Australian study undertaken by Su et al., and which used the DFI, concluded that caring for patients with moderate and severe AD is more stressful than caring for children with insulin-dependent diabetes.30 In a study carried out in Switzerland, Gånemo et al. assessed the quality of life of children and their families using the IDQOL, the DFI and the Children's Dermatology Life Quality Index (CDLQI) simultaneously. Their results showed a concordance between the scores for the three questionnaires, indicating that itching, lost sleep, mood changes, treatment costs and parents' feelings of guilt were the factors most responsible for the impact on quality of life.22
In the results for the IDQOL control group, the highest scores were for itching and problems related to treatment, but the scores in these domains were significantly lower than for the patients with AD. The presence of dermatoses with itching and physical discomfort, such as scabies, miliaria and impetigo, among patients in the control group may explain these results.
The results found in this study are in agreement with those of other international studies in terms of the correlation between AD severity and its impact on the quality of life of patients and their families. They therefore underline the significant impact of AD on pediatric patients and the need to utilize instruments that make it possible to measure that impact. It is ever more necessary to be aware of patients' families' perceptions of the disease, as a tool to aid physicians in their choice of the appropriate treatment, improving the patient-doctor relationship and becoming an important factor in improving these people's quality of life.
References
- 1. Minayo MC, Hartz ZM, Buss PM. Qualidade de vida e saúde: um debate necessário. Cienc Saude Coletiva. 2000;5:7-18.
- 2. Frisén A. Measuring health-related quality of life in adolescence. Acta Paediatr. 2007;96:963-8.
- 3. Dantas RA, Sawada NO, Malerbo MB. Pesquisas sobre qualidade de vida: revisão da produção científica das universidades públicas do estado de São Paulo. Rev Latino Am Enfermagem. 2003;11:532-8.
- 4. Fleck MP, Leal OF, Louzada S, Xavier M, Chachamovich E, Vieira G, et al. Desenvolvimento da versão em português do instrumento de avaliação de qualidade de vida da OMS (WHOQOL-100). Rev Bras Psiquiatria.1999;21:19-28.
- 5. Grange A, Bekker H, Noyes J, Langley P. Adequacy of health-related quality of life measures in children under 5 years old: systematic review. J Adv Nurs. 2007;59:197-220.
- 6. Klatchoian DA, Len CA, Terreri MT, Silva M, Itamoto C, Ciconelli RM, et al. Quality of life of children and adolescents from São Paulo: reliability and validity of the Brazilian version of the Pediatric Quality of Life InventoryTM version 4.0 Generic Core Scales. J Pediatr (Rio J). 2008;84:308-15.
- 7. Misery L, Finlay AY, Martin N, Boussetta S, Nguyen C, Myon E, et al. Atopic dermatitis: impact on the quality of life of patients and their partners. Dermatology. 2007;215:123-9.
- 8. Lewis-Jones MS, Finlay AY. The children's Dermatology Life Quality Index (CDLQI): initial validation and pratical use. Br J Dermatol. 1995;132:942-9.
- 9. Finlay AY, Khan GK. Dermatology Life Quality Index (DLQI): a simple practical measure for routine clinical use. Clin Exp Dermatol. 1994;19:210-6.
- 10. Bello RT. Qualidade de vida em dermatologia. Rev SPDV. 2005, 63:35-57.
- 11. LeBovidge JS, Kelley SD, Lauretti A, Bailey EP, Timmons KV, Horn MV et al. Integrating medical and psychological health care for children with atopic dermatitis. J Pediatr Psychol. 2007;32:61725.
- 12. Mancini AJ, Kaulback K, Chamlin SL. The socioeconomic impact of atopic dermatitis in the United States: a systematic review. Pediatr Dermatol. 2008;25:1-6.
- 13. Beattie PE, Lewis-Jones MS. A comparative study of impairment of quality of life in children with skin disease and children with other chronic childhood diseases. Br J Dermatol. 2006;155:145-51.
- 14. Hanifin JM, Rajka G. Diagnostic features of atopic dermatitis. Acta Derm Venereol Suppl (Stockh). 1980;92:44-7.
- 15. Hanifin JM, Thurston M, Omoto M, Cherill R, Tofte SJ, Graeber M. The eczema area and severity index (EASI) assessment of reliability in atopic dermatitis. Exp Dermatol. 2001;10:11-8.
- 16. Lewis-Jones MS, Finlay AY. Wales College of Medicine, Quality of life research. http://www.dermatology.org.uk . Acesso:10/04/2006.
- 17. Weber MB, Fontes Neto PT, Prati C, Soirefman M, Mazzotti NG, Barzenski B, et al.Improvement of pruritus and quality of life of children with atopic dermatitis and their families after joining support groups. JEADV. 2008;22:992-9.
- 18. Lewis-Jones MS, Finlay AY, Dykes PJ. The infant's Dermatitis Quality of Life Index. Br J Dermatol. 2001;144:104-10.
- 19. Leung DY, Coter NA. Cellular and immumologic mechanisms in atopic dermatitis. JAM Acad Dermatol. 2001;44:S1-12.
- 20. Luiz RR, Magnanini MM. A lógica da determinação do tamanho da amostra em investigações epidemiológicas. Cad Saude Publica. 2000;8:9-28.
- 21. Ricci G, Bendandi B, Bellini F, Patrizi A, Masi M. Atopic dermatitis: quality of life of young Italian children and their families and correlation with severity score. Pediatr Allergy Immunol. 2007;18:245-9.
- 22. Gĺnemo A, Svensson A, Lindberg M, Wahlgren CF. Quality of life in Swedish children with eczema. Acta Derm Venereol. 2007;87:345-9.
- 23. Eiser C, Morse R. Can parents rate their child's health related quality of life? Results of a systematic review. Qual Life Res. 2001;10:347-57.
- 24. Coghi S, Bortoletto MC, Sampaio SA, Andrade Junior HF, Aoki V. Quality of life is severely compromised in adult patients with atopic dermatitis in Brazil, especially due to mental components. Clinics (Sao Paulo). 2007;62:235-42
- 25. Weber MB, Petry V, Weis L, Mazzotti NG, Cestari TF. Avaliação da relação do prurido e níveis sangüíneos de IgE com a gravidade do quadro clínico em pacientes com dermatite atópica. An Bras Dermatol. 2005;80:245-8.
- 26. Chamlin SL, Frieden IJ, Williams ML, Chren MM. Effects of atopic dermatitis on young American children and their families. Pediatrics. 2004;3:607-11.
- 27. Chamlin SL, Mattson CL, Frieden IJ, Williams ML, Mancini AJ, Cella D, et al. The price of pruritus: sleep disturbance and cosleeping in atopic dermatitis. Arch Pediatr Adolesc Med. 2005;159:745-50.
- 28. Ben-Gashir MA. Relationship between quality of life and disease severity in atopic dermatitis/eczema syndrome during childhood. Curr Opin Allergy Clin Immunol. 2003;3:369-73.
- 29. Ben-Gashir MA, Seed PT, Hay RJ. Quality of life and disease severity are correlated in children with atopic dermatitis. Br J Dermatol. 2004;150:284-90.
- 30. Su JC, Kemp AS, Varigos GA, Nolan TM. Atopic Eczema: its impact on the family and financial cost. Arch Dis Child. 1997;76:159-62.
Correspondência:
Publication Dates
-
Publication in this collection
28 Oct 2009 -
Date of issue
Oct 2009
History
-
Accepted
17 June 2009 -
Received
29 Dec 2008