Abstracts
OBJECTIVES: To review publications about the main features of post-infectious bronchiolitis obliterans and its history, etiology, epidemiology, risk factors, pathogenesis, histological findings, clinical presentation, complementary tests, diagnostic criteria, differential diagnosis, treatment and prognosis. SOURCES: Non-systematic review of MEDLINE and LILACS databases and selection of 66 most relevant studies. SUMMARY OF THE FINDINGS: In the post-infectious bronchiolitis obliterans there is an insult to respiratory epithelial cells, and its clinical severity is associated with the degree of lesion and inflammation. Diagnosis is made according to clinical signs and symptoms, by exclusion of main differential diagnoses and with the aid of complementary tests. High resolution CT, particularly images obtained during inspiration and expiration, provide information for the evaluation of the small airways. Pulmonary function tests show fixed airway obstructions and marked decrease of FEF25-75%. Treatment has not been definitely established, and corticoids have been administered as pulse therapy or by inhalation of high doses of steroids. However, data about its efficacy are scarce in the literature. Long-term prognosis is variable, and there might be either clinical improvement or deterioration into respiratory insufficiency and death. CONCLUSION: Post-infectious bronchiolitis obliterans is a disease with a high morbidity rate; it should be treated by a multidisciplinary team, and patients should be followed up for a long period of time.
Bronchiolitis obliterans; diagnosis; spirometry; CT; child
OBJETIVOS: Revisar os trabalhos publicados sobre os principais aspectos da bronquiolite obliterante pós-infecciosa, relacionados com sua história, etiologia, epidemiologia, fatores de risco, patogenia, alterações histológicas, manifestações clínicas, exames complementares, critérios diagnósticos, diagnóstico diferencial, tratamento e prognóstico. FONTES DOS DADOS: Realizou-se uma revisão não sistemática nas bases de dados MEDLINE e LILACS, selecionando-se 66 referências mais relevantes. SÍNTESE DOS DADOS: Na bronquiolite obliterante pós-infecciosa ocorre lesão do epitélio respiratório, e a gravidade clínica está relacionada aos diferentes graus de lesão e ao processo inflamatório. O diagnóstico baseia-se no quadro clínico, na exclusão dos principais diagnósticos diferenciais e no auxílio dos exames complementares. A tomografia computadorizada de alta resolução, principalmente com imagens em inspiração e expiração, possibilta a avaliação das pequenas vias aéreas. As provas de função pulmonar caracterizam-se por padrão obstrutivo fixo com redução acentuada do FEF25-75%. O tratamento não está bem estabelecido, e o uso de corticoides tem sido preconizado em forma de pulsoterapia ou por via inalatória em elevadas doses, no entanto, os dados da literatura a respeito de sua eficácia ainda são escassos. O prognóstico a longo prazo é variável, podendo haver melhora clínica ou evolução para insuficiência respiratória crônica e óbito. CONCLUSÃO: A bronquiolite obliterante pós-infecciosa é uma doença que cursa com elevada morbidade e deve ser abordada por equipe multidisciplinar com acompanhamento em longo prazo.
Bronquiolite obliterante; diagnóstico; espirometria; tomografia; criança
REVIEW ARTICLE
Post-infectious bronchiolitis obliterans in children
Natália da Silva ChampsI; Laura M. L. B. F. LasmarII; Paulo A. M. CamargosIII; Christophe MarguetIV; Gilberto Bueno FischerV; Helena Teresinha MocelinVI
IMestre, Saúde da Criança e do Adolescente. Especialista, Pneumologia Pediátrica. Universidade Federal de Minas Gerais (UFMG), Belo Horizonte, MG, Brazil
IIDoutora. Professora adjunta, Departamento de Pediatria, Faculdade de Medicina, UFMG, Belo Horizonte, MG, Brazil
IIIDoutor. Professor titular, Departamento de Pediatria, Faculdade de Medicina, UFMG, Belo Horizonte, MG, Brazil
IVProfessor, Unité de Pneumologie Pédiatrique, Centre Hospitalier Universitaire, Université de Rouen, Rouen, France
VDoutor. Professor titular, Pediatria, Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, RS, Brazil. Professor, Programa de Pós-Graduação em Pediatria, Universidade Federal do Rio Grande do Sul (UFRGS), Porto Alegre, RS, Brazil. Médico, Serviço de Pneumologia Pediátrica, Hospital da Criança Santo Antônio (HCSA), Irmandade Santa Casa de Misericórdia de Porto Alegre (ISCMPA), Porto Alegre, RS, Brazil
VIDoutora. Médica, Serviço de Pneumologia Pediátrica, HCSA, ISCMPA, Porto Alegre, RS, Brazil
Correspondence Correspondence: Laura Maria Belizário Lasmar Departamento de Pediatria, Faculdade de Medicina, UFMG Av. Alfredo Balena, 190, Sala 4061 CEP 30130-100 â Belo Horizonte, MG â Brazil Tel.: +55 (31) 3292.0309 Fax: +55 (31) 3409.5664, +55 (31) 3248.9664 E-mail: laurabl@medicina.ufmg.br
ABSTRACT
OBJECTIVES: To review publications about the main features of post-infectious bronchiolitis obliterans and its history, etiology, epidemiology, risk factors, pathogenesis, histological findings, clinical presentation, complementary tests, diagnostic criteria, differential diagnosis, treatment and prognosis.
SOURCES: Non-systematic review of MEDLINE and LILACS databases and selection of 66 most relevant studies.
SUMMARY OF THE FINDINGS: In the post-infectious bronchiolitis obliterans there is an insult to respiratory epithelial cells, and its clinical severity is associated with the degree of lesion and inflammation. Diagnosis is made according to clinical signs and symptoms, by exclusion of main differential diagnoses and with the aid of complementary tests. High resolution CT, particularly images obtained during inspiration and expiration, provide information for the evaluation of the small airways. Pulmonary function tests show fixed airway obstructions and marked decrease of FEF25-75%. Treatment has not been definitely established, and corticoids have been administered as pulse therapy or by inhalation of high doses of steroids. However, data about its efficacy are scarce in the literature. Long-term prognosis is variable, and there might be either clinical improvement or deterioration into respiratory insufficiency and death.
CONCLUSION: Post-infectious bronchiolitis obliterans is a disease with a high morbidity rate; it should be treated by a multidisciplinary team, and patients should be followed up for a long period of time.
Keywords: Bronchiolitis obliterans, diagnosis, spirometry, CT, child.
Introduction
Bronchiolitis obliterans (BO) is a clinical syndrome characterized by the chronic obstruction of small airways that, by definition, are less than 2 mm in diameter.1,2 Several causes of BO have been described, such as infections caused by viruses, atypical germs or bacteria, foreign body or gastric content aspiration, inhalation of toxins, collagenosis, or lung or bone marrow transplant. The purpose of this review was to describe the several aspects associated with post-infectious bronchiolitis obliterans (PIBO), the most common form of BO in children.3
History
The term "bronchiolitis obliterans" was first used in 1835, and then again only in 1901 for two patients with chronic cough and dyspnea of undetermined origin who later died.4,5
In 1953, Swyer & James described the case of a 6 year old boy with unilateral pulmonary hyperlucency, reduced lung volume and ipsilateral reduction of pulmonary artery caliber.6 In 1954, MacLeod described this syndrome in nine adult patients that had unilateral pulmonary hyperlucency.7 Today, the Swyer-James or MacLeod syndrome is one of the presentations of PIBO.
Since the end of the 1980s, after the description of new etiologic factors and the advent of high resolution CT (HRCT), which provides detailed images of the small airways, the interest in BO has been growing, and the number of publications about it has multiplied. During this time, particularly in Latin America, outbreaks of adenovirus infection have been detected, and patients that developed PIBO had been previously affected by that infection.
Etiology
BO is a final process common to several lung injuries. However, in 1/3 of the cases, the cause is unknown.8 Most studies associate PIBO with adenovirus infections, although other viruses may also be implicated: measles, influenza, parainfluenza, and respiratory syncytial virus (RSV). Adenovirus has already been identified as a cause of PIBO in up to 72%9 of the cases; in different study populations, about 303 to 60%10 of the cases of adenovirus infection progress into PIBO. Serotypes 3,11 7,3,12 1113 and 2114,15 have been described as the most virulent.
Questions have been raised about RSV infection being a possible cause of PIBO because, although it is the agent most often associated with acute viral bronchiolitis (AVB), there are few reports in the literature of PIBO secondary to RSV infection.5,8 However, this virus has been detected in patients that develop PIBO in up to 30% of the cases.10 The occurrence of simultaneous infection by adenovirus and RSV has been associated with the worst outcomes.16,17 Coinfection of viruses and bacteria may be associated with greater risk of lung lesions.3
PIBO has also been described as a consequence of infection by bacteria, such as Staphylococcus aureus18 and Streptococcus pneumoniae,19 or atypical germs, such as Mycoplasma pneumoniae.20 Mycoplasma infection, in some cohorts, was the second most frequent etiologic agent and explained about 26% of the cases.19,21
Epidemiology
No epidemiological data about PIBO have been produced so far, but there seems to be a predominance of cases in the southern hemisphere (Argentina, Brazil, Chile, and New Zealand) and a lower frequency in the United States and Europe.5 Some authors suggested that people of Asian ethnicities would be more susceptible to the disease because of reports of cases of Polynesian descendents in New Zealand, as well as reports in South Korea and Taiwan.8
Some studies have described a greater occurrence of adenovirus AVB in Amerindian populations.11,15 In Argentina, a greater frequency of a representative allele of the Amerindian population was found in patients with PIBO (HLA haplotype DR8-DQB1*0302)5, which may be associated with a possible genetic predisposition.8 Moreover, epidemiological studies conducted in the early 1990s found a high prevalence of adenovirus 7h, a highly virulent serotype, in Argentina, Chile and Uruguay, which may explain the greater number of cases described in that time period in the region.5
AVB may affect up to 10% of infants in their first year of life, and about 1% of the patients with this condition may develop PIBO.22,23 These data suggest that its prevalence might be greater than previously suspected and that there might be many underdiagnosed cases. The increase in the number of reports in the literature in the last years in different regions, such as Spain,24 China25 and France,26 corroborates this hypothesis.
At the same time, reports delivered during recent scientific meetings in several centers with a tradition in the care of PIBO suggest a reduction in the incidence of the disease in the last few years, probably due to the decrease in microepidemic outbreaks of severe adenovirus infection.27
Risk factors
It remains unclear why some children develop PIBO after AVB. Viral load, immunological response and genetic and environmental factors may be associated.9
Some possible risk factors for the occurrence of PIBO have been described: adenovirus AVB (odds ratio [OR] = 49.9),9 hospitalization for longer than 30 days (OR = 27.2),3 multifocal pneumonia (OR = 26.6),3 need of mechanical ventilation (OR = 11.9)9 and hypercapnia (OR = 5.6).3 In an isolated study, elevated IgE levels were thought to be associated with a worse prognosis,28 but this finding was not reported in any of the studies with larger cohorts. The analysis of mechanical ventilation changes did not clarify whether it is an indicator of disease severity or the factor responsible for, even if only partially, the direct induction of airway lesions.9
Data about the age at the time of event are controversial. In children with adenovirus pneumonia, the younger the child at the time of pneumonia, the greater the risk of having an abnormal pulmonary function tests later on.12 However, a study found that children with PIBO who remained symptomatic had pulmonary lesions at more advanced ages than the children that became asymptomatic during follow-up (mean 5 months and 2 months, respectively).29
Bilateral pulmonary involvement is expected in cases of PIBO and is associated with greater severity. A study found that children with bilateral pulmonary involvement had higher risks of persistent respiratory symptoms, recurrent pneumonia episodes, malnutrition and digital clubbing than children with unilateral involvement.
Pathogenesis
Despite the diversity of microorganisms involved in the etiology of PIBO, all share the capacity to affect epithelial cells of the respiratory tract during the acute phase. The lesions lead to necrosis of bronchiolar epithelial cells and peribronchial, intraepithelial, and interstitial inflammatory infiltrate.30
The specific role of each cell in the pathogenesis of PIBO is not fully understood. A study to determine the lymphohistiocytic profile using fragments of pulmonary biopsy tissue of patients with PIBO have found that the CD3+T cells were the most frequently found and that there was a predominance of the CD8+T cell subtype. CD8 cells may lead alveolar epithelial cells to produce cytokines, which intensify inflammation and perpetuate lung lesions.30
Immunocomplexes containing the virus have been found in the lung of patients with adenovirus penumonia.5,8,31 Moreover, the increases of IL-6, IL-8 and TNF-α have been associated with greater severity of adenovirus infection.32
In the bronchoalveolar lavage (BAL) fluid of patients with PIBO, the percentage of neutrophils (Figure 1) is elevated, and there is a discrete increase of lymphocytes, as well as an increase in IL-8 concentrations, a chemokine for neutrophils.33 These findings were confirmed years after pulmonary insult, which suggests that inflammation is persistent.34
The development of animal BO models may contribute to a better understanding of its pathogenic mechanisms and facilitate the study of possible treatment regimes. An experimental model, developed using the intratracheal instillation of nitric acid in rats and inducing histological changes compatible with constrictive BO, may be adapted for use in future experimental studies.35
Histological changes
Histological findings are similar, regardless of etiology, which suggests that BO corresponds to a final response to different insults to the epithelial cells of the small airways, which advances by means of intraluminal healing instead of the normal repair path.5,8
There are two histologically well defined types if BO: constrictive bronchiolitis and proliferative bronchiolitis. The latter is characterized by granulation tissue in the form of a polypoid mass inside the airway. When the granulation tissue is also found inside the alveoli, the patient has bronchiolitis obliterans organized pneumonia (BOOP),36 currently called cryptogenic organizing pneumonia,8 which is not found in PIBO.
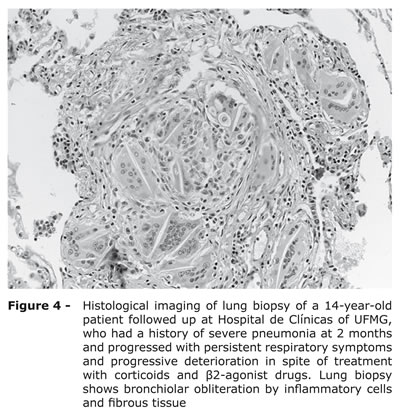
Most patients with PIBO have the constrictive form, which was identified in up to 97% of the cases.36 Total obliteration of bronchioles starts with the necrosis of bronchiolar epithelium and inflammatory infiltration of the mucosa, submucosa, peribronchiolitis area, and the bronchiolar lumen, predominantly in terminal bronchioles. Adjacent pulmonary parenchyma is spared or has very little involvement. Bronchioles often lose their form and have collagen deposition and mucous buffers. In a more advanced stage, there is submucosal fibrosis, which reaches the bronchiolar lumen in a concentric pattern. As fibrosis advances, the bronchiolar lumen reduces progressively to the point of full obliteration.37
Indirect signs of airway obstruction may be seen, such as mucostasis, macrophage accumulation, hyperinflation, bronchiolar distortion and dilatation.5,36 In addition to the changes in the small airways, changes in larger airways may also be seen. There may be hypertrophy or thickening of the bronchial epithelium, and sometimes the lumen of larger bronchi may be occluded, which may lead to BO.8,20
Clinical presentation
The diagnosis of PIBO should always be suspected in previously healthy children that develop chronic respiratory symptoms for longer than 4 to 8 weeks after an episode of acute, usually severe infection.8-10,36-39
The clinical presentation of PIBO is unspecific and both the severity and extension of bronchopulmonary lesions vary widely, which may contribute to the fact that they may be underdiagnosed. Despite possible ethnic or geographic differences, a study with Korean and American patients reported that clinical, radiological, and functional findings were similar in the two countries.40
The persistence of symptoms, such as tachypnea, wheezing, and productive cough, is usually the first noticeable sign. Several of these patients are followed up for several years with a diagnosis of asthma, but the unsatisfactory response to oral or inhaled corticoids and oral or inhaled bronchodilators draws the attention to the possibility of another diagnosis.19 The presence of crackles during follow-up is a remarkable change seen during physical examination of most patients.18,19,28 PIBO should also be suspected in case of chronic coughing or expectoration, decreased vesicular murmur, recurrent pneumonia, impaired weight and height gains, and episodes of hypoxemia and desaturation secondary to physical exercise or during sleep.37 Low arterial oxygen saturation may indicate severity, and hypoxemia has been described in up to half of the patients.40 In more severe cases, there might be thorax deformity, digital clubbing, and signs and symptoms of pulmonary arterial hypertension (PAH).17,18,41
Although the Swyer-James or MacLeod syndrome may be within the PIBO spectrum, no studies have differentiated it from typical forms of PIBO. However, the clinical presentation of this syndrome seems to be milder (Fischer GB, personal communication).
Radiography and laboratory tests
Chest X ray
Ideally, a simple inspiration-expiration chest X ray should be requested. Changes are unspecific, but they may contribute to rule out diseases that are part of the differential diagnosis.
During AVB, there is hyperinflation, interstitial infiltrate, peribronchial thickening, areas of segmental or subsegmental atelectasis and consolidations.18,28,38 Parenchymatous lesions tend to be severe and extensive, particularly when the etiologic agent is adenovirus.15
After the initial episode, pulmonary hyperinflation may persist, as well as air trapping, which may be generalized or localized in one lung. Uni- or bilateral hyperlucency may be a consistent finding,21,28,40 already described in up to 50% of the cases.19 Persistent atelectasis has been found in up to 30% of the cases.18 Although not the best test to evaluate bronchiectasis, a chest X ray may be used to detect it in some patients.28 Sometimes, cardiomegaly and indirect signs of pulmonary hypertension may also be found.37 However, chest X ray changes are usually poor when compared with clinical signs and symptoms and CT findings, and it may be normal in some cases.19
A study has described the five most frequent patterns of PIBO21: 1) unilateral hyperlucency and increased lung volume; 2) persistent atelectasis in one or more lobes; 3) Swyer-James or MacLeod syndrome (hyperlucent lung and normal or decreased lung volume)4,7; 4) bilaterally hyperlucency; 5) mixed pattern of hyperlucency, persistent atelectasis and peribronchial thickening. The latter pattern was the most frequent in that study.21
High-resolution CT
There is a consensus that the diagnosis of PIBO may be made at a good safety margin when clinical findings and noninvasive tests are analyzed.5,18,19,21,29 There is no specific finding for PIBO, but, when correlated with the clinical presentation, chest HRCT is the best exam to diagnose PIBO. HRCT provides information about bronchial lesions or lesions of the small airways21 because it is more sensitive than chest X rays and pulmonary scintigraphy to detect airway and parenchyma abnormalities that occur during PIBO.
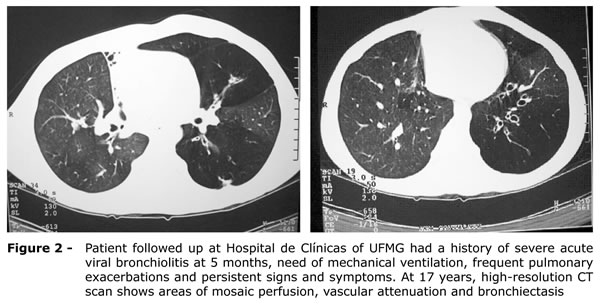
HRCT findings may be divided into direct and indirect.42 Direct signs are wall thickening and bronchiole obliteration, but they are less frequent.43 Indirect signs include bronchiectasis, bronchial wall thickening, mosaic perfusion, air trapping, decreased lung volume, and reduced hilar and peripheral vessel diameters.29
The most frequent signs in several studies were mosaic perfusion, air trapping, vascular attenuation, bronchiectasis and peribronchial thickening (Figure 2 and Table 1). The data about the frequency of CT abnormalities in patients of eight centers are seen in Table 1.
The comparison of inspiration and expiration images provides the chance to combine structural and functional information and evaluate the presence of air trapping and hypoxic vasoconstriction, which may appear as areas of mixed hypo- and hyperattenuation (mosaic perfusion).44 This finding is probably one of the most sensitive markers of PIBO, but it is not very specific because it is found in other pathologies, such as vascular and infiltrative diseases of the lung parenchyma.45 In about 13 to 18% of the cases, inspiration images are normal, and attenuation heterogeneity is only seen during expiration.46 The identification of air trapping in the expiration sections may also be useful to differentiate the airway diseases from other causes of mosaic perfusion.42
Few studies have evaluated lesion extension using HRCT. In one of them, about 64% of the patients had bilateral abnormalities associated with poorer clinical progressions.19 In another series, involvement was bilateral in all cases, and air trapping was diffuse in 55% of the cases and multifocal in 45%. Those authors found no direct signs of bronchiolar lesions in the CT scans under analysis.29 In another study that used densitometry and volumetry, a CT technique that uses three-dimensional reconstructions of lung parenchyma to measure the volume of lung parenchyma with normal and abnormal (non-functional) densities, found an inverse correlation with functional tests (FEV1) and oxygen saturation during the 6-minute walk test.47
The CT differential diagnosis between severe persistent asthma and BO remains challenging. A study compared CT findings of adult patients with severe asthma or BO of several etiologies and diagnosis confirmed by lung biopsy, and found that only mosaic perfusion was statistically significant to differentiate the two diseases (p = 0.0006). Mosaic perfusion was significantly larger in patients with BO, but was found in only one patient with asthma and in a small area of the lung.48 The pulmonary function tests tend to differentiate the two conditions because the pattern of fixed obstruction in PIBO is the opposite in patients that have asthma with reversible obstruction.
The main limitations of CT are the exposure to radiation and the difficulty to obtain quality images in small children, which requires, in many cases, sedation or general anesthesia with endotracheal intubation. Protocols with low radiation doses have been used without any substantial harm to image quality and should always be taken into consideration when using CT for infants or children.49 The lateral decubitus technique has been described as an alternative for the expiration technique in small children.50
Ventilation-perfusion scintigraphy
Changes are also unspecific, but scintigraphy provides valuable information, particularly for the evaluation of extension, distribution, and severity of lung involvement. In most patients, there is a heterogeneous distribution of technetium-99m in the lung parenchyma, as in ventilation-perfusion scintigraphy. Zhang et al. found perfusion abnormalities in all patients with BO in their study.28 The comparison of lung perfusion with chest X ray reveals that scintigraphy changes correspond to the most affected areas in the radiographs, with peribronchial thickening and bronchiectasis.5 Patients with more perfusion defects in scintigraphy also tend to have greater morbidity, and the extension of perfusion defects is associated with the number of exacerbations and hospitalization time (r = 0.66; p = 0.00).51
Bronchoscopy
Although bronchoscopy does not usually reveal any significant changes, it may be useful to rule out diseases that are part of the differential diagnosis. It should be performed in children with signs of unilateral involvement, such as hyperlucency in one lung or lobe, so that foreign body or other causes of localized obstruction may be ruled out.19
Pulmonary function tests
The typical PIBO pattern is a fixed obstructive ventilation disorder whose severity is variable with a marked reduction of terminal flows, particularly the forced expiratory flow between 25 and 75% (FEF25-75%). Its marked decrease (< 30%) is a sensitive indicator of PIBO in infants and children with a compatible clinical presentation.21 In PIBO, the involvement of small airways is more severe than in other diseases, such as asthma and bronchopulmonary dysplasia.5 Spirometric indices vary substantially between studies (Table 2), and this difference may be associated with the differences of each population and the severity of the disease. Expiration curves show a marked concavity (Figure 3), and there might be a reduction in forced vital capacity (FVC) associated with air trapping.44
Plethysmography showed that most patients had normal total lung capacity (TLC).44 However, it might be increased in some cases,5,8 and there might be an increased residual volume (RV) and RV/TLC ratio.34 A study with Brazilian and Chilean children found that there was an important decrease of FEV1 and FEV1 and FEF25-75%, as well as an increase in RV and specific airway resistance (sRaw). The most affected parameters and the percentage means of predicted values were FEV1 = 45.9%; FEF25-75% = 21.5%; RV = 281.1%; VR/TLC = 236.2%; and sRaw = 665.3%.52
When the forced oscillation technique was used, patients with PIBO had a more marked increase in air resistance than patients that also had adenovirus pneumonia but did not develop PIBO.41
The obstructive pattern is found at an early stage in PIBO. Functional studies with infants revealed that forced expiratory maneuvers had a marked effect on the curve and reduced expiratory flows, particular tidal expiratory flows.52 Moreover, there was a decrease of compliance and an increase of airway resistance.53
The evaluation of pulmonary function in infants is not available in all centers. There is evidence that CT changes in children younger than 3 years may predict future pulmonary function.54
Some patients have a positive response to intermittent bronchodilators, although marked obstruction persists.21,40 Whenever it occurs, bronchial hyperresponsiveness differs from that seen in patients with asthma. Most patients with PIBO are hyperresponsive to methacholine, but not to adenosine 5monophosphate, whereas patients with asthma usually respond to the two substances.55
Children with PIBO who underwent cardiopulmonary exercise tests and the 6-minute walk test had a reduced exercise capacity in both. In the 6-minute walk test, there was a reduction in distance walked, a fall in oxygen saturation, and an increased sensation of dyspnea.56,57 This test is easily applied and is useful as a screening tool, particularly where the cardiopulmonary test is not available.27
There is some controversy among authors in relation to findings of pulmonary function in children with PIBO. Some believe that children with mild or moderate ventilation disorders should not be diagnosed with PIBO, and most authors believe that the degree of response to bronchodilator is variable, but does not achieve full reversibility because airway obstruction is fixed.10,28,34,41,52 Most centers require, as PIBO diagnostic criteria, that fixed airway obstruction should be demonstrated, and tend to diagnostic exclusion in cases with normal pulmonary function.27
Lung biopsy
Histological examination of an adequate fragment obtained by lung biopsy remains the most accurate method to diagnose PIBO.38 Open lung biopsy18,28,40 may be obtained by removing a fragment from the lingula or the area believed to be more severely affected.
Although lung biopsy establishes the diagnosis of PIBO, the lesions are distributed heterogeneously; therefore, tissue that has not been affected may be harvested. The absence of characteristic changes in the fragment analyzed does not rule out the diagnosis of PIBO.37 According to reports, biopsies may not diagnose PIBO in up to 30% of the cases.
The changes found may be mild, depending on the disease stage and the region from where the fragment was collected. In initial stages, bronchiolar epithelial necrosis and inflammation may be found, but fibroblastic response may still be at a minimal stage.18 When more advanced, lung tissue may be destroyed, which may complicate the identification of bronchioles.18 Small changes that may suggest airway obstruction should, therefore, be taken into consideration.
Currently, considering the limitations described above and the risks of the procedure, the need to perform lung biopsies to establish the diagnosis of PIBO has been questioned.18,21,28,38 With the advent of HRCT, the method of choice to evaluate small airways,22 lung biopsy has been used only in selected cases for which HRCT findings are not available or are inconclusive.21 Most diagnoses in the differential analysis may be excluded using noninvasive examination. However, in cases of severe progression and gradual deterioration in spite of treatment, a biopsy may be useful to confirm the diagnosis22 (Figure 4).
Cardiovascular evaluation
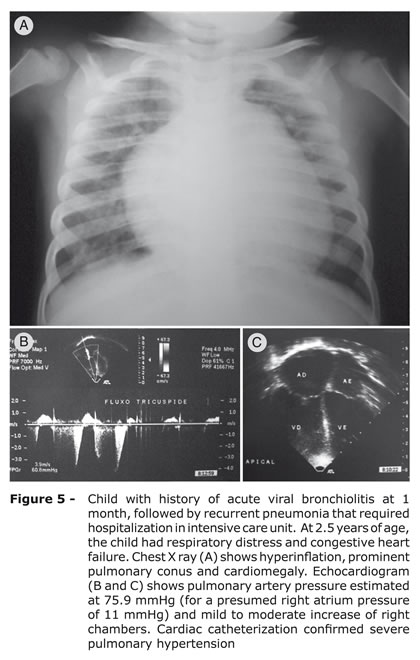
Chronic hypoxemia, particularly during sleep or physical exercise, may lead to PAH and cor pulmonale when not detected at an early stage. PAH secondary to PIBO has been found in 6.5 to 15% of the patients in different cohorts.17,38,40 Overnight oximetry may be a useful evaluation test in patients with severe PIBO that have used continuous oxygen supplementation to evaluate overnight hypoxemia and to prevent cor pulmonale. In children with signs of hypoxemia, a cardiovascular investigation should be considered, including clinical examination, electrocardiogram, echocardiogram and cardiac catheterization, if necessary, to detect patients with PAH at an early stage.37Figure 5 shows a 2.5-year-old child with severe PAH secondary to PIBO.
Diagnostic criteria
Clinical history and the abnormal test results described above suggest the diagnosis of PIBO. However, no specific test has been defined for its diagnosis so far. Therefore, some criteria are suggested to help to define an accurate diagnosis. For most authors, the diagnosis should be made according to clinical history, suggestive functional and radiological abnormalities and the exclusion of other pulmonary diseases. The criteria used for the diagnosis of PIBO may be the following27:
- History of bronchiolitis or acute viral pneumonia in a previously healthy child before 3 years of age;
- Evidence of persistent obstruction of airways after acute event, according to physical exam or pulmonary function tests. This obstruction does not respond to at least 2 weeks of systemic corticoid administration associated with bronchodilator;
- Radiological findings of obstructive pulmonary disease, such as hyperinflation, atelectasis, bronchial thickening, and bronchiectasis;
- Mosaic perfusion and air trapping in chest CT scan;
- Exclusion of other chronic pulmonary diseases that progress with persistent respiratory symptoms, such as tuberculosis, cystic fibrosis, bronchopulmonary dysplasia, immunodeficiency, severe asthma, and α-1 antitrypsin deficiency.
A study with 125 Argentinean children younger than 2 years used a score to predict PIBO based on the following variables: typical clinical history (4 points), adenovirus infections (3 points), and HRCT scan with mosaic perfusion (4 points). A score > 7 may predict a diagnosis of PIBO with a specificity of 100% and sensitivity of 67%. Their study, however, included only patients with a severe form of the disease.39
The search for more accurate criteria to diagnose PIBO is important, but if these criteria are not met, the adoption of support measures should not be delayed because they may improve prognosis and the quality of life of these patients. Numerous patients are referred to specialized centers only at a late stage. A study conducted in the pediatric Pulmonology unit of Hospital de Clínicas of UFMG followed up 23 children for a mean time of 6 years (maximum = 19 years), and found that, although the pulmonary insult occurred before 12 months of age in 69% of the cases and the patients had persistent symptoms after that initial insult, mean age at diagnosis was 5.7 years (p<0.001). Most patients were referred to the pediatric Pulmonology outpatient service of UFMG with a diagnosis of asthma or recurrent pneumonia.58
Differential diagnosis
To establish the diagnosis of PIBO, other pulmonary diseases should be ruled out, particularly those that lead to chronic obstruction of air flow. The differential diagnosis should include cystic fibrosis, chronic lung disease of prematurity, pulmonary tuberculosis, pulmonary hemosiderosis, congestive heart disease, immunodeficiency, α-1 antitrypsin deficiency, congenital malformations, acquired immunodeficiency syndrome (AIDS), foreign body aspiration and gastroesophageal reflux disease (GERD). The results of the following laboratory tests should be reviewed: sweat electrolytes, tuberculin test, serum immunoglobulin concentration, α-1 antitrypsin concentration, pH-metry, echocardiogram, and bronchoscopy.
To define the differential diagnosis of the Swyer-James or MacLeod syndrome, the following congenital malformations should be investigated: congenital absence of the pectoralis major muscle, congenital agenesis of pulmonary artery, and obstructive bronchial lesion.24
Sequelae of viral infections may affect any individual, with or without a primary disease. Studies that investigate changes associated with PIBO should exclude patients with other diseases to avoid introducing a selection bias. However, in clinical practice, a preterm infant or a child with another chronic disease may have AVB that leaves sequelae. Therefore, PIBO should not be ruled out in children with other chronic diseases.
GERD does not exclude PIBO; in fact it may be secondary to PIBO because of the increased intraabdominal pressure resulting from lung hyperinflation.44
A practical way to differentiate PIBO diagnosis in patients with AVB is that airway involvement is predominant in the acute phase, whereas in PIBO a severe case of pneumonia is associated (Fisher GB, personal communication).
Treatment
BO treatment has not been clearly defined in the literature. In addition to clinical, methodological and ethic difficulties, few studies using laboratory animals have been conducted to investigate both induction and pathogenesis or treatment options. Our review of the literature also did not yield any controlled clinical trials. Therefore, pharmacological approaches are often based on the clinical experience of different healthcare workers.
Patients should be treated by a multidisciplinary team that includes at least one pediatric pulmonologist, a pediatric cardiologist, a physical therapy specialist, a nutritionist, a psychologist, and a social worker.
General support measures
Some patients need home oxygen therapy, sometimes for long periods of time (months or years) after acute infection. In a series of 117 children with PIBO secondary to adenovirus infection, 87% had to use oxygen after hospital discharge for a mean time of 33 months.3 Saturation should be measured also during sleep because it may be satisfactory while the patient is awake but fall significantly during sleep. The oxygen concentration necessary to keep oxygen saturation above 94% is usually low (fraction of inspired oxygen [FIO2] = 25 to 40%).44
Respiratory physiotherapy should be prescribed to mobilize secretions and minimize chronic inflammation that may lead to recurrent infection. Together with physiotherapy, a long-term pulmonary rehabilitation program, similar to the one recommended for adults with chronic obstructive pulmonary disease, should be prescribed.
Adequate nutritional support should compensate the greater caloric loss due to the increased respiratory work. A study of nutritional status found some degree of malnutrition in about 20% of the patients.57 Another study examined, in addition to nutrition, body composition, and found a low muscle mass reserve in 51% of the patients (33% had normal weight).59
In addition, general preventive measures should be adopted, such as vaccination against pneumonia and influenza and a reduction in exposure to active or passive smoking.37
Corticoids
Although frequently used in clinical practice, the use of corticoids to treat PIBO is controversial because no clinical trials have confirmed their efficacy so far.28
Some authors recommend their use in the initial phases of the disease, before fibrosis is established.8,18 In a study that included 17 patients with BO, systemic corticoids were administered for at least 1 month, and 64.7% of the patients had clinical improvement, reduction of symptoms or increase in oxygen saturation. However, similar results were not repeated in other studies.17,18
There are reports of beneficial effects of pulse therapy with methylprednisolone (25 to 30 mg/day for 3 days) in the treatment of adenovirus AVB60 and in children with PIBO.37 In the latter case, oxygen saturation was normal 6 months after monthly administration of methylprednisolone.40 However, studies are scarce and no clinical trial has evaluated the efficacy of pulse therapy in children with PIBO.
Bronchodilators
The use of β-adrenergic agonists is controversial and should be based on the positive response to bronchodilator during pulmonary function tests and on clinical criteria.40,44 About 25% of the patients are estimated to benefit from this treatment.40
Antibiotics
In patients with bronchiectasis, antibiotics are often prescribed, particularly during exacerbations, to minimize the risk of new pulmonary lesions. The most frequently found germs are Haemophilus influenzae, Moraxella catarrhalis and S. pneumoniae,61 but the choice of antibiotics should be made according to culture and susceptibility tests of sputum, tracheal aspirate and BAL fluid. It is most often recommended for patients with non-localized bronchiectasis with no indication for surgery and for whom the control of infectious exacerbations may be critical to prevent more extensive lung damage. Courses of treatment with antibiotics during exacerbations are preferred to continuous use because of the risk of emergence of resistant bacterial forms. In the group in the city of Porto Alegre, Brazil, a trend towards colonization and infection by Pseudomonas was found when continuous antibiotic use was adopted. In the same group, inhaled antibiotics had to be administered to the patients infected by Pseudomonas (Fisher GB, personal communication).
Studies with lung transplant recipients with BO showed that azithromycin may have a beneficial effect because of its anti-inflammatory action, associated with the in vitro reduction of IL-8, IL-6 and TNF-α, IL-1β.54 Those patients had an improvement in their pulmonary function after 3 to 4 months of use of this macrolide on alternate days.62,63 Based on those results, some authors have recommended the use of azithromycin for PIBO,8 although no studies have evaluated its efficacy in this group of patients.
Other drugs
As most evidence shows that PIBO is mediated by immunological mechanisms, treatment options have focused on reducing inflammatory responses.5 In addition to corticoids, there are reports of the use of chloroquine and hydroxychloroquine17,37 without satisfactory results. However, the empirical monthly administration of IV immunoglobulin has been used as a treatment option in some cases.8,40
There is also a report of symptom improvement after the use of TNF-α monoclonal antibodies (infliximab) in children with BO after bone marrow transplants,64 but no studies have included patients with PIBO so far.
Surgery and lung transplant
Surgical resection might have to be chosen for patients with localized bronchiectasis areas with persistent purulent secretion and destruction of lung parenchyma or chronic atelectasis not controlled with clinical treatment.
The most severe forms of the disease, in which there is oxygen dependence, important physical limitations and pulmonary faction with severe reduction of expiratory flows, lung transplant is indicated. Some reports describe a satisfactory clinical progression and improvement of pulmonary function in three patients that received lung transplants.40
Prognosis
Studies with case series showed that PIBO progression is variable. In most cases, it seems to be chronic, non-progressive, in contrast with BO after Stevens-Johnson syndrome or bone marrow transplant.8 Most patients with PIBO tend to improve slowly and progressively. However, this clinical improvement may be associated with the growth of airways, and not necessarily resolution of inflammation.22 Moreover, this improvement is partial for most patients, and they still have mild symptoms, particularly during physical exercise, and abnormal clinical findings. In a series of 31 children followed up for a mean time of 3.5 years, 9.7% of the patients died and 67.7% still had symptoms or clinical signs.28 Morbidity associated with PIBO is high. Recurrent wheezing was seen in 42% of the patients in another study, as well as bronchiectasis in 32% and chest deformities in 21%.21
In another study, some patients developed chronic respiratory insufficiency and died. Different studies found mortality rates that range from 3.240 to 16.7%,18 which may result from the differences in severity of the patients included in each study.
The progression of pulmonary function is unclear. The general prognosis of pulmonary function was poor in most publications.5,9,28,41 Most reports showed that spirometry of patients followed up for a long time had the same characteristics of fixed obstructive disease of moderate to severe severity without response to bronchodilators. A study that evaluated children with adenovirus pneumonia found that 65% had an obstructive pattern in their spirometry results after about 13 years.12 Some reports reveal that the obstructive pattern is maintained the same along time,28 but there is also a slow and progressive fall of spirometric values.34 In a study with 11 children with PIBO, there was a fall of 1.02% per year in the FEV1/FVC, of 1.01% per year in FEV1 and of 1.04% per year in FEF25-75%.34
According to some authors, inflammation persists after the acute airway lesion, which might explain the persistence of symptoms,28 the recurrence of radiological changes,29 and the persistent changes in pulmonary function.34
Although the first description of BO was made over one century ago, most studies were conducted in the last 20 years, and no prospective studies have been conducted to define its long-term prognosis. However, it is known that support treatment contributes to reduce exacerbations and minimize the occurrence of new lung lesions, so that children may reach adulthood with a better quality of life. In a study conducted in Porto Alegre, Brazil, results of the Pediatric Qualify of Life Inventory (PedsQL) were similar for both patients with PIBO and controls, probably because the PIBO group had the continuous assistance of a multidisciplinary team. Patients excluded from the study because of irregular attendance to follow-up had severe impacts to their activities and a poorer progression.27
Final considerations
PIBO seems to be more frequent than it was believed in the past, and many cases are underdiagnosed. A significant number of children have persistent symptoms and obstructions demonstrated in the pulmonary function tests. Clinical trials designed to evaluate possible treatments, such as pulse therapy, may help to minimize pulmonary lesions and improve the prognosis for these patients, particularly if used in the early stages of the disease to reduce acute inflammation. As the number of study participants is usually small, multicenter studies should be conducted to better understand inflammation in this disease, risk factors, and the best treatment approaches. A current program, called Bronchiolitis Obliterans in Latin America (BOLAT) has been established to compare data from several Latin American centers and to conduct cooperative studies about interventions.
Acknowledgments
We thank Dr. Zilda Meira, of Hospital das Clínicas of UFMG, Professor Roussel, of the CHU Charles Nicolle, Rouen, France, and the pathological anatomy service of the School of Medicine of UFMG for the permission to use some of their images.
References
1. Schlesinger C, Meyer CA, Veeraraghavan S, Koss MN. Constrictive (obliterative) bronchiolitis: diagnosis, etiology, and a critical review of the literature. Ann Diagn Pathol. 1998;2:321-34.
2. Shaw RJ, Djukanovic R, Tashkin DP, Millar AB, du Bois RM, Orr PA. The role of small airways in lung disease. Respir Med. 2002;96:67-80.
3. Murtagh P, Giubergia V, Viale D, Bauer G, Pena HG. Lower respiratory infections by adenovirus in children. Clinical features and risk factors for bronchiolitis obliterans and mortality. Pediatr Pulmonol. 2009;44:450-6.
4. Gosink BB, Friedman PJ, Liebow AA. Bronchiolitis obliterans. Roentgenologic-pathologic correlation. Am J Roentgenol Radium Ther Nucl Med. 1973;117:816-32.
5. Colom AJ, Teper AM. Postinfectious bronchiolitis obliterans. Arch Argent Pediatr. 2009;107:160-7.
6. Swyer PR, James GC. A case of unilateral pulmonary emphysema. Thorax. 1953;8:133-6.
7. MacLeod WM. Abnormal transradiancy of one lung. Thorax. 1954;9:147-53.
8. Moonnumakal SP, Fan LL. Bronchiolitis obliterans in children. Curr Opin Pediatr. 2008;20:272-8.
9. Colom AJ, Teper AM, Vollmer WM, Diette GB. Risk factors for the development of bronchiolitis obliterans in children with bronchiolitis. Thorax. 2006;61:503-6.
10. Lobo AL, Guardiano M, Nunes T, Azevedo I, Vaz LG. Pos-infectious bronchiolitis obliterans in children. Rev Port Pneumol. 2007;13:495-509.
11. Wenman WM, Pagtakhan RD, Reed MH, Chernick V, Albritton W. Adenovirus bronchiolitis in Manitoba: epidemiologic, clinical, and radiologic features. Chest. 1982;81:605-9.
12. Sly PD, Soto-Quiros ME, Landau LI, Hudson I, Newton-John H. Factors predisposing to abnormal pulmonary function after adenovirus type 7 pneumonia. Arch Dis Child. 1984;59:935-9.
13. Kajon AE, Mistchenko AS, Videla C, Hortal M, Wadell G, Avendaño LF. Molecular epidemiology of adenovirus acute lower respiratory infections of children in the south cone of South America (1991-1994). J Med Virol. 1996;48:151-6.
14. Becroft DM. Bronchiolitis obliterans, bronchiectasis, and other sequelae of adenovirus type 21 infection in young children. J Clin Pathol. 1971;24:72-82.
15. Lang WR, Howden CW, Laws J, Burton JF. Bronchopneumonia with serious sequelae in children with evidence of adenovirus type 21 infection. Br Med J. 1969;1:73-9.
16. Hirschheimer M, Silva PS, Giudici R, Carrilho M, Mauad T, Ishida M. Simultaneous viral infection and childhood bronchiolitis obliterans. Braz J Infect Dis. 2002;6:146-8.
17. Yalçin E, Doğru D, Haliloğlu M, Ozçelik U, Kiper N, Göçmen A. Postinfectious bronchiolitis obliterans in children: clinical and radiological profile and prognostic factors. Respiration. 2003;70:371-5.
18. Hardy KA, Schidlow DV, Zaeri N. Obliterative bronchiolitis in children. Chest. 1988;93:460-6.
19. Chan PW, Muridan R, Debruyne JA. Bronchiolitis obliterans in children: clinical profile and diagnosis. Respirology. 2000;5:369-75.
20. Leong MA, Nachajon R, Ruchelli E, Allen JL. Bronchitis obliterans due to Mycoplasma pneumonia. Pediatr Pulmonol. 1997;23:375-81.
21. Chang AB, Masel JP, Masters B. Post-infectious bronchiolitis obliterans: clinical, radiological and pulmonary function sequelae. Pediatr Radiol. 1998;28:23-9.
22. Zhang L, Silva FA. Bronquiolite obliterante em crianças. J Pediatr (Rio J). 2000;76:185-92.
23. Milner AD, Murray M. Acute bronchiolitis in infancy: treatment and prognosis. Thorax. 1989;44:1-5.
24. Tortajada M, Gracia M, García E, Hernández R. Diagnostic considerations in unilateral hyperlucency of the lung (Swyer-James-MacLeod Syndrome). Allergol Immunopathol (Madr). 2004;32:265-70.
25. Wang W, Shen KL, Zeng JJ. Clinical studies of children with bronchiolitis obliterans. Zhonghua Er Ke Za Zhi. 2008;46:732-8.
26. Sulaiman A, Cavaille A, Vaunois B, Tiffet O. Swyer-James-MacLeod syndrome; repeated chest drainages in a patient misdiagnosed with pneumothorax. Interact Cardiovasc Thorac Surg. 2009;8:482-4.
27. Fischer GB, Sarria EE, Mattiello R, Mocelin HT, Castro-Rodriguez JA. Post infectious bronchiolitis obliterans in children. Paediatr Respir Rev. 2010;11:233-9.
28. Zhang L, Irion K, Kozakewich H, Reid L, Camargo JJ, da Silva Porto N, et al. Clinical course of postinfectious bronchiolitis obliterans. Pediatr Pulmonol. 2000;29:341-50.
29. Arce JD, Mondaca R, Mardones R, Velozo LF, Parra G. Secuelas post-infección por adenovirus en niños: Evaluación con tomografía computada. Rev Chil Radiol. 2002;8:154-63.
30. Mauad T, van Schadewijk A, Schrumpf J, Hack CE, Fernezlian S, Garippo AL, et al. Lymphocytic inflammation in childhood bronchiolitis obliterans. Pediatr Pulmonol. 2004;38:233-9.
31. Mistchenko AS, Lenzi HL, Thompson FM, Mota EM, Vidaurreta S, Navari C, et al. Participation of immune complexes in adenovirus infection. Acta Paediatr. 1992;81:983-8.
32. Mistchenko AS, Diez RA, Mariani AL, Robaldo J, Maffey AF, Bayley-Bustamante G, et al. Cytokines in adenoviral disease in children: association of interleukin-6, interleukin-8, and tumor necrosis factor alpha levels with clinical outcome. J Pediatr. 1994;124:714-20.
33. Koh YY, Jung DE, Koh JY, Kim JY, Yoo Y, Kim CK. Bronchoalveolar cellularity and interleukin-8 levels in measles bronchiolitis obliterans. Chest. 2007;131:1454-60.
34. Cazzato S, Poletti V, Bernardi F, Loroni L, Bertelli L, Colonna S, et al. Airway inflammation and lung function decline in childhood post-infectious bronchiolitis obliterans. Pediatr Pulmonol. 2008;43:381-90.
35. Costa CL, Spilborghs GM, Martins MA, Saldiva PH, Mauad T. Nitric acid-induced bronchiolitis in rats mimics childhood bronchiolitis obliterans. Respiration 2005;72:642-9.
36. Mauad T, Dolhnikoff M; São Paulo Bronchiolitis Obliterans Study Group. Histology of childhood bronchiolitis obliterans. Pediatr Pulmonol. 2002;33:466-74.
37. Camargos P, Champs N, Lasmar L, Fonseca MT, Fischer G. Bronchiolite oblitérante post-infectieuse. In: de Blic J, editor. Pneumologie Pédiatrique. Paris: Médecine-Sciences Flammarion; 2009. p. 72-76.
38. Chiu CY, Wong KS, Huang YC, Lin TY. Bronchiolitis obliterans in children: clinical presentation, therapy and long-term follow-up. J Paediatr Child Health. 2008,44:129-33.
39. Colom AJ, Teper AM. Clinical prediction rule to diagnose post-infectious bronchiolitis obliterans in children. Pediatr Pulmonol. 2009;44:1065-9.
40. Kim CK, Kim SW, Kim JS, Koh YY, Cohen AH, Deterding RR, et al. Bronchiolitis obliterans in the 1990s in Korea and the United States. Chest. 2001;120:1101-6.
41. Castro-Rodriguez JA, Daszenies C, Garcia M, Meyer R, Gonzales R. Adenovirus pneumonia in infants and factors for developing bronchiolitis obliterans: a 5-year follow-up. Pediatr Pulmonol. 2006;41:947-53.
42. Teel GS, Engeler CE, Tashijian JH, duCret RP. Imaging of small airways disease. Radiographics. 1996;16:27-41.
43. Yang CF, Wu MT, Chiang AA, Lai RS, Chen C, Tiao WM, et al.. Correlation of high-resolution CT and pulmonary function in bronchiolitis obliterans: a study based on 24 patients associated with consumption of Sauropus androgynus. AJR Am J Roentgenol. 1997;168:1045-50.
44. Teper A, Fischer GB, Jones MH. Seqüelas respiratórias de doenças virais: do diagnóstico ao tratamento. J Pediatr (Rio J). 2002;78 Suppl 2:S187-94.
45. Arakawa H, Webb WR. Air trapping on expiratory high-resolution CT scans in the absence of inspiratory scan abnormalities: correlation with pulmonary function tests and differential diagnosis. AJR Am J Roentgenol. 1998;170:1349-53.
46. Hansell DM, Rubens MB, Padley ST, Wells AU. Obliterative bronchiolitis: individual CT signs of small airway disease and functional correlation. Radiology. 1997;203:721-6.
47. Mocelin H, Fischer GB, Irion K. Densitovolumetry- correlation with pulmonary function test in children with bronchiolitis obliterans. Eur Resp J. 2004;24:A258.
48. Jensen SP, Lynch DA, Brown KK, Wenzel SE, Newell JD. High-resolution CT features of severe asthma and bronchiolitis obliterans. Clin Radiol. 2002;57:1078-85.
49. Brody AS. Computed Tomography of Pediatric Small Airways Disease. In: Boiselle PM, Lynch DA, editors. CT of the Airways. Totowa: Humana Press; 2008. p. 381-404.
50. Choi SJ, Choi BK, Kim HJ, Lee SH, Choi SH, Park SJ, et al. Lateral decubitus HRCT: a simple technique to replace expiratory CT in children with air trapping. Pediatr Radiol. 2002;32:179-82.
51. Yüksel H, Yilmaz O, Urk V, Yüksel D, Göktan C, Savaş R, et al. Clinical significance of lung perfusion defects in children with post-infectious bronchiolitis obliterans. Tuberk Toraks. 2009;57:376-82.
52. Mattiello R, Mallol J, Fischer GB, Mocelin HT, Rueda B, Sarria EE. Pulmonary function in children and adolescents with postinfectious bronchiolitis obliterans. J Bras Pneumol. 2010;36:453-9.
53. Teper AM, Kofman CD, Maffey AF, Vidaurreta SM. Lung function in infants with chronic pulmonary disease after severe adenoviral illness. J Pediatr. 1999;134:730-3.
54. Mattiello R, Sarria EE, Mallol J, Fischer GB, Mocelin H, Bello R, et al. Post-infectious bronchiolitis obliterans: can CT scan findings at early age anticipate lung function? Pediatr Pulmonol. 2010;45:315-9.
55. Yoo Y, Yu J, Kim DK, Choi SH, Kim CK, Koh YY. Methacholine and adenosine 5'-monophosphate challenges in children with post-infectious bronchiolitis obliterans. Eur Respir J. 2006;27:36-41.
56. Mattiello R, Sarria EE, Stein R, Fischer GB, Mocelin HT, Barreto SS, et al. Functional capacity assessment in children and adolescents with post-infectious bronchiolitis obliterans. J Pediatr (Rio J). 2008;84:337-43.
57. Mocelin HT, Fischer GB, Iriar KL, Cunha LS. A clinical follow-up on Bronchiolitis Obliterans in children. Rev Chil Pediatr. 2004;75:S12-17.
58. Champs NS. Bronquiolite obliterante pós-infecciosa: aspectos clínicos, tomográficos e funcionais; estudo comparativo entre crianças e adolescentes brasileiros e franceses. [Dissertação]. Programa de Pós-graduação em Ciências da Saúde - Saúde da criança e do adolescente: Universidade Federal de Minas Gerais; 2009.
59. Bosa VL, Mello ED, Mocelin HT, Benedetti FJ, Fischer GB. Assessment of nutritional status in children and adolescents with post-infectious bronchiolitis obliterans. J Pediatr (Rio J). 2008;84:323-30.
60. Takahashi I, Takahashi T, Tsuchida S, Mikami T, Saito H, Hatazawa C, et al. Pulse methylprednisolone therapy in type 3 adenovirus pneumonia with hypercytokinemia. Tohoku J Exp Med. 2006;209:69-73.
61. Santos RV, Rosário NA, Ried CA. Bronquiolite obliterante pós-infecciosa: aspectos clínicos e exames complementares de 48 crianças. J Bras Pneumol. 2004;30:20-5.
62. Gerhardt SG, McDyer JF, Girgis RE, Conte JV, Yang SC, Orens JB. Maintenance azithromycin therapy for bronchiolitis obliterans syndrome: results of a pilot study. Am J Respir Crit Care Med. 2003;168:121-5.
63. Yates B, Murphy DM, Forrest IA, Ward C, Rutherford RM, Fisher AJ, et al. Azithromycin reverses airflow obstruction in established bronchiolitis obliterans syndrome. Am J Respir Crit Care Med. 2005;172:772-5.
64. Fullmer JJ, Fan LL, Dishop MK, Rodgers C, Krance R. Successful treatment of bronchiolitis obliterans in a bone marrow transplant patient with tumor necrosis factor-α blockade. Pediatrics. 2005;116:767-70.
65. Furlan SP. Qualidade de vida em crianças e adolescentes com bronquiolite obliterante pós-infecciosa [Dissertação]. Programa de Pós-Graduação em Ciências Médicas - Pediatria: UFRGS; 2007.
66. Linares M, Meyer R, Soto G. Assessment of bronchodilator response in post- adenovirus infection patients. Rev Chil Pediatr. 2004;75;S37-44.
Manuscript submitted Oct 15 2010, accepted for publication Dec 15 2010.
Study submitted as dissertation to obtain the degree of Master in Adolescent and Children Health from the Graduate Program of Health Sciences, Universidade Federal de Minas Gerais, November 2009.
No conflicts of interest declared concerning the publication of this article.
Suggested citation: Champs NS, Lasmar LM, Camargos PA, Marguet C, Fischer GB, Mocelin HT. Post-infectious bronchiolitis obliterans in children. J Pediatr (Rio J). 2011;87(3):187-198.
- 1. Schlesinger C, Meyer CA, Veeraraghavan S, Koss MN. Constrictive (obliterative) bronchiolitis: diagnosis, etiology, and a critical review of the literature. Ann Diagn Pathol. 1998;2:321-34.
- 2. Shaw RJ, Djukanovic R, Tashkin DP, Millar AB, du Bois RM, Orr PA. The role of small airways in lung disease. Respir Med. 2002;96:67-80.
- 3. Murtagh P, Giubergia V, Viale D, Bauer G, Pena HG. Lower respiratory infections by adenovirus in children. Clinical features and risk factors for bronchiolitis obliterans and mortality. Pediatr Pulmonol. 2009;44:450-6.
- 4. Gosink BB, Friedman PJ, Liebow AA. Bronchiolitis obliterans. Roentgenologic-pathologic correlation. Am J Roentgenol Radium Ther Nucl Med. 1973;117:816-32.
- 5. Colom AJ, Teper AM. Postinfectious bronchiolitis obliterans. Arch Argent Pediatr. 2009;107:160-7.
- 6. Swyer PR, James GC. A case of unilateral pulmonary emphysema. Thorax. 1953;8:133-6.
- 7. MacLeod WM. Abnormal transradiancy of one lung. Thorax. 1954;9:147-53.
- 8. Moonnumakal SP, Fan LL. Bronchiolitis obliterans in children. Curr Opin Pediatr. 2008;20:272-8.
- 9. Colom AJ, Teper AM, Vollmer WM, Diette GB. Risk factors for the development of bronchiolitis obliterans in children with bronchiolitis. Thorax. 2006;61:503-6.
- 10. Lobo AL, Guardiano M, Nunes T, Azevedo I, Vaz LG. Pos-infectious bronchiolitis obliterans in children. Rev Port Pneumol. 2007;13:495-509.
- 11. Wenman WM, Pagtakhan RD, Reed MH, Chernick V, Albritton W. Adenovirus bronchiolitis in Manitoba: epidemiologic, clinical, and radiologic features. Chest. 1982;81:605-9.
- 12. Sly PD, Soto-Quiros ME, Landau LI, Hudson I, Newton-John H. Factors predisposing to abnormal pulmonary function after adenovirus type 7 pneumonia. Arch Dis Child. 1984;59:935-9.
- 13. Kajon AE, Mistchenko AS, Videla C, Hortal M, Wadell G, Avendaño LF. Molecular epidemiology of adenovirus acute lower respiratory infections of children in the south cone of South America (1991-1994). J Med Virol. 1996;48:151-6.
- 14. Becroft DM. Bronchiolitis obliterans, bronchiectasis, and other sequelae of adenovirus type 21 infection in young children. J Clin Pathol. 1971;24:72-82.
- 15. Lang WR, Howden CW, Laws J, Burton JF. Bronchopneumonia with serious sequelae in children with evidence of adenovirus type 21 infection. Br Med J. 1969;1:73-9.
- 16. Hirschheimer M, Silva PS, Giudici R, Carrilho M, Mauad T, Ishida M. Simultaneous viral infection and childhood bronchiolitis obliterans. Braz J Infect Dis. 2002;6:146-8.
- 18. Hardy KA, Schidlow DV, Zaeri N. Obliterative bronchiolitis in children. Chest. 1988;93:460-6.
- 19. Chan PW, Muridan R, Debruyne JA. Bronchiolitis obliterans in children: clinical profile and diagnosis. Respirology. 2000;5:369-75.
- 20. Leong MA, Nachajon R, Ruchelli E, Allen JL. Bronchitis obliterans due to Mycoplasma pneumonia. Pediatr Pulmonol. 1997;23:375-81.
- 21. Chang AB, Masel JP, Masters B. Post-infectious bronchiolitis obliterans: clinical, radiological and pulmonary function sequelae. Pediatr Radiol. 1998;28:23-9.
- 22. Zhang L, Silva FA. Bronquiolite obliterante em crianças. J Pediatr (Rio J). 2000;76:185-92.
- 23. Milner AD, Murray M. Acute bronchiolitis in infancy: treatment and prognosis. Thorax. 1989;44:1-5.
- 24. Tortajada M, Gracia M, García E, Hernández R. Diagnostic considerations in unilateral hyperlucency of the lung (Swyer-James-MacLeod Syndrome). Allergol Immunopathol (Madr). 2004;32:265-70.
- 25. Wang W, Shen KL, Zeng JJ. Clinical studies of children with bronchiolitis obliterans. Zhonghua Er Ke Za Zhi. 2008;46:732-8.
- 26. Sulaiman A, Cavaille A, Vaunois B, Tiffet O. Swyer-James-MacLeod syndrome; repeated chest drainages in a patient misdiagnosed with pneumothorax. Interact Cardiovasc Thorac Surg. 2009;8:482-4.
- 27. Fischer GB, Sarria EE, Mattiello R, Mocelin HT, Castro-Rodriguez JA. Post infectious bronchiolitis obliterans in children. Paediatr Respir Rev. 2010;11:233-9.
- 28. Zhang L, Irion K, Kozakewich H, Reid L, Camargo JJ, da Silva Porto N, et al. Clinical course of postinfectious bronchiolitis obliterans. Pediatr Pulmonol. 2000;29:341-50.
- 29. Arce JD, Mondaca R, Mardones R, Velozo LF, Parra G. Secuelas post-infección por adenovirus en niños: Evaluación con tomografía computada. Rev Chil Radiol. 2002;8:154-63.
- 30. Mauad T, van Schadewijk A, Schrumpf J, Hack CE, Fernezlian S, Garippo AL, et al. Lymphocytic inflammation in childhood bronchiolitis obliterans. Pediatr Pulmonol. 2004;38:233-9.
- 31. Mistchenko AS, Lenzi HL, Thompson FM, Mota EM, Vidaurreta S, Navari C, et al. Participation of immune complexes in adenovirus infection. Acta Paediatr. 1992;81:983-8.
- 32. Mistchenko AS, Diez RA, Mariani AL, Robaldo J, Maffey AF, Bayley-Bustamante G, et al. Cytokines in adenoviral disease in children: association of interleukin-6, interleukin-8, and tumor necrosis factor alpha levels with clinical outcome. J Pediatr. 1994;124:714-20.
- 33. Koh YY, Jung DE, Koh JY, Kim JY, Yoo Y, Kim CK. Bronchoalveolar cellularity and interleukin-8 levels in measles bronchiolitis obliterans. Chest. 2007;131:1454-60.
- 34. Cazzato S, Poletti V, Bernardi F, Loroni L, Bertelli L, Colonna S, et al. Airway inflammation and lung function decline in childhood post-infectious bronchiolitis obliterans. Pediatr Pulmonol. 2008;43:381-90.
- 35. Costa CL, Spilborghs GM, Martins MA, Saldiva PH, Mauad T. Nitric acid-induced bronchiolitis in rats mimics childhood bronchiolitis obliterans. Respiration 2005;72:642-9.
- 36. Mauad T, Dolhnikoff M; São Paulo Bronchiolitis Obliterans Study Group. Histology of childhood bronchiolitis obliterans. Pediatr Pulmonol. 2002;33:466-74.
- 37. Camargos P, Champs N, Lasmar L, Fonseca MT, Fischer G. Bronchiolite oblitérante post-infectieuse. In: de Blic J, editor. Pneumologie Pédiatrique. Paris: Médecine-Sciences Flammarion; 2009. p. 72-76.
- 38. Chiu CY, Wong KS, Huang YC, Lin TY. Bronchiolitis obliterans in children: clinical presentation, therapy and long-term follow-up. J Paediatr Child Health. 2008,44:129-33.
- 39. Colom AJ, Teper AM. Clinical prediction rule to diagnose post-infectious bronchiolitis obliterans in children. Pediatr Pulmonol. 2009;44:1065-9.
- 40. Kim CK, Kim SW, Kim JS, Koh YY, Cohen AH, Deterding RR, et al. Bronchiolitis obliterans in the 1990s in Korea and the United States. Chest. 2001;120:1101-6.
- 41. Castro-Rodriguez JA, Daszenies C, Garcia M, Meyer R, Gonzales R. Adenovirus pneumonia in infants and factors for developing bronchiolitis obliterans: a 5-year follow-up. Pediatr Pulmonol. 2006;41:947-53.
- 42. Teel GS, Engeler CE, Tashijian JH, duCret RP. Imaging of small airways disease. Radiographics. 1996;16:27-41.
- 43. Yang CF, Wu MT, Chiang AA, Lai RS, Chen C, Tiao WM, et al.. Correlation of high-resolution CT and pulmonary function in bronchiolitis obliterans: a study based on 24 patients associated with consumption of Sauropus androgynus. AJR Am J Roentgenol. 1997;168:1045-50.
- 44. Teper A, Fischer GB, Jones MH. Seqüelas respiratórias de doenças virais: do diagnóstico ao tratamento. J Pediatr (Rio J). 2002;78 Suppl 2:S187-94.
- 45. Arakawa H, Webb WR. Air trapping on expiratory high-resolution CT scans in the absence of inspiratory scan abnormalities: correlation with pulmonary function tests and differential diagnosis. AJR Am J Roentgenol. 1998;170:1349-53.
- 46. Hansell DM, Rubens MB, Padley ST, Wells AU. Obliterative bronchiolitis: individual CT signs of small airway disease and functional correlation. Radiology. 1997;203:721-6.
- 47. Mocelin H, Fischer GB, Irion K. Densitovolumetry- correlation with pulmonary function test in children with bronchiolitis obliterans. Eur Resp J. 2004;24:A258.
- 48. Jensen SP, Lynch DA, Brown KK, Wenzel SE, Newell JD. High-resolution CT features of severe asthma and bronchiolitis obliterans. Clin Radiol. 2002;57:1078-85.
- 49. Brody AS. Computed Tomography of Pediatric Small Airways Disease. In: Boiselle PM, Lynch DA, editors. CT of the Airways. Totowa: Humana Press; 2008. p. 381-404.
- 50. Choi SJ, Choi BK, Kim HJ, Lee SH, Choi SH, Park SJ, et al. Lateral decubitus HRCT: a simple technique to replace expiratory CT in children with air trapping. Pediatr Radiol. 2002;32:179-82.
- 52. Mattiello R, Mallol J, Fischer GB, Mocelin HT, Rueda B, Sarria EE. Pulmonary function in children and adolescents with postinfectious bronchiolitis obliterans. J Bras Pneumol. 2010;36:453-9.
- 53. Teper AM, Kofman CD, Maffey AF, Vidaurreta SM. Lung function in infants with chronic pulmonary disease after severe adenoviral illness. J Pediatr. 1999;134:730-3.
- 54. Mattiello R, Sarria EE, Mallol J, Fischer GB, Mocelin H, Bello R, et al. Post-infectious bronchiolitis obliterans: can CT scan findings at early age anticipate lung function? Pediatr Pulmonol. 2010;45:315-9.
- 55. Yoo Y, Yu J, Kim DK, Choi SH, Kim CK, Koh YY. Methacholine and adenosine 5'-monophosphate challenges in children with post-infectious bronchiolitis obliterans. Eur Respir J. 2006;27:36-41.
- 56. Mattiello R, Sarria EE, Stein R, Fischer GB, Mocelin HT, Barreto SS, et al. Functional capacity assessment in children and adolescents with post-infectious bronchiolitis obliterans. J Pediatr (Rio J). 2008;84:337-43.
- 57. Mocelin HT, Fischer GB, Iriar KL, Cunha LS. A clinical follow-up on Bronchiolitis Obliterans in children. Rev Chil Pediatr. 2004;75:S12-17.
- 58. Champs NS. Bronquiolite obliterante pós-infecciosa: aspectos clínicos, tomográficos e funcionais; estudo comparativo entre crianças e adolescentes brasileiros e franceses. [Dissertação]. Programa de Pós-graduação em Ciências da Saúde - Saúde da criança e do adolescente: Universidade Federal de Minas Gerais; 2009.
- 59. Bosa VL, Mello ED, Mocelin HT, Benedetti FJ, Fischer GB. Assessment of nutritional status in children and adolescents with post-infectious bronchiolitis obliterans. J Pediatr (Rio J). 2008;84:323-30.
- 60. Takahashi I, Takahashi T, Tsuchida S, Mikami T, Saito H, Hatazawa C, et al. Pulse methylprednisolone therapy in type 3 adenovirus pneumonia with hypercytokinemia. Tohoku J Exp Med. 2006;209:69-73.
- 61. Santos RV, Rosário NA, Ried CA. Bronquiolite obliterante pós-infecciosa: aspectos clínicos e exames complementares de 48 crianças. J Bras Pneumol. 2004;30:20-5.
- 62. Gerhardt SG, McDyer JF, Girgis RE, Conte JV, Yang SC, Orens JB. Maintenance azithromycin therapy for bronchiolitis obliterans syndrome: results of a pilot study. Am J Respir Crit Care Med. 2003;168:121-5.
- 63. Yates B, Murphy DM, Forrest IA, Ward C, Rutherford RM, Fisher AJ, et al. Azithromycin reverses airflow obstruction in established bronchiolitis obliterans syndrome. Am J Respir Crit Care Med. 2005;172:772-5.
- 65. Furlan SP. Qualidade de vida em crianças e adolescentes com bronquiolite obliterante pós-infecciosa [Dissertação]. Programa de Pós-Graduação em Ciências Médicas - Pediatria: UFRGS; 2007.
- 66. Linares M, Meyer R, Soto G. Assessment of bronchodilator response in post- adenovirus infection patients. Rev Chil Pediatr. 2004;75;S37-44.
Correspondence:
Publication Dates
-
Publication in this collection
27 Sept 2011 -
Date of issue
June 2011
History
-
Received
15 Oct 2010 -
Accepted
15 Dec 2010