Abstracts
OBJECTIVES: To determine the prevalence of metal contact allergy among the children seen at a health center and to characterize children with metal allergies in terms of risk factors. METHODS: This was an uncontrolled cross-sectional study undertaken at a health center in Belo Horizonte, Brazil. Children aged from 0 to 12 years were recruited when they presented at the health center for routine pediatric consultations and were given contact tests for chrome, cobalt and nickel. Statistical analyses were conducted on test readings taken at 96 hours. Results classed as weak (+), strong (++) or extreme (+++) were defined as "reaction," while those classed as doubtful, negative or irritant were defined as "no reaction." RESULTS: A total of 144 children completed the study protocol. Of these, 4.9% exhibited a reaction to chrome, 9.7% to cobalt and 20.1% to nickel. Patients with pierced ears were more likely to react to nickel than those without pierced ears (p = 0.031 and odds ratio = 2.8). CONCLUSIONS: In view of the current tendency for the prevalence of nickel allergy to increase, parents should be warned about its association with ear piercing. Further studies are needed to determine the ideal age for ear piercing and the ideal materials for earrings.
Cobalt; child; chrome; contact dermatitis; hypersensitivity; nickel
OBJETIVOS: Determinar a prevalência da alergia de contato aos metais em crianças que frequentam um centro de saúde e caracterizar o subgrupo com alergia em relação aos fatores de risco. MÉTODOS: Estudo transversal não controlado, conduzido em um centro de saúde de Belo Horizonte (MG). Foram incluídas crianças com idades entre 0 e 12 anos que se apresentaram para consulta pediátrica de rotina, sendo aplicados testes de contato para o cromo, o cobalto e o níquel. As análises estatísticas foram realizadas com base na leitura do teste em 96 horas. Leituras classificadas como fraca (+), forte (++) ou extrema (+++) foram consideradas como reação, enquanto as classificadas como duvidosa, negativa ou irritativa foram consideradas como não reação. RESULTADOS: Completaram o estudo 144 crianças. Destas, 4,9% apresentaram reação ao cromo, 9,7% ao cobalto e 20,1% ao níquel. Os pacientes com orelha perfurada tiveram mais chance de reação ao níquel do que aqueles sem essa característica (p = 0,031 e odds ratio = 2,8). CONCLUSÕES: Em face da tendência atual ao aumento da alergia ao níquel, familiares devem ser alertados sobre a sua associação com a perfuração das orelhas. Estudos posteriores são necessários para avaliar a idade ideal para a perfuração das orelhas e o material ideal para brincos.
Cobalto; criança; cromo; dermatite de contato; hipersensibilidade; níquel
ORIGINAL ARTICLE
Ear piercing as a risk factor for contact allergy to nickel
Marilda H. T. BrandãoI;Bernardo GontijoII;Marcela A. GirundiIII;Maria C. M. de CastroIII
IMédica especialista, Pediatria e Dermatologia. Mestre, Ciências da Saúde, Saúde da Criança e do Adolescente, Hospital Infantil João Paulo II, Fundação Hospitalar do Estado de Minas Gerais (FHEMIG), Belo Horizonte, MG, Brazil
IIDoutor, Dermatologia, Faculdade de Medicina, Universidade Federal de Minas Gerais (UFMG), Belo Horizonte, MG, Brazil. Professor associado, Faculdade de Medicina, UFMG, Belo Horizonte, MG, Brazil
IIIAcadêmica, Medicina, UFMG, Belo Horizonte, MG, Brazil
ABSTRACT
OBJECTIVES: To determine the prevalence of metal contact allergy among the children seen at a health center and to characterize children with metal allergies in terms of risk factors.
METHODS: This was an uncontrolled cross-sectional study undertaken at a health center in Belo Horizonte, Brazil. Children aged from 0 to 12 years were recruited when they presented at the health center for routine pediatric consultations and were given contact tests for chrome, cobalt and nickel. Statistical analyses were conducted on test readings taken at 96 hours. Results classed as weak (+), strong (++) or extreme (+++) were defined as "reaction," while those classed as doubtful, negative or irritant were defined as "no reaction."
RESULTS: A total of 144 children completed the study protocol. Of these, 4.9% exhibited a reaction to chrome, 9.7% to cobalt and 20.1% to nickel. Patients with pierced ears were more likely to react to nickel than those without pierced ears (p = 0.031 and odds ratio = 2.8).
CONCLUSIONS: In view of the current tendency for the prevalence of nickel allergy to increase, parents should be warned about its association with ear piercing. Further studies are needed to determine the ideal age for ear piercing and the ideal materials for earrings.
Keywords: Cobalt, child, chrome, contact dermatitis, hypersensitivity, nickel.
Introduction
Several different studies have shown that metals are the most common contact sensitizers in children, particularly to nickel.1-4 Recent data have shown that the prevalence of nickel allergy has increased in industrialized nations.5 Fashions and lifestyle exert a considerable influence over emergence of this sensitization. Nickel contact dermatitis was originally considered to be an occupational dermatosis, but began to affect the general population when the metal began to be used in the manufacture of products such as zippers, suspenders and jewelry.5 It is now known that wearing jewelry, particularly earrings fitted at an early age, is associated with increased nickel sensitivity.6,7 Sensitization may take place at any age, including in newborn infants,8 and generally has a negative effect on patients' lives, including in terms of occupational opportunities.
The proportion of reactivity varies in accordance with the way in which the population tested is selected. One meta-analysis observed contact allergy prevalence rates among the general pediatric population of 8.3% for nickel, 1.9% for cobalt and 1.5% for chrome.9
Once sensitization has been detected, patients are advised to avoid contact with objects that contain the metals to which they are sensitive. However, many children and adolescents find it difficult to follow this guidance since nickel, the metal to which sensitization most frequently occurs, is used in many different products, including fashion clothing.
Reviewing the literature, we observed that few studies have investigated contact sensitization in unselected (healthy) pediatric populations. Published results from children are from patients with suspected contact dermatitis. The fact that a contact test reveals sensitivity to a given allergen does not necessarily mean that contact dermatitis is present.10
A search of the literature did not locate any descriptions of contact sensitization in asymptomatic Brazilian children. The primary objective of this study was to determine the prevalence of metal contact allergy among children seen at a health center. A secondary objective is to characterize the subset who have metal allergies in terms of risk factors.
Patients and methods
Study design
This was an uncontrolled cross-sectional study undertaken at a health center in Belo Horizonte, MG, Brazil between February and September of 2008. All patients had their histories taken, underwent a dermatological examination including a questionnaire and had contact tests for chrome, cobalt and nickel administered The questionnaire included questions on age, sex, color, personal atopic diseases, family atopic disease, personal and family allergic contact dermatitis, ear piercing, age and number of piercings, wearing of dental appliances and age first fitted, wearing of spectacles containing metals and wearing of jewelry. The study protocol was approved by the Research Ethics Committee (Comitê de Ética em Pesquisa, COEP) at the Universidade Federal de Minas Gerais (UFMG) in Belo Horizonte, Brazil, under hearing number ETIC 483/06. All patients and guardians received a written explanation of the study in the form of a free and informed consent form which they were invited to read and analyze. Patients aged 7 to 12 years signed the consent form together with their guardians, as mandated by COEP. Interviewee anonymity was preserved throughout analysis and publication of the results.
Study population
We recruited children aged from 0 to 12 years who presented at the health center of Cachoeirinha, Brazil, for pediatric consultations and whose guardians gave permission to participate. Children were excluded if they had active cases of dermatitis on their backs (which is where the test would be performed), if they had infectious febrile diseases (since there is a chance that exanthema could interfere with interpretation of the results), if they had taken systemic corticoids during the previous month, had immunodeficiency diseases or had suffered intense exposure to the sun during the previous 15 days. The Cachoeirinha health center is part of Belo Horizonte's northeast healthcare district and has a population of 274,060 inhabitants; 42.74% of whom have a monthly income of between one and three times the minimum monthly wage.11
Contact tests
All patients enrolled on the study were tested with 5% nickel sulphate in solid petroleum jelly, 1% cobalt chloride in solid petroleum jelly and 0.5% potassium dichromate in solid petroleum jelly (FDA Allergenic, Rio de Janeiro, Brazil). These mixtures were applied to the upper back between the shoulder blades in Finn Chambers on Scanpor tape® (Epitest Ltd Oy, Tuusula, Finland), and also with Micropore 3M® hypoallergenic porous tape (3M do Brasil Ltda, Sumaré, Brazil). The tests were removed after 48 hours, when the first reading was taken. A second reading was taken after 96 hours. All readings were taken by a single examiner. Reactions to the tests were graded according to the criteria adopted by the International Contact Dermatitis Research Group (ICDRG), which are identical to those recommended by the Brazilian contact dermatitis research group (GBEDC - Grupo Brasileiro de Estudos em Dermatite de Contato). The grades are as follows: - = no cutaneous changes; + = weak reaction (erythema and non-vesicular infiltration), ++ = strong reaction (vesicular); +++ = extreme reaction (with blisters or ulceration); ?+ = doubtful reaction (erythema without infiltration); and IR = irritant reaction.12,13
Statistics
It is estimated that 8.3% of the pediatric population is sensitized to nickel.9 Since this is the most prevalent sensitization, we used this figure for the sample size calculation. Adopting a margin of error of 5% and a 95% confidence level, the minimum sample size was 138 patients.14 Since losses were to be expected, 162 children were enrolled on the study.
Descriptive results for qualitative variables are presented as frequencies and percentages and quantitative variables as measures of central tendency (mean and median) and distribution [standard deviation (SD)]. The readings taken after 96 hours were used for analysis and the reaction classes were grouped together to form two categories, with "reaction" covering weak (+), strong (++) and extreme (+++), and "no reaction" comprising doubtful, negative and irritant.
Qualitative variables were compared with the reaction tests results using contingency tables and Pearson's chi-square test was used to compare proportions. If expected frequency was less than five, Fisher's exact test was used instead. The reference category is indicated in the results tables by a figure of 1.0 in the odds ratio (OR) column. It is important to point out that categories that did not exhibit any observations, and patients who did not provide a reply were excluded from the comparisons.
When the standard assumptions of Student's t test (normality and homogeneity of variance) were met it was used to compare reaction results and qualitative covariables. If these conditions were not met, the Mann-Whitney test was used. The Kolmogorov-Smirnov test was used to test normality and the Levene test was used to test homogeneity of variance.15
Logistic regression models were built by starting with all covariables that had p < 0.25 in the univariate analysis, i.e. those that at least exhibited a tendency towards statistical significance. Variables were then removed step-by-step until only those with statistical significance (p < 0.05) and with clinical significance were left. The Hosmer-Lemeshow test was used to test the final model's goodness of fit. The multivariate analysis was performed using the public-domain software package R.
Results
Descriptive analysis of the sample
A total of 162 children were tested and 144 of them completed the study protocol. Sixty-six (45.8%) of them were male and 78 (54.2%) were female. The remaining 18 children were lost to follow-up or had removed the tests before 48 hours had elapsed. Mean age in months was 64.78 with an SD of 42.11 and a median of 152.76. Mean age in months at time of ear piercing was 9.97 with an SD of 23.92 and a median of 2.00. Seven (4.9%) children were positive for a reaction to chrome, 14 (9.7%) reacted to cobalt and 29 (20.1%) to nickel. The reaction test results are presented in Table 1.
Univariate and multivariate analysis
Factors associated with a reaction to chrome
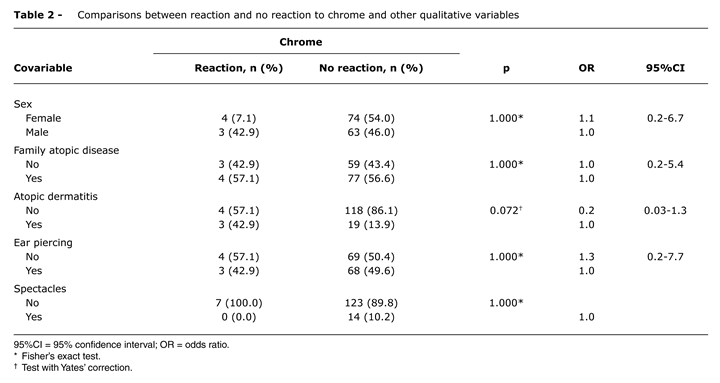
The comparisons between the chrome test results and other variables of interest are given in Table 2. It will be observed that children without atopic dermatitis were less likely to react to chrome than those with atopic dermatitis (p = 0.072 and OR = 0.2). There was no difference in age between those who reacted and those who did not (p = 0.791). The mean age in months of children who did react was 71.9 with an SD of 58.4 and a median of 90.8.
Factors associated with reaction to cobalt
The comparisons between cobalt test results and other variables of interest are given in Table 3. It will be observed that children without family atopic disease were more likely to react to cobalt than those whose families had atopic disease (p = 0.050 and OR = 3.7), although without statistical significance; since there are no indications of clinical significance between cobalt allergy and this covariable. There was no difference in age between those who reacted and those who did not (p = 0.432). The mean age in months of children who did react to cobalt was 56.6 with an SD of 47.9 and a median of 39.0. The covariables family atopic disease and ear piercing had p < 0.25 and were included in the multivariate model. It was observed that these covariables in conjunction were not associated with reaction to cobalt.
Factors associated with reaction to nickel
The comparisons between nickel test results and other variables of interest are given in Table 4. It will be observed that children with pierced ears were more likely to react to nickel than those who did not have pierced ears (p = 0.031 and OR = 2.8). There was a tendency towards a difference in age between those who reacted and those who did not (p = 0.059), with statistical significance. Those who reacted tended to be older than those who did not. The mean age in months of children who did react to nickel was 76.8 with an SD of 43.3 and a median of 90.3. The covariables sex, ear piercing, spectacles and age had p < 0.25 and were included in the multivariate model. After the process of variable selection, it was observed that none of these covariables in conjunction were associated with reaction to nickel. The only factor associated with a reaction to nickel was ear piercing, where patients with pierced ears were more likely to react.
Discussion
Comparisons with published data are compromised by methodological differences, such as sample population selection and the concentrations of each allergen tested. Marcussen was the first to test unselected children in Denmark in 1963. Testing of 191 hospitalized children aged from 0 to 10 years of age found that 29% were positive for reactions to 5% nickel sulphate at the 48-hour reading.16 Weston et al. tested 314 healthy child volunteers aged 6 months to 18 years, finding that 7.6% were positive for chrome at the 72-hour reading, 24 hours after the test had been removed.1 In 1991, Barros et al. administered contact tests to 562 children aged 5 to 14 from four schools in Portugal and found that after 48 hours there were five (0.89%) reactions to nickel, three (0.53%) to cobalt and one (0.18%) to chrome,17 which is a much lower rate than we observed in our sample. Bruckner et al. tested 85 children aged 6 months to 5 years who presented for routine pediatric consultations at a pediatric clinic in Denver. Tests were removed after 48 hours and readings taken from 96 to 120 hours demonstrated positive reactions to nickel in 11 children (12.9%) and to cobalt in one child (1.2%), but no children reacted to chrome.18 The majority of the children who had reactions to nickel were girls (69.0%) with pierced ears.
Several different researchers have shown that sensitization to nickel is associated with wearing earrings. The instruments used to pierce ears are generally made from stainless steel, which has a low allergenic potential and are probably not the cause of sensitivity, which is probably caused by the studs put in after piercing.7 Parents tend to pierce children's ears and put gold studs in to keep the holes open, but low-quality gold can contain nickel and could therefore be responsible for sensitization.19 Between March of 1992 and March of 1993, Dotterud & Falk tested 424 Norwegian schoolchildren aged from 7 to 12 years. The 48-hour reading was positive in 14.9% for nickel, with greater frequency among girls who wore earrings.7 Jensen et al. studied female students aged from 17 to 22 years, finding that 19% of those with pierced ears were sensitive to nickel, whereas just 5.3% of those without pierced ears were sensitive to this metal.20 Mortz et al. studied Danish schoolchildren aged 12 to 16 and found that 15.9% of the girls with earrings had an allergy to nickel compared with 3.6% of those who did not wear earrings.6 It appears that the risk of allergy increases with the number of piercings in the ear lobes7 and when piercing is performed before 20 years of age.21 Rystedt & Fischer observed that 24 of 109 women with ears that had been pierced before they were 20 years old had nickel allergy against six out of 69 women with ears pierced after they were 30 years old (p < 0.05).21 Possible reasons are that young people use cheaper jewelry and the fact that the ear-piercing environment in the ear lobe is closed and humid, making irritant dermatitis more likely, which in turn encourages sensitivity.21 In Nigeria, where both men and women wear jewelry equally, no difference between sexes has been observed in the prevalence of sensitivity to nickel.22
Five (17.2%) of the 29 children who reacted to nickel did not have a history of dermatitis triggered by contact with metals, demonstrating a rate of clinical relevance of 82.8%. Mortz et al. tested 1,146 Danish schoolchildren aged 12 to 16 years. Readings taken at 72 hours, 24 hours after removing the test, were positive for nickel reaction in 8.6% with a clinical relevance of 69.4%.23 Individuals with a positive test, but without clinical relevance, may be a group at risk of developing contact dermatitis if exposed to the allergen in question at concentrations exceeding their personal tolerance.23
Thirty-four children (23.6%) reported that they had had cutaneous reactions to wearing jewelry. Fifteen (44.1%) of these children did not react to the metals tested. Similar data have been published by Gawkrodger et al., who observed that 50% of 449 individuals with a history of reactions to jewelry had negative contact test results, with no significant difference in atopic disease between those whose contact tests were negative and those whose tests were positive.24 Occasionally, people may truly have an allergy, but still exhibit a negative reaction to the contact test.19 Other possible causes are irritant cutaneous reactions of allergies to other metals, such as palladium.24
Although 5% nickel sulphate can produce irritant reactions in children, we observed just three such reactions (2.1% of the children tested). This observation is shared by other authors who did not observe irritant reactions to nickel at this concentration in children.4,25 No serious side effects were detected either and the contact test was safe for the population tested.
The majority of studies do not detect a significant association between atopic disease and reactivity to metals,6,26-28 which is comparable to our observations.
In conclusion, although ear piercing is probably not the only cause of sensitization to nickel, in the sample studied here it was the only covariable that exhibited a significant association. In view of the current trend for nickel allergies to increase, parents should be warned about the association with ear piercing. Further studies are needed to determine the ideal age for ear piercing and the ideal materials for earrings., which could impact on the incidence of nickel contact allergic dermatitis in the general population.
References
- 1. Weston WL, Weston JA, Kinoshita J, Kloepfer S, Carreon L, Toth S, et al. Prevalence of positive epicutaneous tests among infants, children, and adolescents. Pediatrics. 1986;78:1070-4.
- 2. Hogeling M, Pratt M. Allergic contact dermatitis in children: the Ottawa hospital patch-testing clinic experience, 1996 to 2006. Dermatitis. 2008;19:86-9.
- 3. Lewis VJ, Statham BN, Chowdhury MM. Allergic contact dermatitis in 191 consecutively patch tested children. Contact Dermatitis. 2004;51:155-6.
- 4. Stables GI, Forsyth A, Lever RS. Patch testing in children. Contact Dermatitis. 1996;34:341-4.
- 5. Thyssen JP, Johansen JD, Menné T. Contact allergy epidemics and their controls. Contact Dermatitis. 2007;56:185-95.
- 6. Mortz CG, Lauritsen JM, Bindslev-Jensen C, Andersen KE. Nickel sensitization in adolescents and association with ear piercing, use of dental braces and hand eczema. The Odense Adolescence Cohort Study on Atopic Diseases and Dermatitis (TOACS). Acta Derm Venereol. 2002;82:359-64.
- 7. Dotterud LK, Falk ES. Metal allergy in Norwegian schoolchildren and its relationship with ear piercing and atopy. Contact Dermatitis. 1994;31:308-13.
- 8. Fisher AA. Allergic contact dermatitis in early infancy. Cutis. 1994;54:300-2.
- 9. Spiewak R. Allergische Kontaktdermatitis im Kindesalter. Eine Übersiht und Meta-Analyse. Allergologie. 2002;25:374-81.
- 10. Johnke H, Norberg LA, Vach W, Bindslev-Jensen C, Host A, Andersen KE. Reactivity to patch tests with nickel sulfate and fragrance mix in infants. Contact Dermatitis. 2004;51:141-7.
-
11Secretaria Municipal de Saúde de Belo Horizonte. [website] Belo Horizonte, MG. http://www.pbh.gov.br/smsa Acesso: 02/12/2008.
» link - 12. Grupo Brasileiro de Estudo em Dermatite de Contato (GBEDC) do Departamento Especializado de Alergia em Dermatologia da Sociedade Brasileira de Dermatologia. Estudo multicêntrico para a elaboração de uma bateria-padrão brasileira de teste de contato. An Bras Dermatol. 2000;75:147-56.
- 13. Wilkinson DS, Fregert S, Magnusson B, Bandmann HJ, Calnan CD, Cronin E, et al. Terminology of contact dermatitis. Acta Derm Venereol. 1970;50:287-92.
- 14. Bolfarine H, Bussab WO. Elementos de Amostragem. São Paulo: Edgard Blücher; 2005. p. 290.
- 15. Triola MF. Introdução à estatística. Rio de Janeiro: LTC; 2005. p. 410.
- 16. Marcussen PV. Primary irritant patch-test reactions in children. Arch Dermatol. 1963;87:378-82.
- 17. Barros MA, Baptista A, Correia TM, Azevedo F. Patch testing in children: a study of 562 schoolchildren. Contact Dermatitis. 1991;25:156-9.
- 18. Bruckner AL, Weston WL, Morelli JG. Does sensitization to contact allergens begin in infancy? Pediatrics. 2000;105:e3. Disponível em: http://www.pediatrics.org/cgi/contente/full/105/1/e3 Acesso: 09/03/2009.
- 19. Rietschel RL, Fowler JF Jr. Metals. Fisher's contact dermatitis. 6th ed. Hamilton, Ontario: BC Decker Inc; 2008. p. 641-99.
- 20. Jensen CS, Lisby S, Baadsgaard O, Vřlund A, Menné T. Decrease in nickel sensibilization in a Danish schoolgirl population with ears pierced after implementation of a nickel-exposure. Br J Dermatol. 2002;146:636-42.
- 21. Rystedt I, Fischer T. Relationship between nickel and cobalt sensitization in hard metal workers. Contact Dermatitis. 1983;9:195-200.
- 22. Olumide YM. Contact dermatitis in Nigeria. Contact Dermatitis. 1985;12:241-6.
- 23. Mortz CG, Lauritsen JM, Bindslev-Jensen C, Andersen KE. Prevalence of atopic dermatitis, asthma, allergic rhinitis, and hand and contact dermatitis in adolescents. The Odense Adolescence Cohort Study on Atopic Diseases and Dermatitis. Br J Dermatol. 2001;144:523-32.
- 24. Gawkrodger DJ, Lewis FM, Shah M. Contact sensitivity to nickel and other metals in jewelry reactors. J Am Acad Dermatol. 2000;43:31-6.
- 25. Manzine BM, Ferdani G, Simonetti V, Donini M, Seidenari S. Contact sensitization in children. Pediatr Dermatol. 1998;15:12-7.
- 26. Brasch J, Schnuch A, Uter W; German Contact Dermatitis Research Group (DKG) and the Information Network of Departments of Dermatology (IVDK). Strong allergic patch test reactins may indicate a general disposition for contact allergy. Allergy. 2006;61:364-9.
- 27. Uter W, Pfahlberg A, Gefeller O, Geier J, Schnuch A. Risk factors for contact allergy to nickel - results of a multifactorial analysis. Contact Dermatitis. 2003;48:33-8.
- 28. Brasch J, Schnuch A, Uter W. Patch-test reaction patterns with a predisposition to atopic dermatitis. Contact Dermatitis. 2003;49:197-201.
Publication Dates
-
Publication in this collection
30 Apr 2010 -
Date of issue
Apr 2010
History
-
Accepted
23 Dec 2009 -
Received
07 Oct 2009