Abstracts
OBJECTIVE: To assess the effect of prone and supine positions on breathing pattern variables, thoracoabdominal motion and peripheral oxygen saturation of hemoglobin of premature newborn infants recovering from respiratory distress syndrome, while breathing spontaneously and in rapid eye movement sleep. METHODS: This was a quasi-experimental study. Twelve preterms weighing > 1,000 g at enrollment were studied in both positions, in random order. Respiratory inductive plethysmography was used to analyze breathing pattern (tidal volume, respiratory rate, minute ventilation, mean inspiratory flow) and thoracoabdominal motion (labored breathing index, phase relation in inspiration, phase relation in expiration, phase relation in total breath and phase angle). Pulse oximetry was used to evaluate peripheral oxygen saturation. Student's t test for paired samples or the Wilcoxon test were used for statistical analysis. Significance was set at p < 0.05. RESULTS: A total of 9,167 respiratory cycles were analyzed. The prone position was associated with significant reductions in labored breathing index (-0.84±0.69; p = 0.001; 95%CI -1.29 to -0.40), phase relation in inspiration (-27.36±17.55; p = 0.000; 95%CI -38.51 to -16.20), phase relation in expiration (-32.36±16.20; p = 0.000; 95%CI -42.65 to -22.06) and phase relation in total breath (-30.20±14.76; p = 0.000; 95%CI -39.59 to -20.82). There were no significant differences between the two positions in any of the other variables analyzed. CONCLUSION: The prone position resulted in a significant reduction in thoracoabdominal asynchrony, without affecting breathing pattern or peripheral oxygen saturation.
Prone position; supine position; premature newborn infants; respiratory distress syndrome
OBJETIVO: Avaliar a influência das posições prona e supina em recém-nascidos prematuros pós-síndrome do desconforto respiratório, respirando espontaneamente e em estado de sono ativo, sobre variáveis de padrão respiratório, movimento toracoabdominal e saturação periférica da hemoglobina pelo oxigênio. MÉTODOS: Estudo quase experimental. Doze prematuros com peso > 1.000 g no momento do estudo foram estudados nas duas posições, em ordem randomizada. A pletismografia respiratória por indutância foi utilizada para avaliação do padrão respiratório (volume corrente, frequência respiratória, ventilação minuto, fluxo inspiratório médio) e do movimento toracoabdominal (índice de trabalho respiratório, relação de fase inspiratória, relação de fase expiratória, relação de fase respiratória total e ângulo de fase). A oximetria de pulso registrou a saturação periférica de oxigênio. Para a análise estatística foram realizados os testes t de Student para amostras pareadas ou Wilcoxon. Foi considerado significativo p < 0,05. RESULTADOS: Foram analisados 9.167 ciclos respiratórios. Na posição prona, houve redução significativa do índice de trabalho respiratório (-0,84±0,69; p = 0,001; IC95% -1,29 a -0,40), das relações de fase inspiratória (-27,36±17,55; p = 0,000; IC95% -38,51 a -16,20), expiratória (-32,36±16,20; p = 0,000; IC95% -42,65 a -22,06) e total (-30,20±14,76; p = 0,000; IC95% -39,59 a -20,82). Não houve diferença significativa entre as posições nas demais variáveis analisadas. CONCLUSÃO: A posição prona promoveu diminuição significativa da assincronia toracoabdominal, sem influenciar o padrão respiratório e a saturação periférica de oxigênio.
Decúbito ventral; decúbito dorsal; recém-nascido prematuro; síndrome do desconforto respiratório
ORIGINAL ARTICLE
Prone position and reduced thoracoabdominal asynchrony in preterm newborns
Trícia G. OliveiraI; Maria A. S. RegoII; Nadja C. PereiraIII; Lorena O. VazIV; Danielle C. FrançaI; Danielle S. R. VieiraI; Verônica F. ParreiraV
IMestre, Ciências da Reabilitação, Universidade Federal de Minas Gerais (UFMG), Belo Horizonte, MG, Brazil
IIDoutora, Ciências da Saúde. Professora adjunta, Departamento de Pediatria, UFMG, Belo Horizonte, MG, Brazil
IIIEngenheira de Controle e Automação, UFMG, Belo Horizonte, MG, Brazil
IVAcadêmica de Fisioterapia, UFMG, Belo Horizonte, MG, Brazil. Bolsista, Programa Institucional de Bolsas de Iniciação Científica Conselho Nacional de Desenvolvimento Científico e Tecnológico (PIBIC-CNPq)
VDoutora, Fisioterapia e Reabilitação. Professora associada, Departamento de Fisioterapia, UFMG, Belo Horizonte, MG, Brazil
Correspondence
ABSTRACT
OBJECTIVE: To assess the effect of prone and supine positions on breathing pattern variables, thoracoabdominal motion and peripheral oxygen saturation of hemoglobin of premature newborn infants recovering from respiratory distress syndrome, while breathing spontaneously and in rapid eye movement sleep.
METHODS: This was a quasi-experimental study. Twelve preterms weighing > 1,000 g at enrollment were studied in both positions, in random order. Respiratory inductive plethysmography was used to analyze breathing pattern (tidal volume, respiratory rate, minute ventilation, mean inspiratory flow) and thoracoabdominal motion (labored breathing index, phase relation in inspiration, phase relation in expiration, phase relation in total breath and phase angle). Pulse oximetry was used to evaluate peripheral oxygen saturation. Student's t test for paired samples or the Wilcoxon test were used for statistical analysis. Significance was set at p < 0.05.
RESULTS: A total of 9,167 respiratory cycles were analyzed. The prone position was associated with significant reductions in labored breathing index (-0.84±0.69; p = 0.001; 95%CI -1.29 to -0.40), phase relation in inspiration (-27.36±17.55; p = 0.000; 95%CI -38.51 to -16.20), phase relation in expiration (-32.36±16.20; p = 0.000; 95%CI -42.65 to -22.06) and phase relation in total breath (-30.20±14.76; p = 0.000; 95%CI -39.59 to -20.82). There were no significant differences between the two positions in any of the other variables analyzed.
CONCLUSION: The prone position resulted in a significant reduction in thoracoabdominal asynchrony, without affecting breathing pattern or peripheral oxygen saturation.
Keywords: Prone position, supine position, premature newborn infants, respiratory distress syndrome.
Introduction
In newborn infants, the configuration and complacency of the chest wall puts the respiratory system at a clear mechanical disadvantage with relation to adults.1 The more horizontal position of the ribs makes the chest wall more rounded, rather than elliptical as is seen in adults, which changes the ratio between length and tension of rib cage muscles.2 The reduction in diaphragmatic apposition compromises the muscle's capacity to generate force, which in turn compromises chest wall stability, resulting in distortions during respiratory movement.3 With relation to complacency, ribs have a low mineral content, offering less stability against various different distorting forces to which the chest wall is subjected.1,2 This can be observed in asynchronous respiratory movement, which involves increased diaphragmatic work, respiratory work and energy expenditure.4
The sleep state known as rapid eye movement (REM), which is described as eyes closed, irregular breathing, and small movements, and in which preterm newborns spend 90% of their time asleep, has an effect on respiratory system work.5
The combination of factors described above can result in paradoxical chest wall motion, reducing even further the efficiency of diaphragm movement and increasing its work, making it incapable of responding to increases in ventilatory demand.1,3,6
The literature indicates that the prone position offers certain benefits when compared with the supine position: increased peripheral oxygen saturation of hemoglobin (SpO2),7,8 increased tidal volume (Vt),9,10 reduction in the number of central apnea events,11 reduced energy expenditure,4 improved thoracoabdominal synchronization,12-14 reduced need for reintubation of preterm newborn infants after weaning from mechanical ventilation15 and a reduction in the number of episodes of gastroesophageal reflux.16 However, the prone position is strongly associated with sudden infant death syndrome and is not recommended for healthy full term newborns.17 Other studies have found no differences in SpO215,18 and in Vt19 between the two positions.
Studies that evaluated respiratory parameters analyzed a very low number of respiratory cycles,20 or recorded for a short period,12-14 or did not present detailed methodological designs.21,22 Therefore, questions remain about the true benefits of prone and supine positions for full term and preterm newborns.8,15,23
In this background, the objective of this study was to assess the influence of prone and supine positions on breathing pattern variables, thoracoabdominal motion and SpO2 in preterm newborn infants recovering from respiratory distress syndrome, breathing spontaneously and in REM sleep.
Materials and methods
Sample
The sample size was calculated for a statistical power of 80% and a significance level of 0.05, after a pilot study with nine newborn infants, resulting in n = 5 for Vt, n = 3 for labored breathing index (LBI) and n = 12 for phase angle (PhAng). Therefore, 12 preterm newborn infants were recruited for this quasi-experimental study from the neonatal intensive care unit at a University Hospital. The inclusion criteria were: gestational age at birth between 28 and 36 weeks; free from congenital malformations, such as gastroschisis, omphalocele and/or myelomeningocele; free from clinical or surgical conditions that would prevent positioning from being studied; clinically stable, defined as spontaneous breathing, on room air or oxygen therapy with fraction of inspired oxygen under 0.40 for a period higher than 72 hours; recovering from respiratory distress syndrome, defined by radiological (ground glass sign) and clinical criteria (tachypnea, retraction of the rib cage, moaning and cyanosis); and weight above 1,000 g at the time of enrollment. The exclusion criterion was any factor preventing the infant being put in either position. The study was approved by Research Ethics Committee at the institution (ETIC 381/07), and all parents signed informed consent forms.
Measurement instruments
Respiratory inductive plethysmography (RIP) was used to measure breathing pattern and thoracoabdominal motion variables. In 1978, M. Cohn introduced RIP, which is an instrument that makes it possible to take measurements non-invasively based on principles described by Konno & Mead in 1967.24 This technique studies pulmonary ventilation based on rib cage and abdomen cross-sectional area changes, captured by transducers inside Teflon bands that are positioned around the circumference of the rib cage, the axilla and the circumference of the abdomen, at the height of the umbilical scar.24 These transducers are connected to the plethysmograph which captures the signals corresponding to rib cage and abdomen and the sum of the two displacements, i.e., Vt. This scalar trace is converted by software into a Lissajous figure, in the case of plethysmography, and into a Konno-Mead curve (x-axis-y), for analysis of volume and time breathing pattern components and thoracoabdominal motion.25 The apparatus was calibrated automatically by Qualitative Diagnostic Calibration (QDC)13,26 using an estimated Vt of 6 to 8 mL/kg, as described by Wilkes et al.26
Pulse oximetry was used to measure SpO2 (Dixtal Oxypleth®, DX2405, Manaus, Brazil).
Variables analyzed
The following variables were analyzed for each respiratory cycle, on a cycle-by-cycle basis: Vt, respiratory rate (f), minute ventilation (VE), mean inspiratory flow (Vt/Ti corresponds to the ratio between tidal volume and inspiratory time), LBI, PhAng, phase relation in inspiration (PhRIB expresses the percentage of direction in which rib cage and the abdomen move in opposite directions during inspiration), phase relation in expiration (PhREB expresses the percentage of direction in which rib cage and the abdomen move in opposite directions during expiration) and phase relation in total breath (PhRTB expresses the percentage of direction in which rib cage and the abdomen move in opposite directions during the respiratory cycle). Percentages of 0% indicate perfect synchronization; 100% indicates total asynchrony. All variables were analyzed during REM sleep, according to behavioral observation described by Prechtl.5 RespiEvents 5.2® (NIMS, Miami, United States) software was used to analyze breathing pattern variables and LBI, while MatLab® (MathWorks Inc., Natick, United States) was used to analyze all other thoracoabdominal motion variables. SpO2 was analyzed continuously and recorded every 5 minutes on a dedicated chart.
Procedures
After parents had signed the consent form, newborn infants data were extracted from medical records. Initial positions were decided by drawing lots in sealed envelopes, 60 minutes after feeding. The pulse oximeter was then fitted, followed by Teflon bands, of an appropriate size for preterms, on the chest and abdomen, at the axilla and at the level of the umbilical scar, respectively. Soon after, the newborn was placed in the first position and the sensors connected to the cables from the apparatus. Vital signs were recorded and sleep state was determined by behavioral observation. The apparatus was switched on at this point, entering calibration mode, which lasts about 5 minutes. After calibration, data were recorded for 30 minutes in the first position. Soon after saving the data collected, the apparatus was switched off, the cables disconnected and the newborn placed in the second position. The calibration and data collection phases were then repeated.
In the prone position, the preterms were positioned on a cloth roll and surrounded by another roll, in order to guarantee semi-flexion of the hips and knees. In the supine position, the newborn infants were lain down with a roll of cloth surrounding them and another behind the knees, once more to guarantee semi-flexion of the hips and knees.14 In order to reduce the chance of neck position influencing the parameters assessed, the infants' heads were turned to the right in both positions.27
Statistical analysis
Data are presented as measures of central tendency and dispersion. The Shapiro-Wilk test was used to test each variable for normality. Variables with normal distribution were compared using Student's t test for paired samples and the Wilcoxon test was used for the remaining variables. The significance level adopted was α de 0.05.28 Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS, Chicago, United States) version 13.0.
Results
Fifteen newborn infants were eligible for the study between November of 2007 and July of 2008. Two newborn infants were transferred to the Kangaroo care ward before data could be collected, and informed consent was not granted for a third. Therefore, 12 premature newborn infants were studied. There are no breathing pattern data available for neonates number 1 in supine position, because the apparatus had not been calibrated for this position and these variables are dependent of volume calibration.
All of these infants received a dose of exogenous surfactant during mechanical ventilation; after extubation, all received methylxanthines. The infants were studied between their 9th and 20th day of life, when they were already clinically stable, in conformity with the inclusion criteria. During the study the preterm newborn infants were in incubators with the temperature set according to their requirements.
A total of 9,167 respiratory cycles were analyzed, 6,349 in prone position and 2,818 in supine position. Out of 30 minutes of registered traces in each position, all periods that included more than 10 consecutive acceptable respiratory cycles were analyzed. The mean number of cycles analyzed per newborn was 819±541.
Table 1 lists demographic, anthropometric data and other characteristics of the 12 newborn infants studied.
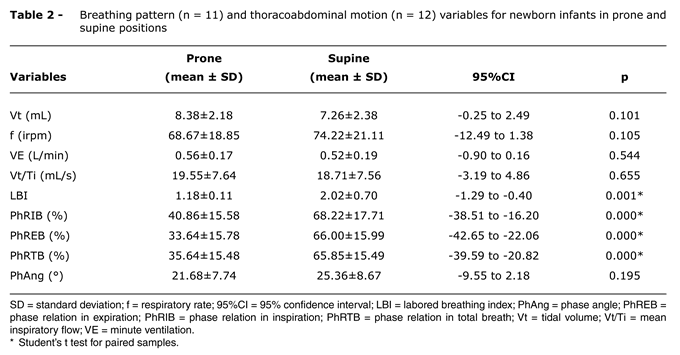
Table 2 shows breathing pattern and thoracoabdominal motion data of newborn infants in prone and supine positions. No significant difference was observed in any of the breathing pattern variables (Vt, f, VE and Vt/Ti; p > 0.05). A significant reduction was observed in thoracoabdominal motion variables in prone position (LBI, PhRIB, PhREB, and PhRTB), with the exception of PhAng (p > 0.05).
With relation to SpO2, no significant difference was observed between the prone (95.10±0.65%) and supine positions (93.44±0.74%); p = 0.084.
Discussion
The main results of this study demonstrate that prone position significantly reduced the values of LBI, PhRIB, PhREB and PhRTB in relation to supine position; having a positive effect on thoracoabdominal motion without altering PhAng, breathing pattern variables or SpO2.
In our study, in prone position there was a significant reduction in LBI scores while in REM sleep, in comparison with supine position, in agreement with a study published by Adams et al.13 According to those authors, the greater the rib cage distortion, the better the results when this is stabilized, reflecting thoracoabdominal coordination improvement.
This coordination can also be evaluated using the phase relations (PhRIB, PhREB and PhRTB), which have been recorded previously for healthy adults. As far as we are aware, there have not yet been any studies evaluating these variables in premature infants. The significant reduction in PhRIB, PhREB and PhRTB values when in prone position indicates a positive influence of this position on thoracoabdominal coordination. According to Wolfson et al.,12 this finding is related to mechanical changes to chest wall. According to this theory, the mechanisms involved include, foremost, the direct effect of gravity on the diaphragm, increasing its area of apposition to the anterior chest wall and, therefore, the mechanical efficiency of the muscle. Added to this, the axial displacement of the rib cage, also due to the direct action of gravity, increases the passive tension of the rib cage muscles, helping to stabilize the anterior chest wall during inspiration.
Some studies have speculated that the prone position, by increasing abdominal pressure, may increase the area of apposition of the diaphragm, contributing to stabilization of the lower rib cage.1 However, no studies have tested this hypothesis.
Our findings with relation to breathing pattern variables are comparable with the results of some earlier studies12,19 which also failed to detect significant differences between the prone and supine positions. However, other studies have observed significant increases in Vt and a significant reduction in f in the prone position.9,10 It is possible that this difference in results is related to the influence of mechanical ventilation and the oxygen dependency of the premature infants included in those studies,10 In contrast with our newborn infants, who were breathing spontaneously and were not oxygen-dependent. No significant difference was observed in terms of Vt/Ti among positions, thereby indicating that stimulation of the respiratory center did not differ. A study undertaken by Adams et al.,13 with full term newborns found a significant increase in this parameter in prone position, with a significant increase also in Vt with no change in Ti.
In this study no significant difference was observed in SpO2 levels as a result of positioning. This is in agreement with the findings of some authors,14,15,18 but contrasts with the results observed in other studies, which have demonstrated significant improvements in SpO2 in preterm newborn infants put in the prone position.17,8,19 The samples in those studies were composed of newborn infants on mechanical ventilation, or, in the majority, of oxygen-dependent newborn infants. In a meta-analysis conducted by Wells et al.,29 all eight studies analyzed detected a significant increase in this variable; however, they stated that these studies were heterogeneous in terms of methodology, concluding that there was evidence for short-term improvements in SpO2 among preterm newborn infants on mechanical ventilation put in the prone position, as is also supported by the systematic review conducted by Balaguer et al.23
The thoracoabdominal motion variable most cited in the literature is PhAng.12,14,22 In the majority of studies this angle is calculated from the ratio between the excursion of the abdomen at mid-excursion of the rib cage and maximum excursion of the abdomen. This analysis is traditionally based on evaluation of sine waves producing elliptical curves. Since the chest wall movements of newborns are not perfectly sinusoidal and the Lissajous figures that correspond to them are not perfectly elliptical, the term PhAng can be considered to be an approximation. For this reason we decided to employ another method to calculate this variable, which is independent of morphology. Calculating from the inverse tangent of the regression curve is independent of the relative or absolute contributions of the rib cage and abdomen signals, providing quantitative information on thoracoabdominal asynchrony.14 In our study, there was no significant difference in PhAng in relation to position. Our findings are in agreement with results published by Levy et al.,22 who investigated PhAng and respiratory work indirectly using RIP and esophageal manometry, demonstrating that there was no significant difference between the two positions in terms of these two variables.
Certain methodological aspects deserve to be mentioned. Earlier studies that have analyzed the influence of position on the respiratory function of preterm newborn infants have chosen to assess a specific number of respiratory cycles. Allen et al.20 only examined consistently reproducible respiratory cycles, during a non-REM phase of sleep; Wolfson et al.12 analyzed a minimum of 10 respiratory cycles, also in non-REM sleep, determined by behavioral observation.12,20 Maynard et al.14 studied 20 minutes' records, irrespective of the neonate's behavioral state, without, however, reporting the number of respiratory cycles analyzed. In our study, 30 minutes' records were analyzed for each position with a minimum of 10 consecutive acceptable respiratory cycles. Taking both positions, 9,167 respiratory cycles were analyzed. As far as we know, this is the analysis of the largest number of cycles reported in the literature to date. It should be emphasized that the number of cycles analyzed in the prone position was 225% greater than in the supine position. Taking into consideration that the selection criteria for cycles was a sequence of at least 10 consecutive cycles in a stable trace, it could be speculated that the prone position resulted in a more stable trace, reflecting increased stability of breathing pattern and thoracoabdominal motion. No episodes of apnea were observed during data collection in either position. Despite the period of data collection, we take a risk to hypothesize that the mechanism involved in the association between sudden infant death syndrome and prone position does not involve thoracoabdominal motion, rather other factors which still need to be better elucidated.
Another methodological issue is automatic calibration by QDC in each position, despite earlier studies having shown that calibration in just one position should be sufficient, as long as the neonate's behavioral state is maintained.13 We considered that this could be a source of error, since our population was made up of preterm newborn infants who changed behavioral state during the study period. It is also worth pointing out that the sequence of positions was randomized, with the intention of avoiding superimposition of the effect of one position on the other. Another precaution was the exclusion of extremely premature infants because there are no studies demonstrating the accuracy of RIP for measuring volumes in this subpopulation.
In conclusion, the results of this study suggest that the prone position improved thoracoabdominal synchrony without affecting breathing pattern or SpO2.
References
- 1. Heldt GP, McIlroy MB. Dynamics of chest wall in preterm infants. J Appl Physiol. 1987;62:170-4.
- 2. Mortola JP, Saetta M, Fox G, Smith B, Weeks S. Mechanical aspects of chest wall distortion. J Appl.Physiol. 1985;59:295-304.
- 3. Papastamelos C, Panitch HB, England SE, Allen JL. Developmental changes in chest wall compliance in infancy and early childhood. J Appl Physiol. 1995;78:179-84.
- 4. Masterson J, Zucker C, Schulze K. Prone and supine positioning effects on energy expenditure and behavior of low birth weight neonates. Pediatrics. 1987;80:689-92.
- 5. Prechtl HF. The behavioural states of the newborn infant (a review). Brain Res. 1974;76:185-212.
- 6. Fleming PJ, Muller NL, Bryan MH, Bryan AC. The effects of abdominal loading on rib cage distortion in premature infants. Pediatrics. 1979;64:425-8.
- 7. Chang YJ, Anderson GC, Dowling D, Lin CH. Decreased activity and oxygen desaturation in prone ventilated preterm infants during the first postnatal week. Heart Lung. 2002;31:34-42.
- 8. Kassim Z, Donaldson N, Khetriwal B, Rao H, Sylvester K, Rafferty GF et al. Sleeping position, oxygen saturation and lung volume in convalescent, prematurely born infants. Arch.Dis.Child Fetal Neonatal Ed. 2007;92:F347-50.
- 9. Hutchison AA, Ross KR, Russell G. The effect of posture on ventilation and lung mechanics in preterm and light-for-date infants. Pediatrics. 1979;64:429-32.
- 10. Leipala JA, Bhat RY, Rafferty GF, Hannam S, Greenough A. Effect of posture on respiratory function and drive in preterm infants prior to discharge. Pediatr Pulmonol. 2003;36:295-300.
- 11. Heimler R, Langlois J, Hodel DJ, Nelin LD, Sasidharan P. Effect of positioning on the breathing pattern of preterm infants. Arch Dis Child. 1992;67:312-4.
- 12. Wolfson MR, Greenspan JS, Deoras KS, Allen JL, Shaffer TH. Effect of position on the mechanical interaction between the rib cage and abdomen in preterm infants. J Appl Physiol. 1992;72:1032-8.
- 13. Adams JA, Zabaleta IA, Sackner MA. Comparison of supine and prone noninvasive measurements of breathing patterns in fullterm newborns. Pediatr Pulmonol. 1994;18:8-12.
- 14. Maynard V, Bignall S, Kitchen S. Effect of positioning on respiratory synchrony in non-ventilated pre-term infants. Physiother Res Int. 2000;5:96-110.
- 15. Antunes LC, Rugolo LM, Crocci AJ. Efeito da posição do prematuro no desmame da ventilação mecânica. J Pediatr (Rio J). 2003;79:239-44.
- 16. Mezzacapa MA, Goulart LM, Brunelli MM. Influência dos decúbitos dorsal e ventral na monitorização do pH esofágico em recém-nascidos de muito baixo peso. Arq Gastroenterol. 2004;41:42-8.
- 17. American Academy of Pediatrics AAP Task Force on Infant Positioning and SIDS: Positioning and SIDS. Pediatrics. 1992;89:1120-6.
- 18. Elder DE, Campbell AJ, Doherty DA. Prone or supine for infants with chronic lung disease at neonatal discharge? J Paediatr Child Health. 2005;41:180-5.
- 19. Mendoza JC, Roberts JL, Cook LN. Postural effects on pulmonary function and heart rate of preterm infants with lung disease. J Pediatr. 1991;118:445-8.
- 20. Allen JL, Wolfson MR, McDowell K, Shaffer TH. Thoracoabdominal asynchrony in infants with airflow obstruction. Am Rev Respir Dis. 1990;141:337-42.
- 21. Mizuno K, Aizawa M. Effects of body position on blood gases and lung mechanics of infants with chronic lung disease during tube feeding. Pediatr Int. 1999;41:609-14.
- 22. Levy J, Habib RH, Liptsen E, Singh R, Kahn D, Steele AM et al. Prone versus supine positioning in the well preterm infant: effects on work of breathing and breathing patterns. Pediatr Pulmonol. 2006;41:754-8.
- 23. Balaguer A, Escribano J, Roque M. Infant position in neonates receiving mechanical ventilation. Cochrane Database Syst Rev. 2006;(4):CD003668.
- 24. Konno K, Mead J. Measurement of the separate volume changes of rib cage and abdomen during breathing. J Appl Physiol. 1967;22:407-22.
- 25. Chadha TS, Watson H, Birch S, Jenouri GA, Schneider AW, Cohn MA, et al. Validation of respiratory inductive plethysmography using different calibration procedures. Am Rev Respir Dis. 1982;125:644-9.
- 26. Wilkes DL, Revow M, Bryan MH, England SJ. Evaluation of respiratory inductive plethysmography in infants weighing less than 1,500 grams. Am Rev Respir Dis. 1987;136:416-9.
- 27. Downs JA, Stocks J. Effect of neck rotation on the timing and pattern of infant tidal breathing. Pediatr Pulmonol. 1995;20:380-6.
- 28. Munro BH. Statistics methods for health care research. Philadelphia, PA: Lippincott Williams & Wilkins, 2005.
- 29. Wells DA, Gillies D, Fitzgerald DA. Positioning for acute respiratory distress in hospitalised infants and children. Cochrane Database Syst Rev. 2005;(2):CD003645.
Correspondência:
Publication Dates
-
Publication in this collection
28 Oct 2009 -
Date of issue
Oct 2009
History
-
Accepted
19 June 2009 -
Received
22 Apr 2009